Abstract
Layered materials embrace rich intercalation reactions to accommodate high concentrations of foreign species within their structures, and find many applications spanning from energy storage, ion exchange to secondary batteries. Light alkali metals are generally most easily intercalated due to their light mass, high charge/volume ratio and in many cases strong reducing properties. An evolving area of materials chemistry, however, is to capture metals selectively, which is of technological and environmental significance but rather unexplored. Here we show that the layered telluride T2PTe2 (T=Ti, Zr) displays exclusive insertion of transition metals (for example, Cd, Zn) as opposed to alkali cations, with tetrahedral coordination preference to tellurium. Interestingly, the intercalation reactions proceed in solid state and at surprisingly low temperatures (for example, 80 °C for cadmium in Ti2PTe2). The current method of controlling selectivity provides opportunities in the search for new materials for various applications that used to be possible only in a liquid.
Similar content being viewed by others
Introduction
Intercalation compounds allow incorporation or exchange of foreign atoms or molecules into the voids of various topologies in the host lattices such as cages (zeolites and so on)1, channels (h-WO3 and so on)2 and two-dimensional (2D) spaces (graphite and so on)3 and represent an important frontier in solid state chemistry. The structural diversity of these solids gives rise to a vast array of applications too extensive to summaries. However, two key areas of particular note are those materials serving as reservoirs to store and release alkali metal ions, for example, Li+ for high performance for energy storage devices4 and the tuning of exotic superconductivity in NaxCoO2·1.3H2O, Lix(THF)yHfNCl (THF, tetrahydrofuran) and CuxBi2Se3 upon intercalation5,6,7.
The rich intercalation chemistry that has been discovered for layered materials with van der Waals (vdW) interactions, which includes V2O5, MNCl (M=Ti, Zr), MX2 (M=Ti, Zr, Ta and so on; X=S, Se), MPX3 (M=Mg, Fe, Ni and so on; X=S, Se), MoS2 and MOX (M=Ti, V, Fe and so on; X=Cl, Br)8,9,10,11,12, yields various chemical and physical properties. Rather weak interlayer (vdW) interactions and a flexible interlayer spacing allows for incorporation of not only the lowest charged small alkali metal cations, but also many other heavier metals in the periodic table. For instance, TaS2 is capable of intercalating alkali metals, alkali earth metals and nearly all 3d transition metals as well as organic amines13,14,15,16,17,18,19. However, the poor selectivity in these materials hinders the preferential sorption of heavy metals, which is of environmental significance in the remediation of important toxic heavy metals pollutants, such as Cd, Pb and Hg20. Traditional absorbents and ion-exchangers like activated carbon, clays and zeolites also suffer from this problem21,22,23.
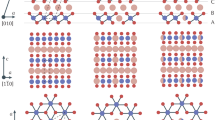
Recently, Kanatzidis and colleagues have demonstrated that several layered sulfides exhibit highly selective ion-exchange properties for Sr, Hg, Pd and Cd within K2xMnxSn3–xS6 (0.5<x<0.95; refs 24, 25) and Cs within [(CH3)2NH2]2Ga2Sb2S7·H2O (ref. 26), via an aqueous solutions with excess lighter alkali metal ions and protons. In both systems, the high selectivity is ascribed to the favourable interactions between the soft Lewis base S2– ions of the host layer and the soft Lewis acid of the metal ions as a guest. In this study, we utilize the smaller electronegativity of tellurium to provide a softer Lewis base, compared with O, S and Se27, within the layered tellurides, Ti2PTe2 and Zr2PTe2, which we expect will promote a higher degree of covalency to selectively bind heavy metals. Prior intercalation studies are largely confined to binary tellurides such as TiTe2, ZrTe2 and IrTe2, with a primary interest in superconductivity and ferromagnetism28,29. The structure of T2PTe2 (T=Ti, Zr) is built up of slabs of hexagonal closed-packed triple Te–P–Te layers with T atom being octahedrally coordinated by three Te and three P atoms (see Fig. 1a)30,31. The individual slabs are stacked such that a rhombohedral structure is formed. It is isostructural with 3R-type Ta2CS2 (ref. 32). The absence of metal species between the double Te layers suggests relatively weak interlayer interactions and thus intercalation chemistry.
(a) T2PTe2 (3R-Ta2CS2 type with space group R-3m) and (b) metal-intercalated MxT2PTe2 (1T-Ta2CS2 type with space group P-3m1), where black, blue, yellow and purple spheres, respectively denote Ti/Zr, P, Te and M atoms. A, B, C are used to represent anion (Te, P) stacking arrangement, while α, β, γ represent the Ti/Zr stacking sequence. The unit cells are shown by the dotted lines. (c) Coordination environment around an octahedral void (dotted circle) in the 3R structure. (d) Coordination environment around a tetrahedral void in the 1T structure. In MxT2PTe2, the voids are partially occupied by M (for example, ∼20% for Zn). (e) Crystal structure of TiTe2 and MX2 compounds in general (M=Ti, Zr, Ta and so on; X=S, Se).
