Abstract
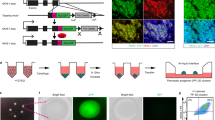
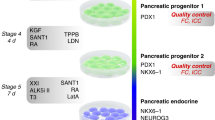
Stepwise differentiation from embryonic stem cells (ESCs) to functional insulin-secreting beta cells will identify key steps in beta-cell development and may yet prove useful for transplantation therapy for diabetics. An essential step in this schema is the generation of pancreatic progenitors—cells that express Pdx1 and produce all the cell types of the pancreas. High-content chemical screening identified a small molecule, (−)-indolactam V, that induces differentiation of a substantial number of Pdx1-expressing cells from human ESCs. The Pdx1-expressing cells express other pancreatic markers and contribute to endocrine, exocrine and duct cells, in vitro and in vivo. Further analyses showed that (−)-indolactam V works specifically at one stage of pancreatic development, inducing pancreatic progenitors from definitive endoderm. This study describes a chemical screening platform to investigate human ESC differentiation and demonstrates the generation of a cell population that is a key milepost on the path to making beta cells.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Anonymous. Regenerative Medicine. National Institutes of Health http://stemcells.nih.gov/info/scireport/2006report.htm (2006).
Lumelsky, N. et al. Differentiation of embryonic stem cells to insulin-secreting structures similar to pancreatic islets. Science 292, 1389–1394 (2001).
Soria, B. In-vitro differentiation of pancreatic beta-cells. Differentiation 68, 205–219 (2001).
Rajagopal, J., Anderson, W.J., Kume, S., Martinez, O.I. & Melton, D.A. Insulin staining of ES cell progeny from insulin uptake. Science 299, 363 (2003).
Stainier, D.Y. A glimpse into the molecular entrails of endoderm formation. Genes Dev. 16, 893–907 (2002).
D'Amour, K.A. et al. Efficient differentiation of human embryonic stem cells to definitive endoderm. Nat. Biotechnol. 23, 1534–1541 (2005).
Kubo, A. et al. Development of definitive endoderm from embryonic stem cells in culture. Development 131, 1651–1662 (2004).
D'Amour, K.A. et al. Production of pancreatic hormone-expressing endocrine cells from human embryonic stem cells. Nat. Biotechnol. 24, 1392–1401 (2006).
McLean, A.B. et al. Activin a efficiently specifies definitive endoderm from human embryonic stem cells only when phosphatidylinositol 3-kinase signaling is suppressed. Stem Cells 25, 29–38 (2007).
Jiang, W. et al. In vitro derivation of functional insulin-producing cells from human embryonic stem cells. Cell Res. 17, 333–344 (2007).
Ku, H.T. et al. Committing embryonic stem cells to early endocrine pancreas in vitro. Stem Cells 22, 1205–1217 (2004).
Gu, G., Brown, J.R. & Melton, D.A. Direct lineage tracing reveals the ontogeny of pancreatic cell fates during mouse embryogenesis. Mech. Dev. 120, 35–43 (2003).
Jonsson, J., Carlsson, L., Edlund, T. & Edlund, H. Insulin-promoter-factor 1 is required for pancreas development in mice. Nature 371, 606–609 (1994).
Offield, M.F. et al. PDX-1 is required for pancreatic outgrowth and differentiation of the rostral duodenum. Development 122, 983–995 (1996).
Stafford, D. & Prince, V.E. Retinoic acid signaling is required for a critical early step in zebrafish pancreatic development. Curr. Biol. 12, 1215–1220 (2002).
Tiso, N., Filippi, A., Pauls, S., Bortolussi, M. & Argenton, F. BMP signalling regulates anteroposterior endoderm patterning in zebrafish. Mech. Dev. 118, 29–37 (2002).
Roy, S., Qiao, T., Wolff, C. & Ingham, P.W. Hedgehog signaling pathway is essential for pancreas specification in the zebrafish embryo. Curr. Biol. 11, 1358–1363 (2001).
diIorio, P.J., Moss, J.B., Sbrogna, J.L., Karlstrom, R.O. & Moss, L.G. Sonic hedgehog is required early in pancreatic islet development. Dev. Biol. 244, 75–84 (2002).
