Abstract
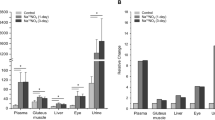
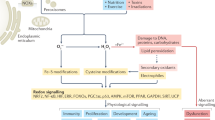
Mammalian tissues produce nitric oxide (NO) to modify proteins at heme and sulfhydryl sites, thereby regulating vital cell functions. The majority of NO produced is widely assumed to be neutralized into supposedly inert oxidation products including nitrite (NO2−). Here we show that nitrite, also ubiquitous in dietary sources, is remarkably efficient at modifying the same protein sites, and that physiological nitrite concentrations account for the basal levels of these modifications in vivo. We further find that nitrite readily affects cyclic GMP production, cytochrome P450 activities, and heat shock protein 70 and heme oxygenase-1 expression in a variety of tissues. These cellular activities of nitrite, combined with its stability and abundance in vivo, suggest that this anion has a distinct and important signaling role in mammalian biology, perhaps by serving as an endocrine messenger and synchronizing agent. Thus, nitrite homeostasis may be of great importance to NO biology.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Thomas, D.D. et al. Heme proteins and nitric oxide (NO): the neglected, eloquent chemistry in NO redox signaling and regulation. Antioxid. Redox Signal. 5, 307–317 (2003).
Schulz, R., Kelm, M. & Heusch, G. Nitric oxide in myocardial ischemia/reperfusion injury. Cardiovasc. Res. 61, 402–413 (2004).
Ignarro, L.J., Fukuto, J.M., Griscavage, J.M., Rogers, N.E. & Byrns, R.E. Oxidation of nitric oxide in aqueous solution to nitrite but not nitrate: comparison with enzymatically formed nitric oxide from L-arginine. Proc. Natl. Acad. Sci. USA 90, 8103–8107 (1993).
Meah, M.N., Harrison, N. & Davies, A. Nitrate and nitrite in foods and the diet. Food Addit. Contam. 11, 519–532 (1994).
Spiegelhalder, B., Eisenbrand, G. & Preussmann, R. Influence of dietary nitrate on nitrite content of human saliva: possible relevance to in vivo formation of N-nitroso compounds. Food Cosmet. Toxicol. 14, 545–548 (1976).
Gangolli, S.D. et al. Nitrate, nitrite, and N-nitroso compounds. Eur. J. Pharmacol. 292, 1–38 (1994).
Zweier, J.L., Wang, P., Samouilov, A. & Kuppusamy, P. Enzyme-independent formation of nitric oxide in biological tissues. Nat. Med. 1, 804–809 (1995).
Gladwin, M.T. et al. Role of circulating nitrite and S-nitrosohemoglobin in the regulation of regional blood flow in human. Proc. Natl. Acad. Sci. USA 97, 11482–11487 (2000).
Cosby, K. et al. Nitrite reduction to nitric oxide by deoxyhemoglobin vasodilates the human circulation. Nat. Med. 9, 1498–1505 (2003).
Duranski, M.R. et al. Cytoprotective effects of nitrite during in vivo ischemia-reperfusion of the heart and liver. J. Clin. Invest. 115, 1232–1240 (2005).
Lundberg, J.O. & Weitzberg, E. NO generation from nitrite and its role in vascular control. Arterioscler. Thromb. Vasc. Biol. 25, 915–922 (2005).
Gladwin, M.T. Haldane, hot dogs, halitosis, and hypoxic vasodilation: the emerging biology of the nitrite anion. J. Clin. Invest. 113, 19–21 (2004).
Parks, N.J. et al. Nitrogen-13-Labeled nitrite and nitrate: distribution and metabolism after intratracheal administration. Science 212, 58–61 (1981).
Thayer, J.R., Chasko, J.H., Swartz, L.A. & Parks, N.J. Gut reactions of radioactive nitrite after intratracheal administration in mice. Science 217, 151–153 (1982).
Kelm, M. Nitric oxide metabolism and breakdown. Biochim. Biophys. Acta 1411, 273–289 (1999).
Rath, M.M. & Krantz, J.C.J. Nitrites: a further study of the mechanism of the action of organic nitrates. J. Pharmacol. Exp. Ther. 76, 33–38 (1942).
Vleeming, W., van de Kuil, A., te Biesebeek, J.D., Meulenbelt, J. & Boink, A.B. Effect of nitrite on blood pressure in anaesthetized and free-moving rats. Food Chem. Toxicol. 35, 615–619 (1997).
Reichert, E.T. & Mitchell, S.W. On the physiological action of potassium nitrite, with a note on the physiological action on man. Am. J. Med. Sci. 159, 158–180 (1880).
Crawford, J.H. et al. Transduction of NO-bioactivity by the red blood cell in sepsis: novel mechanisms of vasodilation during acute inflammatory disease. Blood 104, 1375–1382 (2004).
Malyshev, I.Y., Manukhina, E.B., Mikoyan, V.D., Kubrina, L.N. & Vanin, A.F. Nitric oxide is involved in heat-induced Hsp70 accumulation. FEBS Lett. 370, 159–162 (1995).
Motterlini, R., Green, C.J. & Foresti, R. Regulation of heme oxygenase-1 by redox signals involving nitric oxide. Antioxid. Redox Signal. 4, 615–624 (2002).
Wink, D.A., Darbyshire, J.F., Nims, R.W., Saavedra, J.E. & Ford, P.C. Reactions of the bioregulatory agent nitric oxide in oxygenated aqueous media: determination of the kinetics for oxidation and nitrosation by intermediates generated in the NO/O2 reaction. Chem. Res. Toxicol. 6, 23–27 (1993).