Herein, we show that T2PTe2 displays exclusive insertion of transition metals of Cu, Zn and Cd with tetrahedral coordination preference to tellurium. The intercalation reactions proceed in solid state and at surprisingly low temperatures—as low as 80 °C for cadmium in Ti2PTe2 and 70 °C for copper in Zr2PTe2. The metal intercalation induces a structural transition involving the change in stacking sequence from the 3R- to the 1T-type. Our first-principles calculations demonstrate a unique band structure with a pseudogap just above the Fermi energy, implying that the selective intercalation originates predominantly from the thermodynamic stabilization of the intercalated phase. The observed low temperature solution-free metal capturing, together with the reversible reaction enabling separation of hazardous metal from a powder metal mixture or an alloy, suggest potential applicability of our materials in environmental remediation.
Results
Metal intercalation and structural characterization
We tested a series of alkali, alkali earth, transition and post-transition metals (M) for their intercalation properties into Ti2PTe2. The reactions were typically conducted by heating a pelletized mixture of Ti2PTe2 and M in vacuum at temperatures up to 400 °C (see the ‘Methods’ section for a detailed synthesis procedure). Powder X-ray diffraction patterns collected on the samples after attempted intercalation revealed little or no intercalation in most cases, including alkali metals, as summarized in Supplementary Fig. 1. These reactions either lead to decomposition of Ti2PTe2 or no reaction was observed. In contrast, Zn, Cu and Cd readily intercalated. Fe and Mn intercalation occurred only at high temperature (400 °C), yet leaving a large amount of Ti2PTe2 unreacted (Supplementary Fig. 2). Zr2PTe2 shows the same reaction result, except the fact that no intercalation was observed for Fe and Mn (Supplementary Fig. 1).
As a representative example, we first show the intercalation of Zn into Ti2PTe2. The product prepared at 300 °C for 48 h with a Zn/Ti2PTe2 molar ratio (p) of 0.4 exhibits a similar X-ray diffraction profile to the parent phase (Fig. 2). However, a close inspection of the pattern shows a significant shift of the (00l) reflections. Moreover, a clear change in stacking sequence from the rhombohedral to a primitive cell is evident from the appearance of new characteristic reflections. Here, the lattice constants for the precursor (R) and product (P) phases are correlated by the relation: aP≈aR, 3cP≈cR. The clear expansion of the normalized c axis (cP–cR/3=0.348 Å) suggests successful intercalation of Zn, presumably between the double Te layers. As shown in Supplementary Fig. 3, energy dispersive X-ray spectroscopy (EDX) measurements show that the Zn content is approximately x≈0.36(5) (in ZnxTi2PTe2). The identical Bravais lattice change from R to P along with the elongated normalized c parameter is also observed for Cd (Fig. 2), Cu, Fe and Mn in Ti2PTe2 (Supplementary Fig. 2), and Cd, Zn and Cu in Zr2PTe2 (Supplementary Fig. 4). We note that Fe and Mn intercalated phases for T=Ti were not obtained as a single phase, but together with a large amount of unreacted Ti2PTe2. The lattice parameters of the precursor and the intercalated compounds are shown in Table 1 and the results of elemental analysis by SEM/EDX are shown in Supplementary Figs 5 and 6.
For a more precise structural characterization of ZnxTi2PTe2, synchrotron X-ray diffraction and neutron diffraction Rietveld refinements were carried out. Here, we recall that Ta2CS2 has two polymorphs with different stacking sequences, the 3R-type (R–3m) being isostructural with Ti2PTe2 and the 1T-type (P–3m1), as displayed in Fig. 1a,b, respectively33. Both forms of Ta2CS2 are capable of accepting various foreign cations, but since the stacking sequences are different, intercalated cations find themselves in different coordination environments. For example, Fe and Co in 3R-MxTa2CS2 (M=Fe, Co) partially occupy octahedral voids (site 6c), whereas Cu in 1T-MxTa2CS2 occupies tetrahedral voids (site 2d)33,34,35, as illustrated in Supplementary Fig. 7. Given the observed unit cell change in Ti2PTe2 upon Zn intercalation, it is likely that ZnxTi2PTe2 also adopts the 1T-CuxTa2CS2 structure. Therefore, this structure was adopted as a starting model for a Rietveld refinement, by placing Zn at the 2d (1/3, 2/3, z) site (Fig. 1b,d). The synchrotron pattern also shows minor impurities, TiP and ZnTe, which were included in the refinement. The refinements converged comfortably with Rwp=7.51% and Rp=5.20% for synchrotron X-ray diffraction and Rwp=6.36% and Rp=4.95% for neutron diffraction (see Fig. 3a,b and Supplementary Table 1). The refined Zn composition of x=0.40 is close to the value obtained from the EDX measurements. Placing Zn at the octahedral 1b site (Supplementary Fig. 7) did not lead to better convergence. Likewise, the synchrotron X-ray diffraction refinement for CuxTi2PTe2, ZnxZr2PTe2 and CdxZr2PTe2 revealed the occupation of Cu, Zn and Cd at the tetrahedral (2d) site and a composition of x=0.282(2), 0.337(6) and 0.194(5) (Fig. 3d, Supplementary Fig. 8 and Supplementary Table 2).