Bhushan, A. et al. Fgf10 is essential for maintaining the proliferative capacity of epithelial progenitor cells during early pancreatic organogenesis. Development 128, 5109–5117 (2001).
Hart, A., Papadopoulou, S. & Edlund, H. Fgf10 maintains notch activation, stimulates proliferation, and blocks differentiation of pancreatic epithelial cells. Dev. Dyn. 228, 185–193 (2003).
Kroon, E. et al. Pancreatic endoderm derived from human embryonic stem cells generates glucose-responsive insulin-secreting cells in vivo. Nat. Biotechnol. 26, 443–452 (2008).
Ding, S. & Schultz, P.G. A role for chemistry in stem cell biology. Nat. Biotechnol. 22, 833–840 (2004).
Chen, S. et al. Self-renewal of embryonic stem cells by a small molecule. Proc. Natl. Acad. Sci. USA 103, 17266–17271 (2006).
Osafune, K. et al. Marked differences in differentiation propensity among human embryonic stem cell lines. Nat. Biotechnol. 26, 313–315 (2008).
Frandsen, U., Porneki, A.D., Floridon, C., Abdallah, B.M. & Kassem, M. Activin B mediated induction of Pdx1 in human embryonic stem cell derived embryoid bodies. Biochem. Biophys. Res. Commun. 362, 568–574 (2007).
Kim, S.K. & Melton, D.A. Pancreas development is promoted by cyclopamine, a hedgehog signaling inhibitor. Proc. Natl. Acad. Sci. USA 95, 13036–13041 (1998).
Jacquemin, P., Lemaigre, F.P. & Rousseau, G.G. The Onecut transcription factor HNF-6 (OC-1) is required for timely specification of the pancreas and acts upstream of Pdx-1 in the specification cascade. Dev. Biol. 258, 105–116 (2003).
Kawaguchi, Y. et al. The role of the transcriptional regulator Ptf1a in converting intestinal to pancreatic progenitors. Nat. Genet. 32, 128–134 (2002).
Beck, F., Erler, T., Russell, A. & James, R. Expression of Cdx-2 in the mouse embryo and placenta: possible role in patterning of the extra-embryonic membranes. Dev. Dyn. 204, 219–227 (1995).
Dziadek, M.A. & Andrews, G.K. Tissue specificity of alpha-fetoprotein messenger RNA expression during mouse embryogenesis. EMBO J. 2, 549–554 (1983).
Bendayan, M. & Ito, S. Immunohistochemical localization of exocrine enzymes in normal rat pancreas. J. Histochem. Cytochem. 27, 1029–1034 (1979).
Osafune, K. et al. Marked differences in differentiation propensity among human embryonic stem cell lines. Nat. Biotechnol. 26, 313–315 (2008).
Korsgren, O., Jansson, L., Eizirik, D. & Andersson, A. Functional and morphological differentiation of fetal porcine islet-like cell clusters after transplantation into nude mice. Diabetologia 34, 379–386 (1991).
Movassat, J., Beattie, G.M., Lopez, A.D. & Hayek, A. Exendin 4 up-regulates expression of PDX 1 and hastens differentiation and maturation of human fetal pancreatic cells. J. Clin. Endocrinol. Metab. 87, 4775–4781 (2002).
Seymour, P.A. et al. SOX9 is required for maintenance of the pancreatic progenitor cell pool. Proc. Natl. Acad. Sci. USA 104, 1865–1870 (2007).
Sherwood, R.I. et al. Prospective isolation and global gene expression analysis of definitive and visceral endoderm. Dev. Biol. 304, 541–555 (2007).
Hirota, M. et al. Synthetic analogues (indolactams) of (−)-indolactam-V are new congeners of the teleocidin class of tumor promoters. Jpn. J. Cancer Res. 78, 577–582 (1987).
Heikkila, J. & Akerman, K.E. (−)-Indolactam V activates protein kinase C and induces changes in muscarinic receptor functions in SH-SY5Y human neuroblastoma cells. Biochem. Biophys. Res. Commun. 162, 1207–1213 (1989).
Griner, E.M. & Kazanietz, M.G. Protein kinase C and other diacylglycerol effectors in cancer. Nat. Rev. Cancer 7, 281–294 (2007).
