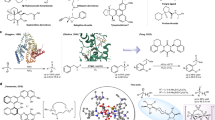
Abstract
Catalytic use of peroxomolybdate for asymmetric transformations has attracted increasing attention due to its catalytic properties and application in catalysis. Herein, we report chiral bisguanidinium dinuclear oxodiperoxomolybdosulfate [BG]2+[(μ-SO4)Mo2O2(μ-O2)2(O2)2]2− ion pair, as a catalyst for enantioselective sulfoxidation using aqueous H2O2 as the terminal oxidant. The ion pair catalyst is isolatable, stable and useful for the oxidation of a range of dialkyl sulfides. The practical utility was illustrated using a gram-scale synthesis of armodafinil, a commercial drug, with the catalyst generated in situ from 0.25 mol% of bisguanidinium and 2.5 mol% of Na2MoO4·2H2O. Structural characterization of this ion pair catalyst has been successfully achieved using single-crystal X-ray crystallography.
Similar content being viewed by others
Introduction
Metalloenzymes containing molybdenum, responsible for nitrogen and sulfur metabolism1,2,3,4, continue to fuel interest in the exploration of novel molybdenum complexes with catalytic activities. Various neutral coordination complexes of Mo(VI) containing chiral organic ligands have been synthesized and studied extensively for enantioselective reactions5,6,7,8,9,10,11. In particular, one interesting example of highly enantioselective sulfoxidation of alkyl aryl sulfides was demonstrated using a complex derived from MoO2(acac)2 and chiral bis-hydroxamic acids12. In contrast to their neutral counterparts, there have been no successful attempts to utilize peroxomolybdate13 for asymmetric reactions, even though many of these species have been comprehensively characterized.
It is well known that peroxomolybdates are formed on the treatment of molybdate salts with aqueous H2O2 oxidant14,15. Monomeric, oligomeric and polymeric peroxomolybdate species could be generated under similar conditions (Fig. 1)14. The addition of different organic ligands can further increase the structural and functional diversity of peroxomolybdate complexes16,17,18. Other ligands such as silanol19, phosphate20, arsenate21 and sulfate22,23,24,25 have been used to bridge molybdates to construct dinuclear or trinuclear peroxomolybdate complexes. Peroxomolybdates species have been shown to be excellent catalysts for the oxidation of numerous substrates, including alkenes, alcohols26 and sulfides. The complexity of peroxomolybdates is thus recognized to be a challenging obstacle for elaborating them into highly enantioselective catalysts.
We have recently developed pentanidium27,28,29,30 and dicationic bisguanidinium (BG) as efficient phase-transfer31,32 and ion pair catalysts33,34,35,36,37. We have utilized bisguanidinium permanganate ion pair catalyst for the enantioselective oxidation of alkenes38. The precise stereocontrol exhibited by bisguanidinium encouraged us to explore other anionic metallic species for asymmetric transformations39. Herein, we describe our serendipitous discovery of chiral bisguanidinium dinuclear oxodiperoxomolybdosulfate [BG]2+[(μ-SO4)Mo2O2(μ-O2)2(O2)2]2− ion pair catalyst (Fig. 2). This ion pair catalyst is stable and isolatable or it can be generated in situ. In a continuation of our current efforts towards developing practical approaches to enantiopure sulfoxides29,40,41, we report a simple and scalable methodology for enantioselective sulfoxidation using this ion pair catalyst42,43,44,45.
Results
Catalytic application of molybdate in sulfoxidation
At the onset of this work, we realized that we were unable to approach enantiopure 2-sulfinyl esters through enantioselective alkylation of sulfenate anion, as the reaction was incompatible with α-halogenated carboxylates (Supplementary Fig. 1)29. We were attracted to the low cost and easy accessibility of molybdate salts and thus we attempted to investigate the direct sulfoxidation of 2-sulfanyl acetate, by utilizing a catalytic amount of molybdate salts and aqueous H2O2 as terminal oxidant. Methyl 2-(benzhydrylsulfanyl)acetate 2a was chosen as the model substrate (Table 1), since 2-sulfinyl acetate 3a could be easily transformed to armodafinil, a commercial drug used for the treatment of narcolepsy and shift work sleep disorder46,47,48.