Refined (a) Synchrotron X-ray diffraction and (b) neutron diffraction patterns of Zn0.4Ti2PTe2, showing observed (red), calculated (green) and difference (blue) profiles. The upper, middle and lower ticks represent the positions of the calculated Bragg reflections of Zn0.4Ti2PTe2, TiP and ZnTe, respectively. Inset of a shows the result of MEM analysis, where a strong Zn–Te bond along the c axis was observed. (c) Fourier transforms of the extended X-ray absorption fine structure (EXAFS) spectrum (blue) at the Zn K-edge of Zn0.4Ti2PTe2, which is compared with calculated Fourier transforms of (red) the anisotropic tetrahedron based on the neutron diffraction refined crystal structure, (dotted line) the equidistant tetrahedron and (dashed line) octahedron. (d) Refined synchrotron X-ray diffraction pattern of Cu0.28Ti2PTe2. The upper, middle and lower ticks represent the positions of the calculated Bragg reflections of Cu0.28Ti2PTe2, TiP and Cu, respectively.
For Ti2PTe2, the refined atomic coordinate z for M is slightly different between Zn and Cu. The Cu position (z=0.452) is nearly at the centre of the Te4 tetrahedron. In contrast, the Zn position (z=0.4347) is slightly off-centred as found in 1T-CuxTa2CS2 (ref. 34), giving Zn–Te distances of 2.287 Å ( × 1) and 2.517 Å ( × 3). The former value is shorter than those of common phases such as ZnTe (2.643 Å) and ZnAl2Te4 (2.578 Å; refs 36, 37). This is rather curious, but a number of techniques support this observation. The charge density distributions around Zn obtained by a MEM analysis shows a strong covalent Zn–Te bond (due to the smaller electronegativity of tellurium), indicative of the single short Zn–Te distance (Fig. 3a). Extended X-ray absorption fine structure analysis at the Zn K-edge (Fig. 3c, Supplementary Fig. 9 and Supplementary Table 3) also led to a better fit with the Rietveld structure incorporating the long/short Zn–Te bonds, as opposed to an isotropic model where all four Zn–Te bonds are the same length. The octahedral model (Zn at the 1d site) also failed to reproduce the experimental data. The off-centring of Zn may arise to reduce repulsion from the Ti located on the top of Zn (see Fig. 1d).
The reversibility of the intercalation process is demonstrated by regeneration of the original Ti2PTe2 host upon exposure to I2 in acetonitrile at room temperature, as determined by X-ray diffraction (Fig. 2). The I2 reaction gives rise to extra tiny peaks such as at 27.8°, which can be attributed to Te metal. This possibly results from the decomposition of ZnTe, a tiny impurity phase already present in Ti2PTe2. For the initial Zn intercalation, we further examined the reaction products while varying the Zn/Ti2PTe2 molar ratio p (0.1≤p≤0.5), with a fixed reaction temperature and time (300 °C, 48 h). For p=0.1, the 3R phase coexists with traces of the 1T phase. With increasing p, the amount of the latter phase increases, whereas the former decreases, and the lattice constants of the rhombohedral phase do not change (Supplementary Fig. 10). In other words, there is no uptake of guest ions in the original 3R phase and Zn intercalation readily induces structural transition and the phase separation. This has been confirmed by SEM/EDX results (Supplementary Fig. 3).
Theoretical calculations
As discussed above, upon intercalation 3R-T2PTe2 transforms to the 1T polytype, and metals amenable to intercalation (Cd, Zn, Cu, Fe, Mn for T=Ti and Cd, Zn, Cu for T=Zr) are less electropositive and favour tetrahedral coordination with Te38. However, no intercalation of alkali metals was observed. Such intercalation selectivity in Ti2PTe2 is quite unprecedented and clearly different from other layer materials based on oxide, sulphide and chloride with weaker interlayer vdW bonding, such as V2O5, MNCl, MX2 (X=S, Se) and MPX3 (X=S, Se)8,9,10,11. Apparently, the use of less electronegative Te layers (versus O, S, Se and so on) hampers alkali metals intercalation. Interestingly, the related telluride TiTe2 is known to accommodate Li and Rb, together with transition metals28,39,40,41,42. Namely, the intercalation selectivity of TiTe2 and ZrTe2 can be greatly enhanced by Ti2PTe2 and Zr2PTe2. This clear difference in intercalativity may originate from the difference in crystal structure and electronic structure. Compared with the structure of TTe2 having TTe6 octahedra (Fig. 1e), T2PTe2 possesses an additional P layer, thus providing an anisotropic octahedral coordination of TTe3P3 (Fig. 1a,b). An additional notable feature in T2PTe2—being distinct from TTe2 and other intercalation materials with vdW layers—is the presence of one extra electron per formula in the conduction band, which is described by the nominal charge configuration: (T4+)2(P3−)(Te2−)2(e−), though this and intercalated compounds are basically covalent in nature. This is evidenced by X-ray absorption near edge structure (XANES) results showing the Ti4+ state and fairly good metallic conductivity (ρ=40 μΩ) at room temperature31.
