Abstract
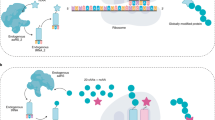
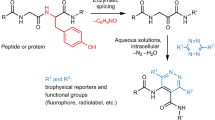
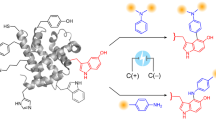
Methods for introducing bioorthogonal functionalities into proteins have become central to protein engineering efforts. Here we describe a method for the site-specific introduction of aldehyde groups into recombinant proteins using the 6-amino-acid consensus sequence recognized by the formylglycine-generating enzyme. This genetically encoded 'aldehyde tag' is no larger than a His6 tag and can be exploited for numerous protein labeling applications.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
Accession codes
References
Schmidt, B., Selmer, T., Ingendoh, A. & von Figura, K. Cell 82, 271–278 (1995).
Cosma, M.P. et al. Cell 113, 445–456 (2003).
Dierks, T. et al. Cell 113, 435–444 (2003).
Dierks, T., Lecca, M.R., Schlotterhose, P., Schmidt, B. & von Figura, K. EMBO J. 18, 2084–2091 (1999).
Landgrebe, J., Dierks, T., Schmidt, B. & von Figura, K. Gene 316, 47–56 (2003).
Gilmore, J.M., Scheck, R.A., Esser-Kahn, A.P., Joshi, N.S. & Francis, M.B. Angew. Chem. Int. Edn Engl. 45, 5307–5311 (2006).
Link, A.J., Mock, M.L. & Tirrell, D.A. Curr. Opin. Biotechnol. 14, 603–609 (2003).
Chen, I., Howarth, M., Lin, W.Y. & Ting, A.Y. Nat. Methods 2, 99–104 (2005).
Griffin, B.A., Adams, S.R. & Tsien, R.Y. Science 281, 269–272 (1998).
Wang, L., Xie, J. & Schultz, P.G. Annu. Rev. Biophys. Biomol. Struct. 35, 225–249 (2006).
Clarke, K.M., Mercer, A.C., La Clair, J.J. & Burkart, M.D. J. Am. Chem. Soc. 127, 11234–11235 (2005).
George, N., Pick, H., Vogel, H., Johnsson, N. & Johnsson, K. J. Am. Chem. Soc. 126, 8896–8897 (2004).
Yin, J., Liu, F., Li, X. & Walsh, C.T. J. Am. Chem. Soc. 126, 7754–7755 (2004).
Dierks, T. et al. J. Biol. Chem. 273, 25560–25564 (1998).
Harris, J.M. & Chess, R.B. Nat. Rev. Drug Discov. 2, 214–221 (2003).
Acknowledgements
We thank M. Francis and J. Rush for helpful discussions and D. King and A. Falick for MS expertise. I.S.C. was supported by a postdoctoral fellowship from the US National Institutes of Health. B.L.C. was supported by a predoctoral fellowship from the US National Science Foundation. This work was supported by a grant from the US National Institutes of Health to C.R.B. (GM59907).
Author information
Authors and Affiliations
Contributions
I.S.C. and B.L.C. carried out cloning, expression, purification and fluorescent tagging of the constructs. I.S.C. quantified conversion to fGly and performed multiple epitope assays. B.L.C. performed PEGylation assays. C.R.B. directed the project. All authors worked together to compose the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Fig. 1
Mass spectra confirming the presence of fGly in a tryptic peptide from ald13-Stf0. (PDF 222 kb)
Supplementary Fig. 2
Quantitation of the conversion of cysteine to formylglycine using mass spectrometry. (PDF 156 kb)
Supplementary Fig. 3
Quantitation of the conversion of cysteine to formylglycine using Alexa Fluor 647 C5-aminooxyacetamide labeling. (PDF 251 kb)
Supplementary Fig. 4
PEGylation of ald6-MBP with 2 kDa and 5 kDa aminooxy-PEG. (PDF 184 kb)
Supplementary Table 1
Oligonucleotides used in this study. (PDF 274 kb)
Rights and permissions
About this article
Cite this article
Carrico, I., Carlson, B. & Bertozzi, C. Introducing genetically encoded aldehydes into proteins. Nat Chem Biol 3, 321–322 (2007). https://doi.org/10.1038/nchembio878
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nchembio878
This article is cited by
-
Traceless cysteine-linchpin enables precision engineering of lysine in native proteins
Nature Communications (2022)
-
Selective and predicable amine conjugation sites by kinetic characterization under excess reagents
Scientific Reports (2021)
-
Assembling custom side chains on proteoglycans to interrogate their function in living cells
Nature Communications (2020)
-
Selective loading and processing of prespacers for precise CRISPR adaptation
Nature (2020)
-
Expanding the limits of the second genetic code with ribozymes
Nature Communications (2019)