When the reaction was performed in the presence of 1 mol% of (S,S)-1a, 5 mol% of (NH4)6Mo7O24·4H2O and 1.05 equiv. 35% aqueous H2O2, poor yield and no enantioselectivity were observed (Table 1, entry 1). With the addition of acetic acid49, the enantioselectivity was slightly improved, albeit with low yield (entries 2 and 3). Using trifluoroacetic acid as additive, a marked enhancement of the reactivity was achieved, but with negligible enantioselectivity (entry 4). With the addition of sodium or potassium hydrogen sulfate, we observed significant improvement of yield, as well as enantioselectivity (entries 5 and 6)50,51. Switching to other additives, such as dihydrogen phosphate or hydrogen phosphate led to poor results (entries 7 and 8). Further investigation of reaction parameters (entries 9–14), such as the source of molybdate, solvent and stoichiometry of KHSO4, led to the enhancement of reactivity, as well as enantioselectivity (entry 14, 93% ee; Supplementary Table 1). The optimal condition was established by lowering the temperature to 0 °C and using just 1 mol% of (S,S)-1a together with 2.5 mol% Na2MoO4·2H2O in iPr2O, affording 2-sulfinyl acetate 3a in 99% yield with 94% ee (entry 15). The absolute configuration of 3a was confirmed to be S through comparison with the reported data46.
Substrate scope of various sulfides using (S,S)-1a
The reaction scope was examined using a series of substrates with a relatively low reactivity, in which the electron density of sulfur is reduced, due to strong electron-withdrawing groups like ester, ketone and nitrile (Table 2). The reactions performed efficiently and were generally completed within 1 h. For benzyl 2-sulfanylacetates with different substituents on the aromatic ring, dialkyl sulfoxides 3b–3i were obtained in high yields and excellent enantioselectivities. Sulfoxide 3j bearing 2-thienyl was obtained in high yield and good enantioselectivity without oxidation at the thiophene. With a slight variation of reaction conditions, using 0.25 equivalent of KHSO4 and 2.5 mol% of K2MoO4, various aromatic 2-sulfanylacetates were efficiently converted to alkyl aryl sulfoxides 3k–3q in high yields with good enantioselectivities. For the oxidation of sulfide-bearing para-OMe substituent, leading to sulfoxide 3l, slight over-oxidation to sulfone was observed. For a less reactive substrate 2r, the reaction was conducted at room temperature using 1.5 equiv. H2O2, affording sulfoxide 3r with good enantioselectivity. With less favourable substrates such as tert-butyl substituted 2-sulfanyl acetate 2s, low enantioselective induction was observed (Table 2, 3s).
To further explore the scope, a diverse range of substrates bearing different functional groups were examined (Table 2, 3t–3y). 3-Sulfinyl propanoate 3t was produced with excellent enantioselectivity. Sulfoxides 3u–3y bearing amide, ketone, acrylate, nitrile and aldehyde moieties were furnished with good to excellent enantioselectivities. The absolute configurations of 3f and 3o were confirmed to be R and S, respectively, using single-crystal X-ray diffraction; thus, absolute configurations of sulfoxides 3 were assigned by analogy to either 3f or 3o. The practical utility was successfully demonstrated using a gram-scale synthesis of (R)-modafinil (armodafinil), a commercial drug, using 0.25 mol% of (R,R)-1a (Fig. 3).
Identification and characterization of ion pair (R,R)-1b
We attempted to identify the reactive catalytic species by mimicking the reaction conditions in the absence of sulfide substrate (Fig. 4). After a simple workup procedure, (R,R)-1b was isolated and a single crystal suitable for X-ray diffraction was grown by vapour diffusion of Et2O into a dimethylformamide (DMF) solution of (R,R)-1b. The structure of (R,R)-1b was fully characterized using X-ray analysis (Fig. 2b), 95Mo nuclear magnetic resonance (NMR) (Fig. 5b) and fourier transform-infrared spectroscopy (FT-IR) (Supplementary Fig. 2).