Calculations based on density functional theory have revealed unique features in the electronic structures and bonding character of this material. The results show that 3R-Ti2PTe2 is indeed metallic with a nearly half-filled electron Fermi surface, as illustrated in Fig. 4 and Supplementary Fig. 11. This metallic feature is in sharp contrast to TiTe2, where hole and electron Fermi surfaces are compensated and a semimetallic-like electronic state is realized43. Despite the two-dimensional crystal structure in Ti2PTe2, the Fermi surface has a three-dimensionally deformed shape (Supplementary Fig. 11b) and the estimated Fermi velocity is vx=2.70 × 107 cm s−1 and vz=1.83 × 107 cm s−1, giving rise to a rather small anisotropy (vz/vx)=0.68. States around the Fermi energy consist mainly of Ti d orbitals hybridized with Te p orbitals, as found in the partial density of states (DOS) in Supplementary Fig. 11. Moreover, the sizable Te–Te hybridization accounts for the emergence of the three-dimensionality of the Fermi surface. Interestingly, the calculated DOS for the dummy ‘host’ material 1T-Ti2PTe2 share similar features with those of 3R-Ti2PTe2, such as a pseudogap structure (valley) just above the Fermi energy (Fig. 4), naively implying that electron doping up to the pseudogap possibly stabilizes the intercalated phases within a rigid band picture.
Total energy calculations for ZnxTi2PTe2 with different Zn concentrations (0≤x≤1), intercalation sites (tetrahedral or octahedral) and ‘host’ structures (3R or 1T) provide useful information on the phase stability of the present system (see Fig. 4). Calculated heats of formation Ef=E(1T-ZnxTi2PTe2)−E(3R-Ti2PTe2)−xE(Zn) in the 1T phase show very small positive values (in the order of 10 meV) up to x=0.5, implying that an entropic term may stabilize the telluride upon intercalation of Zn. This dopant level is fairly consistent with the experimentally obtained value of ∼0.4. Further electron doping beyond the pseudogap results in the appearance of a peak around the Fermi energy (see the x=1 case in Fig. 4), indicating non-bonding electrons. On the other hand, the Zn intercalation in the 3R phase gives rise to a much larger  (in the order of 100 meV) even with a small dopant concentration such as x=1/3, and the DOS exhibit a peak structure around the Fermi energy as shown in Fig. 4, indicative of the deviation from the rigid band picture. The DOS peak is composed mainly of Ti-d and Zn-s orbitals, preventing the Zn intercalation. As for ZnxZr2PTe2, qualitative features in the calculated electronic structure are quite similar to ZnxTi2PTe2, showing even smaller (still positive) heats of formation in the 1T phase.
(in the order of 100 meV) even with a small dopant concentration such as x=1/3, and the DOS exhibit a peak structure around the Fermi energy as shown in Fig. 4, indicative of the deviation from the rigid band picture. The DOS peak is composed mainly of Ti-d and Zn-s orbitals, preventing the Zn intercalation. As for ZnxZr2PTe2, qualitative features in the calculated electronic structure are quite similar to ZnxTi2PTe2, showing even smaller (still positive) heats of formation in the 1T phase.
To obtain further insight into the metal selectivity, we performed the first principles calculations for 1T-M0.25Ti2PTe2 (M=Cr, Mn, Fe, Co, Ni, Cu, Zn and Cd) and 1T-M0.25Zr2PTe2 (M=Ni, Cu, Zn and Cd) with M sitting on the tetrahedral interstitial site, and estimated the heat of formation, defined as Ef=E(1T-M0.25Ti2PTe2)−E(3R-Ti2PTe2)−0.25E(M). As shown in Fig. 5, a positive but very small value of Ef for Zn intercalated both in Ti2PTe2 and Zr2PTe2 is clearly seen, while marginally small values are achieved for M=Ni, Cu and Cd compared with the intercalated systems with M=Cr, Mn, Fe and Co, which reasonably confirms the experimentally observed metal selectivity. These results demonstrate that the thermodynamic stability of the intercalated compound is the decisive factor in determining the selectivity. We note that unintercalated Ni has similarly small values of Ef=+0.06 eV (Ti2PTe2) and +0.04 eV (Zr2PTe2), the reason of which is not clear but may be related to kinetic aspects of reactions (diffusion of metal, surface reactions and so on).