Kim-Shapiro, D.B., Gladwin, M.T., Patel, R.P. & Hogg, N. The reaction between nitrite and hemoglobin: the role of nitrite in hemoglobin-mediated hypoxic vasodilation. J. Inorg. Biochem. 99, 237–246 (2005).
Bryan, N.S. et al. Cellular targets and mechanisms of nitros(yl)ation: an insight into their nature and kinetics in vivo. Proc. Natl. Acad. Sci. USA 101, 4308–4313 (2004).
Pilz, R.B. & Casteel, D.E. Regulation of gene expression by cyclic GMP. Circ. Res. 93, 1034–1046 (2003).
Panesar, N.S. & Chan, K.W. Decreased steroid hormone synthesis from inorganic nitrite and nitrate: studies in vitro and in vivo. Toxicol. Appl. Pharmacol. 169, 222–230 (2000).
Marshall, H.E., Merchant, K. & Stamler, J.S. Nitrosation and oxidation in the regulation of gene expression. FASEB J. 14, 1889–1900 (2000).
Papp, E., Nardai, G., Soti, C. & Csermely, P. Molecular chaperones, stress proteins and redox homeostasis. Biofactors 17, 249–257 (2003).
Balashova, N., Chang, F.J., Lamothe, M., Sun, Q. & Beuve, A. Characterization of a novel type of endogenous activator of soluble guanylyl cyclase. J. Biol. Chem. 280, 2186–2196 (2005).
Webb, A. et al. Reduction of nitrite to nitric oxide during ischemia protects against myocardial ischemia-reperfusion damage. Proc. Natl. Acad. Sci. USA 101, 13683–13688 (2004).
Otterbein, L.E., Soares, M.P., Yamashita, K. & Bach, F.H. Heme-oxygenase-1: unleashing the protective properties of heme. Trends Immunol. 24, 449–455 (2003).
Sunamura, M. et al. Heme oxygenase-1 accelerates tumor angiogenesis of human pancreatic cancer. Angiogenesis 6, 15–24 (2003).
Fang, J. et al. In vivo antitumor activity of pegylated zinc protoporphyrin: targeted inhibition of heme oxygenase in solid tumor. Cancer Res. 63, 3567–3574 (2003).
Feelisch, M. & Martin, J. The early role of nitric oxide in evolution. Trends Ecol. Evol. 10, 496–499 (1995).
Lundberg, J.O. & Govoni, M. Inorganic nitrate is a possible source for systemic generation of nitric oxide. Free Radic. Biol. Med. 37, 395–400 (2004).
Lundberg, J.O., Weitzberg, E., Cole, J.A. & Benjamin, N. Nitrate, bacteria and human health. Nat. Rev. Microbiol. 2, 593–602 (2004).
Marley, R., Feelisch, M., Holt, S. & Moore, K. A chemiluminscent-based assay for S-nitrosoalbumin and other plasma S-nitrosothiols. Free Radic. Res. 32, 1–9 (2000).
Feelisch, M. et al. Concomitant S-, N-, and heme-nitros(yl)ation in biological tissues and fluids: implications for the fate of NO in vivo. FASEB J. 16, 1775–1785 (2002).
Rassaf, T., Bryan, N.S., Kelm, M. & Feelisch, M. Concomitant presence of N-nitroso and S-nitroso proteins in human plasma. Free Radic. Biol. Med. 33, 1590–1596 (2002).
Burke, M.D. et al. Ethoxy-, pentoxy- and benzyloxyphenoxazones and homologues: a series of substrates to distinguish between different induced cytochrome P-450. Biochem. Pharmacol. 34, 3337–3345 (1985).
Ghosal, A. et al. Rapid determination of enzyme activities of recombinant human cytochromes P450, human liver microsomes and hepatocytes. Biopharm. Drug Dispos. 24, 375–384 (2003).
Acknowledgements
We wish to thank F. Saijo and D. Perlman for skillful technical assistance and M. Kelm, M. Bausero and C. Brame for helpful comments. N.S.B. and B.O.F. are recipients of the Kirschstein–National Research Service Award Cardiovascular Training Grant from the US National Institutes of Health (NIH). This work was supported in part by a grant from the NIH (HL69029 to M.F.). T.R. is a research fellow sponsored by the Deutshe Forschungsgesellschaft.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
M.F. is a paid consultant and member of the Scientific Advisory Board of Nitromed, Inc.
Supplementary information
Rights and permissions
About this article
Cite this article
Bryan, N., Fernandez, B., Bauer, S. et al. Nitrite is a signaling molecule and regulator of gene expression in mammalian tissues. Nat Chem Biol 1, 290–297 (2005). https://doi.org/10.1038/nchembio734
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nchembio734
This article is cited by
-
Influence of acute dietary nitrate supplementation timing on nitrate metabolism, central and peripheral blood pressure and exercise tolerance in young men
European Journal of Applied Physiology (2024)
-
Quantitative aspects of nitric oxide production in the heart
Molecular Biology Reports (2022)
-
S-nitrosylation-mediated activation of a histidine kinase represses the type 3 secretion system and promotes virulence of an enteric pathogen
Nature Communications (2020)
-
Sodium nitrate preconditioning prevents progression of the neuropathic pain in streptozotocin-induced diabetes Wistar rats
Journal of Diabetes & Metabolic Disorders (2020)
-
Functional Nitric Oxide Nutrition to Combat Cardiovascular Disease
Current Atherosclerosis Reports (2018)