The achiral anionic metallic species [(μ-SO4)Mo2O2(μ-O2)2(O2)2]2− is revealed by X-ray crystallography to be embedded within the chiral cavity formed by two side arms of the chiral bisguanidinium dication (Fig. 2b). The coordination geometry surrounding the Mo was clearly elucidated (Fig. 5a). The SO42− ligand plays a crucial role in constructing the dimeric symmetric structure. Each Mo centre comprises one bridging peroxo ligand, one side-on peroxo group and a terminal oxo ligand, with the sulfate group acting as a bipodal ligand to the two Mo atoms. Each Mo atom is 7-coordinated with oxygen atoms in a pentagonal bipyramidal arrangement. The two associated pentagonal bipyramids share one edge [O9–O10] and the two Mo atoms are connected by two μ–η1:η2 peroxo-bridges, [O8–O9 and O11–O10]. Both Mo1–O5 and Mo2–O12 bonds have the same length (1.659(7) Å) that falls in a typical range for the Mo=O bond. Generally, the bridging peroxo O8–O9 (1.482(9) Å) and O10–O11 (1.473(10) Å) bond lengths are slightly longer than the other side-on peroxo O6–O7 (1.458(10) Å) and O13–O14 (1.467(10) Å) bond lengths. 95Mo NMR spectrum of (R,R)-1b was also obtained in DMF-d7 at 22 °C, using 2 M Na2MoO4·2H2O solution in D2O as an external reference (assigned to 0 p.p.m.). The chemical shift at −199.3 p.p.m. is characteristic of oxodiperoxomolybdate species (Fig. 5b)52.
We found that (R,R)-1b (1.0 equiv.), prepared using the method in Fig. 4, can be used directly as the oxidant for sulfoxidation, without additional aqueous H2O2, providing sulfoxide ent-3a in 90% yield and 80% ee in 0.5 h (Fig. 6a, equation 1). This result indicates that (R,R)-1b is the actual oxidizing specie providing high enantiodiscrimination. Utilizing 0.25 equiv. (R,R)-1b led to the formation of ent-3a in 50% yield in 24 h with 31% ee (Fig. 6a, equation 2), demonstrating that two out of four peroxo moieties on (R,R)-1b are active oxygen donors, as two equivalent of active oxygen from (R,R)-1b are transferred to the sulfides53. The second oxygen transfer is slower and less enantioselective than the first. As expected, (R,R)-1b can be used catalytically in the presence of H2O2, providing sulfoxide ent-3a in 95% yield with 91% ee at a loading of 1 mol% (Fig. 6b, equation 3). This result is comparable to the reaction, in which this active catalyst is prepared in situ from (R,R)-1a (Fig. 3). The catalyst can be recycled from the reaction (Fig. 6b, equation 3) and used for a second round of reaction, but an additional amount of 0.5 equiv. of KHSO4 must be added to restore the reactivity and enantioselectivity (Fig. 6b, equation 4).
Computational studies of (R,R)-1b using the ONIOM method revealed that a more stable ion-pairing arrangement, with the distance between the anionic cluster and the cationic bisguanidinium noticeably reduced compared with that in the crystal structure (Fig. 7). As a result of this rearrangement, approach of the substrate to most of the peroxo-oxygen atoms is obstructed by bisguanidinium and sulphate groups. Only one of side-on peroxo-oxygen atoms (marked as O14 in Fig. 7) remains accessible for reaction. This is consistent with the experimentally observed high enantioselectivity, since restricted access to secondary reaction sites will result in a reaction with higher selectivity.
Structure of (R,R)-1b obtained from ONIOM geometry optimization, where atoms are color-coded as follows: C (grey), N (blue), H (white), S (yellow), O (red) and Mo (pink). The displayed NCI surface of bisguanidinium indicates interactions between the anionic cluster [(μ-SO4)Mo2O2(μ-O2)2(O2)2] and bisguanidinium.