Towards practical reaction conditions
A remarkable aspect in Ti2PTe2 and Zr2PTe2 is the facile uptake of heavy metals at mild temperatures and in the solid state. We tested the effect of reaction temperature for M=Zn and Cd, with a fixed reaction time of 48 h. Shown in Fig. 6 and Supplementary Fig. 12 is the fraction of the intercalated (1T) phase as a function of reaction temperature. In the case of Ti2PTe2, the volume fraction of Cd-intercalated phase readily increases at temperatures above 50 °C and reaches almost 100% at 150 °C. In contrast, a higher temperature of 200 °C is necessary to attain a full uptake of Zn. The Zr2PTe2 system has quantitatively a similar tendency in intercalation behaviour, but the required temperatures, 190 °C (Cd) and 220 °C (Zn), are higher than those in Ti2PTe2. In Ti2PTe2, Cu requires 300 °C and Fe and Mn requires >400 °C, given that the same reaction period of 48 h is applied. Furthermore, the reaction of Zr2PTe2 with Cu at 70 °C for 20 days yielded a single phase of 1T-CuxZr2PTe2 (Supplementary Fig. 13). These results suggest that this type of intercalation could be further expanded, with a versatile potential to control the reactivity and selectivity.
Reaction temperature dependence of the volume fraction of (a) MxTi2PTe2 and (b) MxZr2PTe2 where M=Zn (red) and Cd (blue). Both the samples were heated together with and elemental metal (Zn or Cd) at various temperatures for 48 h. The molar volume fraction was estimated from the Rietveld refinement. Solid lines are drawn as a guide for visualization. The error bars are smaller than the size of the symbols.
Discussion
The difference in the intercalation temperature makes Ti2PTe2 and Zr2PTe2 of interest as adsorbents to chemically separate Cd from other metals simply by tailoring the temperature. In many forms, Cd has been (or was) widely used and discharged from the electroplating industry, electrical contact devices, nickel cadmium batteries and pigments44,45. Traditional metal separation procedures (smelting, precipitation, ion exchange and solvent extraction) are solution-based46. As shown in Supplementary Figs 14 and 15, test reactions of Ti2PTe2 with an equimolar mixture of Cd-Mn at 100 °C for 96 h and Cd-Ti at 300 °C for 48 h yielded only the Cd intercalated material. Selective intercalation of Cd-containing alloys might also be possible. Reactions with commercially available alloys, Cu0.75Pb0.25 and Cu0.80Sn0.20, at 300 °C, 48 h resulted in only Cu accommodation into Ti2PTe2 as shown in Supplementary Fig. 16. The present solution-free approach may find a new route for capturing Cd and other toxic elements. It is noteworthy that the vacuum environment is not a requirement; reactions in ambient conditions yielded the same result (see Supplementary Fig. 17). What we showed here is a prototype application in environmental remediation. Given the fact that all the current technologies for metal capturing are based on solution chemistry, our demonstration of the selective solid-state metal capturing (in particular, the Cd capturing above 80 °C for Ti2PTe2) is the first of its kind in inorganic layered materials, opening new possibilities for applications to solve environmental issues.
In addition, the observed reactivity and highly mobile nature of the heavy metals at very low temperatures suggests a possibility to develop multivalent ion conductors at significantly lowered working temperatures. This compound represents a member of a larger structure family as in Ta2CS2, with possibilities for improved performance as well as new properties. We believe that layered compounds with less electronegative anions such as telluride and antimonide could provide fertile ground for the development of exotic functionalities.
Methods
Materials synthesis
The stoichiometric polycrystalline samples of the host material Ti2PTe2 and Zr2PTe2 were prepared by the conventional solid state reaction method with excess P to compensate its loss due to sublimation above 500 °C. Ti/Zr (Kojundo Chemical, 3N), P (Kojundo Chemical, 2N) and Te (Kojundo Chemical, 3N) powders were mixed in the molar ratio of 2: 1.1: 2 and pelletized in a nitrogen-filled glove box. The obtained pellet was sealed into an evacuated silica tube (<10−2 Pa) and heated to 850 °C at a rate of 20 °C h−1, and annealed for 24 h. To ensure full incorporation of both phosphorus and tellurium, a slow cooling rate of 2 °C h−1 was applied from 850 to 400 °C before rapid cooling to room temperature.