Gschwendt, M. et al. Inhibition of protein kinase C mu by various inhibitors. Differentiation from protein kinase c isoenzymes. FEBS Lett. 392, 77–80 (1996).
Martiny-Baron, G. et al. Selective inhibition of protein kinase C isozymes by the indolocarbazole Go 6976. J. Biol. Chem. 268, 9194–9197 (1993).
Takahashi, K. et al. Induction of pluripotent stem cells from adult human fibroblasts by defined factors. Cell 131, 861–872 (2007).
Nakagawa, M. et al. Generation of induced pluripotent stem cells without Myc from mouse and human fibroblasts. Nat. Biotechnol. 26, 101–106 (2008).
Yu, J. et al. Induced pluripotent stem cell lines derived from human somatic cells. Science 318, 1917–1920 (2007).
Park, I.H. et al. Reprogramming of human somatic cells to pluripotency with defined factors. Nature 451, 141–146 (2008).
Ochoa, W.F. et al. Retinoic acid binds to the C2-domain of protein kinase C(alpha). Biochemistry 42, 8774–8779 (2003).
Radominska-Pandya, A. et al. Direct interaction of all-trans-retinoic acid with protein kinase C (PKC). Implications for PKC signaling and cancer therapy. J. Biol. Chem. 275, 22324–22330 (2000).
Rosewicz, S., Brembeck, F., Kaiser, A., Marschall, Z.V. & Riecken, E.O. Differential growth regulation by all-trans retinoic acid is determined by protein kinase C alpha in human pancreatic carcinoma cells. Endocrinology 137, 3340–3347 (1996).
Bouzinba-Segard, H., Fan, X.T., Perderiset, M. & Castagna, M. Synergy between phorbol esters and retinoic acid in inducing protein kinase C activation. Biochem. Biophys. Res. Commun. 204, 112–119 (1994).
Mailhos, C., Howard, M.K. & Latchman, D.S. A common pathway mediates retinoic acid and PMA-dependent programmed cell death (apoptosis) of neuronal cells. Brain Res. 644, 7–12 (1994).
Acknowledgements
D.M. and S.L.S. are investigators of the Howard Hughes Medical Institute. S.C. is supported by the Howard Hughes Medical Institute and Juvenile Diabetes Research Foundation. R.M. is supported by the Howard Hughes Medical Institute. L.F.P. is supported by the American Gastroenterological Association and the National Pancreas Foundation. The authors thank D. Huangfu for thoughtful discussion, H. Ngo for assistance with the high-content screen, S. Huang for help with NMR analysis and the Beta Cell Biology Consortium for providing antibodies.
Author information
Authors and Affiliations
Contributions
S.C., D.M. and S.L.S. designed the experiments, analyzed data and wrote the manuscript; S.C., M.B., R.M. and K.O. performed the experiments; J.L.F., L.F.P., S.L.S. and L.L.R. provided the chemical library; and S.C., L.D., K.L. and L.L.R. performed the high-content screen.
Corresponding author
Ethics declarations
Competing interests
The authors have filed a patent on the ILV molecule described in the paper.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–13, Supplementary Table 1 and Supplementary Methods (PDF 2306 kb)
Rights and permissions
About this article
Cite this article
Chen, S., Borowiak, M., Fox, J. et al. A small molecule that directs differentiation of human ESCs into the pancreatic lineage. Nat Chem Biol 5, 258–265 (2009). https://doi.org/10.1038/nchembio.154
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nchembio.154
This article is cited by
-
Pancreatic β-cell heterogeneity in adult human islets and stem cell-derived islets
Cellular and Molecular Life Sciences (2023)
-
Stepwise differentiation of functional pancreatic β cells from human pluripotent stem cells
Cell Regeneration (2022)
-
Optimizing Generation of Stem Cell-Derived Islet Cells
Stem Cell Reviews and Reports (2022)
-
Recent progress in pancreatic islet cell therapy
Inflammation and Regeneration (2021)
-
Stabilization of heterochromatin by CLOCK promotes stem cell rejuvenation and cartilage regeneration
Cell Research (2021)