Reaction selectivity was evaluated using 50 mol% tetrabutylammonium hydrogen sulfate (nBu4NHSO4) as an achiral ion-pairing reagent (Fig. 8). A high level of enantiocontrol can still be achieved, which indicates ion pairing interaction between chiral bisguanidinium and dinuclear oxodiperoxomolybdosulfate anion [(μ-SO4)Mo2O2(μ-O2)2(O2)2]2− accelerates the reaction rate significantly over tetrabutylammonium, promoting the reaction through the desired asymmetric pathway. The stereoinduction observed in the current methodology may be ascribed to ion pairing interaction and other non-covalent interactions between dicationic bisguanidinium, oxodiperoxomolybdosulfate anion, as well as the substrates in the stereoselectivity determining transition state (Supplementary Fig. 5)33,54.
Discussion
In the present study, we have described the first catalytic use of peroxomolybdate for enantioselective sulfoxidation; a series of enantioenriched dialkyl sulfoxides and alkyl aryl sulfoxides have been obtained using inexpensive molybdates and aqueous H2O2 through a simple experimental protocol. The practical value of current methodology was demonstrated using a gram-scale synthesis of armodafinil, a commercial drug, with a low loading (0.25 mol%) of bisguanidinium. The ‘active’ catalyst is isolatable, stable and has been identified to be bisguanidinium dinuclear oxodiperoxomolybdosulfate ion pair [BG]2+[(μ-SO4)Mo2O2(μ-O2)2(O2)2]2−. Its structure is also unambiguously confirmed by X-ray analysis.
Methods
General information
The synthesis of sulfide substrates 2 are provided in Supplementary Note 1. For the details of mechanistic studies, see Supplementary Figs 3–5, Supplementary Tables 1–2 and Supplementary Note 2. For details of X-ray analysis, see Supplementary Figs 6–9, Supplementary Methods and Supplementary Data 2–9. For details of computational studies, see Supplementary Figs 10–13, Supplementary Tables 3–5, Supplementary Methods and Supplementary Data 1. For the 1H, 13C, 19F and 95Mo NMR data, and spectra of the compounds in the article, see Supplementary Figs 14–70 and Supplementary Methods. For the high-performance liquid chromatography spectra of sulfoxide product 3 and 4 in this article, see Supplementary Figs 71–96.
Preparation of ion pair (R,R)-1b
To the solution of Na2MoO4·2H2O (24.1 mg, 0.1 mmol) dissolved in 1 M H2SO4 (1 ml), 35% H2O2 (345 μl, 4.0 mmol) was added dropwise to give a yellow solution at room temperature. Then the above solution was added dropwise to a solution of (R,R)-1a (56.4 mg, 0.04 mmol) in Et2O (2 ml). After vigorously stirring for 15 min, a pale yellow precipitate was formed in the Et2O layer. After further stirring for 2 h and removal of Et2O by evaporation, 4 ml deionized water was added and the resulting heterogeneous mixture was submitted to ultrasound for 1 min. Then the pale yellow solid was filtered off and washed with deionized water (40 ml). After drying over concentrated H2SO4 under vacuum at room temperature, (R,R)-1b was obtained as a pale yellow powder (65.5 mg, 91% yield) and its structure was characterized and determined by X-ray single-crystal diffraction. Increase of the amount of Na2MoO4·2H2O to 0.1 equivalent or replacement of 1 M H2SO4 with 0.5 equivalent of solid KHSO4 all led to the formation of identical complex (R,R)-1b, which is confirmed by X-ray diffraction analysis.
General procedure for synthesis of sulfoxides 3
A 10 ml round-bottom flask (RBF) was charged with a solution of sulfide 2 (0.2 mmol) and bisguanidinium phase-transfer catalyst (S,S)-1a (2.8 mg, 0.002 mmol) in iPr2O (4 ml). Then Na2MoO4·2H2O (1.2 mg, 0.005 mmol) and KHSO4 (13.6 mg, 0.1 mmol) were added. The reaction mixture was stirred for 5 min in an ice bath, and then aqueous 35% H2O2 (18.1 μl, 0.21 mmol) was added in one portion. The resulting mixture was stirred vigorously at 0 °C and monitored by thin-layer chromatography until 2 was completely consumed. Purification using silica gel column chromatography (CH2Cl2/EtOAc=2:1) afforded the desired sulfoxide 3. Minor changes in the amount of KHSO4 and choice of molybdate salt (K2MoO4) and solvent (nBu2O) were conducted for some substrates to achieve slightly better enantioselectivity.