The low temperature reactions of Ti2PTe2 and Zr2PTe2 with various elemental metals (M=Li, Na, Mg, Al, Si, K, Ca, Sc, Ti, V, Cr, Mn, Fe, Co, Ni, Cu, Zn, Ga, Ge, Sr, Y, Zr, Nb, Mo, Ru, Pd, Ag, Cd, In, Sn, Hf, Ta, Ir, Hg, Pb) were performed in the solid state. Ti2PTe2 and the metal powder as received (Kojundo Chemical, 4N) were mixed with various molar ratios (typically 1: 1) and pelletized in a nitrogen-filled glove box. The pellet was sealed in an evacuated silica tube (<10−2 Pa) and heated at temperatures between 100 °C and 400 °C for 48–96 h. Reactions with equimolar mixtures of Cd and Ti and of Cd and Mn in Ti2PTe2 were conducted, respectively, at 300 °C for 48 h, and at 100 °C for 96 h. Also, reactions with Cu0.75Pb0.25 alloy and with Cu0.80Sn0.20 alloy (Kojundo Chemical, 3N) were performed at 300 °C for 48 h. Furthermore, for lithium intercalation, 1 M n-butyl lithium solution in hexane was used and the reaction allowed to proceed for 72 h. Deintercalation of Zn was performed at room temperature by mixing Zn0.4Ti2PTe2 with I2 dissolved in acetonitrile at the molar ratio of 1: 1 for 2 days.
X-ray and neutron diffraction
Laboratory powder X-ray diffraction were collected using a Bruker D8 diffractometer with Cu Kα radiation. The diffraction data for structural refinement were recorded in a 2θ range from 5° to 80° with a step interval of 0.02°. The diffraction pattern for Ti2PTe2 was fit using the space group R–3m and the cell parameters a=3.63949(6) Å and c=28.4885(4) Å, in agreement with reported values in the literature27. High resolution synchrotron X-ray diffraction experiments were performed on ZnxTi2PTe2, CuxTi2PTe2, ZnxZr2PTe2 and CdxZr2PTe2 at room temperature on a Debye-Scherrer camera installed at beamline BL02B2, SPring-8. The incident beam from a bending magnet was monochromatized to 0.35479(1) Å. The powder samples were loaded into a glass capillary (0.1 mm inner diameter) and rotated during measurements to reduce preferential orientation. The diffraction data were recorded in a 2θ range from 0° to 60° with a step interval of 0.01°. Powder neutron diffraction measurements were carried out at room temperature on an approximately 2 g sample at BT-1 (λ=1.5403 Å), National Institute of Standards and Technology.
Structural analysis
The obtained X-ray and neutron data were analysed by the Rietveld method using the RIETAN-FP program47. The agreement indices used were Rp=Σ|yio−yic|/Σyio, Rwp=[Σwi(yio−yic)2/Σwi(yio)2]1/2 and the goodness of fit, χ2=[Rwp/Rexp]2 where Rexp=[(N−P)/Σwiyio2]1/2, yio and yic are the observed and calculated intensities, wi is the weighting factor, N is the total number of yio data when the background is refined and P is the number of adjusted parameters. The energy dispersive X-ray spectroscopy (EDX) measurements were performed using an Oxford Instruments IE-250 detector attached to a scanning electron microscope (SEM, HITACHI S-3400N). For each composition, 10–40 randomly selected spots were examined.
For X-ray absorption spectroscopy (XAS) measurements, Zn0.4Ti2PTe2 was homogeneously dispersed in dried boron nitride powder and pelletized. Zn K-edge spectra were recorded in transmission mode at beam line BL01B1 in SPring-8, Japan with a double-crystal Si(111) monochrometer. The energy scale was calibrated using Cu foil. Data were collected at room temperature. Extended X-ray absorption fine structure (EXAFS) analysis was performed using REX2000 data analysis software, with the theoretical backscattering phases and amplitudes calculated with the code FEFF8 (ref. 48). Radial structure functions were obtained using Fourier transformation of the oscillations between 3.0 and 14.0 Å. To obtain local structural parameters, inverse Fourier transforms were calculated from the radial structure functions between 1.688 and 2.700 Å. Curve fitting was performed in k space.
First principles calculations
First principles density functional theory calculations are performed with all-electron full-potential linearized augmented plane wave method in the scalar-relativistic scheme. Fractional intercalation is simulated by assuming super cell models appropriate to given concentrations. Lattice constants and internal atomic positions are fully optimized by calculating total energy and atomic forces with preserving the original crystal symmetry.
Data availability
The data that support the findings of this study are available from the corresponding author upon request.
Additional information
How to cite this article: Yajima, T. et al. Selective and low temperature transition metal intercalation in layered tellurides. Nat. Commun. 7, 13809 doi: 10.1038/ncomms13809 (2016).
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
Blanchard, G., Maunaye, M. & Martin, G. Removal of heavy metals from waters by means of natural zeolites. Water. Res. 18, 1501–1507 (1984).
Lee, K. S., Seo, D. K. & Whangbo, M. H. Electronic band structure study of the anomalous electrical and superconducting properties of hexagonal alkali tungsten bronzes AxWO3 (A= K, Rb, Cs). J. Am. Chem. Soc. 119, 4043–4049 (1997).
Dresselhaus, M. S. & Dresselhaus, G. Intercalation compounds of graphite. Adv. Phys. 51, 1–186 (2002).
Goodenough, J. B. & Park, K. S. The Li-ion rechargeable battery: a perspective. J. Am. Chem. Soc. 135, 1167–1176 (2013).