Gram-scale experiment for synthesis of armodafinil 4
A 250 ml round-bottom flask was charged with a solution of methyl 2-(benzhydrylthio)acetate 2a (1.36 g, 5 mmol) and bisguanidinium phase-transfer catalyst (R,R)-1a (17.6 mg, 0.0125, mmol) in nBu2O (100 ml). Then Na2MoO4·2H2O (30 mg, 0.125 mmol), KHSO4 (340 mg, 2.5 mmol) and 35% aq. H2O2 (453 μl, 5.25 mmol) were added at room temperature. The resulting mixture was stirred vigorously and monitored by thin-layer chromatography and 2a was completely consumed within 8 h. Purification using silica gel column chromatography (CH2Cl2/EtOAc=2:1) afforded the sulfoxide product ent -3a with R configuration, 1.32 g, 91% yield, 91% ee. Then the obtained sulfoxide (1.32 g, 4.58 mmol) was treated with 2 M ammonical methanol (23 ml, 10.0 equiv.) and the resulting solution was stirred at room temperature for 24 h. Purification using silica gel column chromatography (CH2Cl2/MeOH=20:1) afforded 4 as a white solid, 1.19 g, 95% yield, 91% ee.
Data availability
CCDC 1456987-1456990 contain the supplementary crystallographic data for this paper. These data can be obtained free of charge from The Cambridge Crystallographic Data Centre via www.ccdc.cam.ac.uk/data_request/cif. The data that support the findings of this study are available from the corresponding authors on request.
Additional information
How to cite this article: Zong, L. et al. Bisguanidinium dinuclear oxodiperoxomolybdosulfate ion pair-catalyzed enantioselective sulfoxidation. Nat. Commun. 7, 13455 doi: 10.1038/ncomms13455 (2016).
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
Burgess, B. K. & Lowe, D. J. Mechanism of molybdenum nitrogenase. Chem. Rev. 96, 2983–3012 (1996).
Schwarz, G., Mendel, R. R. & Ribbe, M. W. Molybdenum cofactors, enzymes and pathways. Nature 460, 839–847 (2009).
Hille, R., Hall, J. & Basu, P. The mononuclear molybdenum enzymes. Chem. Rev. 114, 3963–4038 (2014).
Nishibayashi, Y. Recent progress in transition-metal-catalyzed reduction of molecular dinitrogen under ambient reaction conditions. Inorg. Chem. 54, 9234–9247 (2015).
Yamada, S., Mashiko, T. & Terashima, S. (Acetylacetonato)[(-)-N-alkylephedrinato]dioxomolybdenum, a new class of chiral chelate complexes which catalyze asymmetric epoxidation of allylic alcohol. J. Am. Chem. Soc. 99, 1988–1990 (1977).
Kagan, H. B., Mimoun, H., Mark, C. & Schurig, V. Asymmetric epoxidation of simple olefins with an optically active molybdenum(VI) peroxo complex. Angew. Chem. Int. Ed. 18, 485–486 (1979).
Trost, B. M. & Lautens, M. Molybdenum catalysts for allylic alkylation. J. Am. Chem. Soc. 104, 5543–5545 (1982).
Fujimura, O. & Grubbs, R. H. Asymmetric ring-closing metathesis: kinetic resolution catalyzed by a chiral molybdenum alkylidene complex. J. Am. Chem. Soc. 118, 2499–2500 (1996).
Barlan, A. U., Basak, A. & Yamamoto, H. Enantioselective oxidation of olefins catalyzed by a chiral bishydroxamic acid complex of molybdenum. Angew. Chem. Int. Ed. 45, 5849–5852 (2006).
Malcolmson, S. J., Meek, S. J., Sattely, E. S., Schrock, R. R. & Hoveyda, A. H. Highly efficient molybdenum-based catalysts for enantioselective alkene metathesis. Nature 456, 933–937 (2008).
Brito, J. A., Royo, B. & Gomez, M. An overview of chiral molybdenum complexes applied in enantioselective catalysis. Catal. Sci. Tech. 1, 1109–1118 (2011).