Takada, K. et al. Superconductivity in two-dimensional CoO2 layers. Nature 422, 53–55 (2003).
Yamanaka, S., Hotehama, K. I. & Kawaji, H. Superconductivity at 25.5K in electron-doped layered hafnium nitride. Nature 392, 580–582 (1998).
Sasaki, S. et al. Topological superconductivity in CuxBi2Se3 . Phys. Rev. Lett. 107, 217001 (2011).
Bruce, D. W. & O'Hare, D. Inorg. Mater. Wiley (1996).
Yamanaka, S. Intercalation and superconductivity in ternary layer structured metal nitride halides (MNX: M= Ti, Zr, Hf; X= Cl, Br, I). J. Mater. Chem. 20, 2922–2933 (2010).
Whittingham, M. S. Chemistry of intercalation compounds: metal guests in chalcogenide hosts. Prog. Solid State Chem. 12, 41–99 (1978).
Clement, R. A novel route to intercalation into layered MnPS3 . J. Chem. Soc. Chem. Commun. 14, 647–648 (1980).
Herber, R. H. & Maeda, Y. Intercalation compounds of iron (III) oxychloride: systematics of nitrogen-containing Lewis base intercalants. Inorg. Chem. 20, 1409–1415 (1981).
Rouxel, J. in Intercalated Layered Materials 201–250Springer (1979).
Lerf, A. & Schöllhorn, R. Solvation reactions of layered ternary sulfides AxTiS2, AxNbS2, and AxTaS2 . Inorg. Chem. 16, 2950–2956 (1977).
Omloo, W. P. F. A. M. & Jellinek, F. Intercalation compounds of alkali metals with niobium and tantalum dichalcogenides. J. Less Common Metals 20, 121–129 (1970).
Di Salvo, F. J., Hull, G. W. Jr, Schwartz, L. H., Voorhoeve, J. M. & Waszczak, J. V. Metal intercalation compounds of TaS2: preparation and properties. J. Chem. Phys. 59, 1922–1929 (1973).
Gamble, F. R., Osiecki, J. H. & DiSalvo, F. J. Some superconducting intercalation complexes of TaS2 and substituted pyridines. J. Chem. Phys. 55, 3525–3530 (1971).
Schöllhorn, R. & Zagefka, H. D. Demonstration of the ionic structure of the intercalation compound TaS2 NH3 . Angew. Chem. Int. Ed. 16, 199–200 (1977).
Schöllhorn, R. Intercalation chemistry. Physica B C 99, 89–99 (1980).
Shannon, M. A. et al. Science and technology for water purification in the coming decades. Nature 452, 301–310 (2008).
Huang, C. P. & Blankenship, D. W. The removal of mercury (II) from dilute aqueous solution by activated carbon. Water. Res. 18, 37–46 (1984).
Benhammou, A., Yaacoubi, A., Nibou, L. & Tanouti, B. Adsorption of metal ions onto Moroccan stevensite: kinetic and isotherm studies. J. Colloid Interface Sci. 282, 320–326 (2005).
Kesraoui-Ouki, S., Cheeseman, C. R. & Perry, R. Natural zeolite utilisation in pollution control: a review of applications to metals' effluents. J. Chem. Technol. Biotechnol. 59, 121–126 (1994).
Manos, M. J., Ding, N. & Kanatzidis, M. G. Layered metal sulfides: exceptionally selective agents for radioactive strontium removal. Proc. Natl Acad. Sci. USA 105, 3696–3699 (2008).
Manos, M. J. & Kanatzidis, M. G. Sequestration of heavy metals from water with layered metal sulfides. Chem. Eur. J. 15, 4779–4784 (2009).
Ding, N. & Kanatzidis, M. G. Selective incarceration of caesium ions by Venus flytrap action of a flexible framework sulfide. Nat. Chem 2, 187–191 (2010).
Allred, A. L. Electronegativity values from thermochemical data. J. Inorg. Nucl. Chem. 17, 215–221 (1961).
Baranov, N. V. et al. Ferromagnetism and structural transformations caused by Cr intercalation into TiTe2 . J. Phys. Condens. Matter 21, 506002 (2009).
Kamitani, M. et al. Superconductivity in CuxIrTe2 driven by interlayer hybridization. Phys. Rev. B 87, 180501 (2013).
Philipp, F., Schmidt, P., Milke, E., Binnewies, M. & Hoffmann, S. Synthesis of the titanium phosphide telluride Ti2PTe2: a thermochemical approach. J. Solid State Chem. 181, 758–767 (2008).
Philipp, F., Schmidt, P., Ruck, M., Schnelle, W. & Isaeva, A. The layered metal Ti2PTe2 . J. Solid State Chem. 181, 2859–2863 (2008).
Beckmann, O., Boller, H. & Nowotny, H. Die Kristallstrukturen von Ta2S2C und Ti4S5 (Ti0.81S). Monatsh. Chem. 101, 945–955 (1970).