Basak, A., Barlan, A. U. & Yamamoto, H. Catalytic enantioselective oxidation of sulfides and disulfides by a chiral complex of bis-hydroxamic acid and molybdenum. Tetrahedron 17, 508–511 (2006).
Tytko, K.-H., Fleischmann, W.-D., Gras, D. & Warkentin, E. Mo Molybdenum: Hydrous Molybdates of Groups VA to VIB Metals (System Nos. 18 to 52) Springer (1985).
Tytko, K.-H. & Gras, D. Mo Molybdenum: Oxomolybdenum Species in Aqueous Solutions (Continued) Oxomolybdenum Species in Nonaqueous Solvents Oxomolybdenum Species in Melts Peroxomolybdenum Species Springer (1988).
Dickman, M. H. & Pope, M. T. Peroxo and superoxo complexes of chromium, molybdenum, and tungsten. Chem. Rev. 94, 569–584 (1994).
Zhou, Z.-H., Hou, S.-Y. & Wan, H.-L. Peroxomolybdate(VI)-citrate and -malate complex interconversions by pH-dependence. Synthetic, structural and spectroscopic studies. Dalton Trans. 1393–1399 (2004).
Dengel, A. C., Griffith, W. P., Powell, R. D. & Skapski, A. C. Studies on transition-metal peroxo complexes. Part 7. molybdenum(VI) and tungsten(VI) carboxylato peroxo complexes, and the X-ray crystal structure of K2[MoO(O2)2(glyc)]·2H2O. J. Chem. Soc. Dalton Trans. 991–995 (1987).
Chakravarthy, R. D., Ramkumar, V. & Chand, D. K. A molybdenum based metallomicellar catalyst for controlled and selective sulfoxidation reactions in aqueous medium. Green Chem. 16, 2190–2196 (2014).
Salles, L. et al. Crystal structures of three novel dioxotetraperoxodi(molybdenum) heteropolyanions in [PPh4]2[R2SiO2{Mo2O2(μ-O2)2(O2)2}] (R2=(CH3)2, (i-C4H9)2 or (CH3)(CH2Cl)) and characterization of their tungsten analogues. Polyhedron 26, 4786–4792 (2007).
Gresley, N. M., Griffith, W. P., Laemmel, A. C., Nogueira, H. I. S. & Parkin, B. C. Studies on polyoxo and polyperoxo-metalates part 51: peroxide-catalysed oxidations with heteropolyperoxo-tungstates and -molybdates. J. Mol. Catal. A Chem. 117, 185–198 (1997).
Gresley, N. M., Griffith, W. P., Parkin, B. C., White, A. J. P. & Williams, D. J. The crystal structures of [NMe4][(Me2AsO2){MoO(O2)2}2], [NMe4][(Ph2PO2){MoO(O2)2}2], [NBun4][(Ph2PO2){WO(O2)2}2] and [NH4][(Ph2PO2){MoO(O2)2(H2O)}] and their use as catalytic oxidants. J. Chem. Soc. Dalton Trans. 2039–2045 (1996).
Hashimoto, M., Ichida, H. & Sasaki, Y. Preparation and structure of tetramethylammonium hydrogen triperoxotrimolybdodisulfate trihydrate, [N(CH3)4]3[H(SO4)2(MoO2(O2))3]·3H2O. J. Coord. Chem. 37, 349–359 (1996).
Taube, F., Hashimoto, M., Andersson, I. & Pettersson, L. Characterisation of aqueous peroxomolybdate catalysts applicable to pulp bleaching. J. Chem. Soc. Dalton Trans. 1002–1008 (2002).
Deng, D. L., Helton, M. E., Wang, X. H. & Zhao, W. Bleaching compositions. US Patent 7,645,302 filed 20 Jan. 2006 and issued 12 Jan. 2010.
Salles, L., Robert, F., Semmer, V., Jeannin, Y. & Bregeault, J.-M. Novel di- and trinuclear oxoperoxosulfato species in molybdenum(VI) and tungsten(VI) chemistry: the key role of pairs of bridging peroxo groups. Bull. Soc. Chim. Fr. 133, 319–328 (1996).
Shi, X. & Wei, J. Preparation, characterization and catalytic oxidation properties of bis-quaternary ammonium peroxotungstates and peroxomolybdates complexes. Appl. Organomet. Chem. 21, 172–176 (2007).