Boller, H. & Sobczak, R. Einlagerungsphasen vom TypMex[Ta2S2C]. Monatsh. Chem. 102, 1226–1233 (1971).
Brec, R., Ritsma, J., Ouvrard, G. & Rouxel, J. Alkali metal intercalates of tantalum carbide sulfide. Inorg. Chem. 16, 660–665 (1977).
Suzuki, M., Suzuki, I. S. & Walter, J. Magnetism and superconductivity in McTa2S2C (M=Fe, Co, Ni, and Cu). Phys. Rev. B 71, 224407 (2005).
Singh, H. P. & Dayal, B. Lattice parameters and thermal expansion of zinc telluride and mercury selenide. Acta Cryst. A 26, 363–364 (1970).
Hahn, H., Frank, G., Klingler, W., Störger, A. D. & Störger, G. Untersuchungen über ternäre Chalkogenide. VI. Über Ternäre Chalkogenide des Aluminiums, Galliums und Indiums mit Zink, Cadmium und Quecksilber. Z. Anorg. Allg. Chem. 279, 241–270 (1955).
The Inorganic Crystal Structure Database (ICSD). http://www.fiz-informationsdienste.de/en/DB/icsd/index.html.
Patel, S. N. & Balchin, A. A. Structural studies of lithium intercalated titanium di-telluride. J. Mater. Sci. Lett. 4, 382–384 (1985).
Shorikov, D. O., Titov, A. N., Titova, S. G. & Tolochko, B. P. Structural parameters of intercalation compounds based on titanium dichalcogenides near the temperature of the polaronic band collapse. Nucl. Instrum. Methods Phys. Res. A 470, 215–218 (2001).
Yarmoshenko, Y. M. et al. Spatial dependence of the dichroism of photoemission of Fe1/4TiTe2 upon excitation with circularly polarized radiation. Phys. Solid State 50, 2190–2198 (2008).
Cybulski, Z., Feltz, A. & Andratschke, M. Structure and properties of quaternary chalcogenides. Mater. Res. Bull. 24, 157–162 (1989).
Claessen, R. et al. Complete band-structure determination of the quasi-two-dimensional Fermi-liquid reference compound TiTe2 . Phys. Rev. B 54, 2453–2465 (1996).
Salim, R., Al-Subu, M. M. & Sahrhage, E. Uptake of cadmium from water by beech leaves. J. Environ. Sci. Health A27, 603–627 (1992).
Cheung, C. W., Porter, J. F. & McKay, G. Elovich equation and modified second-order equation for sorption of cadmium ions onto bone char. J. Chem. Technol. Biotechnol. 75, 963–970 (2000).
Chang, Y. H., Hsieh, K. H. & Chang, F. C. Removal of Hg2+ from aqueous solution using a novel composite carbon adsorbent. J. Appl. Polym. Sci. 112, 2445–2454 (2009).
Izumi, F. & Momma, K. Three-dimensional visualization in powder diffraction. Solid State Phenom. 130, 15–20 (2007).
Rehr, J. J. & Albers, R. C. Theoretical approaches to X-ray absorption fine structure. Rev. Mod. Phys. 72, 621–654 (2000).
Acknowledgements
The work was supported by CREST and JSPS KAKENHI (JP16H6439, JP16H6440, JP16H6441, JP16H02267). The early stage of the work was supported by FIRST. We thank C. Tassel (Kyoto University), J. Kim, N. Tsuji and A. Fujiwara (JASRI) for their help during the SPring-8 experiments.
Author information
Authors and Affiliations
Contributions
T.Yaj. and H.K. conceived and designed the study. M.K., W.Y., D.K. and Y.Z. performed the synthesis, laboratory X-ray diffraction and SEM/EDX experiments. M.K., W.Y., T.Yaj. and Y.K. obtained synchrotron X-ray diffraction, while M.A.G. and H.K. obtained the neutron data. The structural refinement was performed by T.Yam., T.Yaj., W.Y. and M.K. Y.O. and Y.U. obtained X-ray absorption spectra and analysed the data. T.O. conducted first principles calculations. All the authors discussed the results. Y.Z., T.Yaj., T.O. and H.K. wrote the manuscript, with comments from Y.K. M.K., Y.O., W.Y. and M.A.G.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
Supplementary figures and supplementary tables (PDF 6428 kb)
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Yajima, T., Koshiko, M., Zhang, Y. et al. Selective and low temperature transition metal intercalation in layered tellurides. Nat Commun 7, 13809 (2016). https://doi.org/10.1038/ncomms13809
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/ncomms13809
This article is cited by
-
Intercalation-driven ferroelectric-to-ferroelastic conversion in a layered hybrid perovskite crystal
Nature Communications (2022)
-
Expanding frontiers in materials chemistry and physics with multiple anions
Nature Communications (2018)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.