Ma, T. et al. Pentanidium-catalyzed enantioselective phase-transfer conjugate addition reactions. J. Am. Chem. Soc. 133, 2828–2831 (2011).
Yang, Y. et al. Pentanidium–Catalyzed Enantioselective α-Hydroxylation of Oxindoles Using Molecular Oxygen. Org. Lett. 14, 4762–4765 (2012).
Zong, L., Ban, X., Kee, C. W. & Tan, C.-H. Catalytic enantioselective alkylation of sulfenate anions to chiral heterocyclic sulfoxides using halogenated pentanidium salts. Angew. Chem. Int. Ed. 53, 11849–11853 (2014).
Zong, L., Du, S., Chin, K. F., Wang, C. & Tan, C.-H. Enantioselective synthesis of quaternary carbon stereocenters: addition of 3-substituted oxindoles to vinyl sulfone catalyzed by pentanidiums. Angew. Chem. Int. Ed. 54, 9390–9393 (2015).
Maruoka, K. Asymmetric Phase Transfer Catalysis Wiley-VCH (2008).
Shirakawa, S. & Maruoka, K. Recent developments in asymmetric phase-transfer reactions. Angew. Chem. Int. Ed. 52, 4312–4348 (2013).
Brak, K. & Jacobsen, E. N. Asymmetric ion-pairing catalysis. Angew. Chem. Int. Ed. 52, 534–561 (2013).
Mahlau, M. & List, B. Asymmetric counteranion-directed catalysis: concept, definition, and applications. Angew. Chem. Int. Ed. 52, 518–533 (2013).
Phipps, R. J., Hamilton, G. L. & Toste, F. D. The progression of chiral anions from concepts to applications in asymmetric catalysis. Nat. Chem. 4, 603–614 (2012).
Uyanik, M., Okamoto, H., Yasui, T. & Ishihara, K. Quaternary ammonium (hypo)iodite catalysis for enantioselective oxidative cycloetherification. Science 328, 1376–1379 (2010).
Waser, M., Novacek, J. & Gratzer, K. in Cooperative Catalysis: Designing Efficient Catalysts for Synthesis ed. Peters R. 197–226Wiley-VCH (2015).
Wang, C., Zong, L. & Tan, C.-H. Enantioselective oxidation of alkenes with potassium permanganate catalyzed by chiral dicationic bisguanidinium. J. Am. Chem. Soc. 137, 10677–10682 (2015).
Dong, S. et al. Organocatalytic oxyamination of azlactones: kinetic resolution of oxaziridines and asymmetric synthesis of oxazolin-4-ones. J. Am. Chem. Soc. 135, 10026–10029 (2013).
Ye, X. et al. Enantioselective sulfoxidation catalyzed by a bisguanidinium diphosphatobisperoxotungstate ion pair. Angew. Chem. Int. Ed. 55, 7101–7105 (2016).
Jahier, C. et al. Optically active tripodal dendritic polyoxometalates: synthesis, characterization and their use in asymmetric sulfide oxidation with hydrogen peroxide. Eur. J. Inorg. Chem. 2011, 727–738 (2011).
Kagan, H. B. in Organosulfur Chemistry in Asymmetric Synthesis eds Takeshi T., Carsten B. 1–29Wiley-VCH (2009).
Bäckvall, J.-E. in Modern Oxidation Methods ed. Jan-Erling B. 277–313Wiley-VCH (2010).
Liao, S., Čorić, I., Wang, Q. & List, B. Activation of H2O2 by chiral confined brønsted acids: a highly enantioselective catalytic sulfoxidation. J. Am. Chem. Soc. 134, 10765–10768 (2012).
Burke, A. J. & Carreiro, E. P. in Comprehensive Inorganic Chemistry II 2nd edn ed. Poeppelmeier K. 309–382Elsevier (2013).
Prisinzano, T., Podobinski, J., Tidgewell, K., Luo, M. & Swenson, D. Synthesis and determination of the absolute configuration of the enantiomers of modafinil. Tetrahedron 15, 1053–1058 (2004).
Rebiere, F., Duret, G., Prat, L. & Piacenza, G. Process for enantioselective synthesis of single enantiomers of modafinil by asymmetric oxidation. US Patent Application 2005/0222257 filed 17 Mar. 2005, and published 6 Oct. 2005.
Ternois, J., Guillen, F., Plaquevent, J.-C. & Coquerel, G. Asymmetric synthesis of modafinil and its derivatives by enantioselective oxidation of thioethers: comparison of various methods including synthesis in ionic liquids. Tetrahedron 18, 2959–2964 (2008).
Hong, L., Sun, W., Yang, D., Li, G. & Wang, R. Additive effects on asymmetric catalysis. Chem. Rev. 116, 4006–4123 (2016).
Firouzabadi, H., Iranpoor, N., Jafari, A. A. & Riazymontazer, E. Metal-free chemoselective oxidation of sulfides to sulfoxides by hydrogen peroxide catalyzed by in situ generated dodecyl hydrogen sulfate in the absence of organic co-solvents. Adv. Synth. Catal. 348, 434–438 (2006).
Miao, C. et al. Proton-promoted and anion-enhanced epoxidation of olefins by hydrogen peroxide in the presence of nonheme manganese catalysts. J. Am. Chem. Soc. 138, 936–943 (2016).
Nardello, V., Marko, J., Vermeersch, G. & Aubry, J. M. 95Mo NMR and kinetic studies of peroxomolybdic intermediates involved in the catalytic disproportionation of hydrogen peroxide by molybdate ions. Inorg. Chem. 34, 4950–4957 (1995).
Thompson, D. J., Cao, Z., Judkins, E. C., Fanwick, P. E. & Ren, T. Peroxo-dimolybdate catalyst for the oxygenation of organic sulfides by hydrogen peroxide. Inorg. Chim. Acta 437, 103–109 (2015).
Zhao, Y., Cotelle, Y., Sakai, N. & Matile, S. Unorthodox Interactions at Work. J. Am. Chem. Soc. 138, 4270–4277 (2016).
Acknowledgements
We acknowledge Nanyang Technological University (M4080946.110, M4011372.110) for financial support. We thank Ms E.L. Goh for her technical support in 95Mo NMR analysis.
Author information
Authors and Affiliations
Contributions
L.Z. and C.-H.T designed and conceived the project. L.Z. performed the experiments and analysed the experimental results. C.-H.T and L.Z wrote the manuscript. C.W. and X.Y. contributed to the initial studies. A.M.P.M. and H.H. performed the computational studies. R.G. and Y.L. contributed to the X-ray data collection.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
Supplementary Figures 1-96, Supplementary Tables 1-5, Supplementary Notes 1-2, Supplementary Methods and Supplementary References. (PDF 15187 kb)
Supplementary Data 1
Cartesian Coordinates for TS Structures. (DOCX 119 kb)
Supplementary Data 2
IUCR's CheckCIF report of compound 1b. (DOC 250 kb)
Supplementary Data 3
Crystallographic information for compound 1b. (CIF 1714 kb)
Supplementary Data 4
IUCR's CheckCIF report of compound 3f. (DOC 159 kb)
Supplementary Data 5
Crystallographic information for compound 3f. (CIF 463 kb)
Supplementary Data 6
IUCR's CheckCIF report of compound 3o. (DOC 153 kb)
Supplementary Data 7
Crystallographic information for compound 3o. (CIF 2398 kb)
Supplementary Data 8
IUCR's CheckCIF report of compound 4. (DOC 160 kb)
Supplementary Data 9
Crystallographic information for compound 4. (CIF 588 kb)
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Zong, L., Wang, C., Moeljadi, A. et al. Bisguanidinium dinuclear oxodiperoxomolybdosulfate ion pair-catalyzed enantioselective sulfoxidation. Nat Commun 7, 13455 (2016). https://doi.org/10.1038/ncomms13455
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/ncomms13455
This article is cited by
-
Thiol-Yne click chemistry of acetylene-enabled macrocyclization
Nature Communications (2022)
-
Cationic Organic Catalysts or Ligands in Concert with Metal Catalysts
Topics in Current Chemistry (2019)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.