Abstract
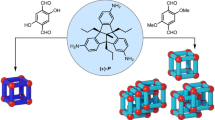
Interlocked molecules possess properties and functions that depend upon their intricate connectivity. In addition to the topologically trivial rotaxanes, whose structures may be captured by a planar graph, the topologically non-trivial knots and catenanes represent some of chemistry's most challenging synthetic targets because of the three-dimensional assembly necessary for their construction. Here we report the synthesis of a cyclic [3]catenane, which consists of three mutually interpenetrating rings, via an unusual synthetic route. Five distinct building blocks self-assemble into a heteroleptic triangular framework composed of two joined FeII3L3 circular helicates. Subcomponent exchange then enables specific points in the framework to be linked together to generate the cyclic [3]catenane product. Our method represents an advance both in the intricacy of the metal-templated self-assembly procedure and in the use of selective imine exchange to generate a topologically complex product.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Evans, N. H. & Beer, P. D. Progress in the synthesis and exploitation of catenanes since the millennium. Chem. Soc. Rev. 43, 4658–4683 (2014).
Forgan, R. S., Sauvage, J. P. & Stoddart, J. F. Chemical topology: complex molecular knots, links, and entanglements. Chem. Rev. 111, 5434–5464 (2011).
Beves, J. E., Blight, B. A., Campbell, C. J., Leigh, D. A. & McBurney, R. T. Strategies and tactics for the metal-directed synthesis of rotaxanes, knots, catenanes, and higher order links. Angew. Chem. Int. Ed. 50, 9260–9327 (2011).
Ponnuswamy, N., Cougnon, F. B., Clough, J. M., Pantos, G. D. & Sanders, J. K. Discovery of an organic trefoil knot. Science 338, 783–785 (2012).
van Dongen, S. F., Cantekin, S., Elemans, J. A., Rowan, A. E. & Nolte, R. J. Functional interlocked systems. Chem. Soc. Rev. 43, 99–122 (2014).
Liang, C. Z. & Mislow, K. Topological chirality and achirality of links. J. Math. Chem. 18, 1–24 (1995).
Dean, F. B. & Cozzarelli, N. R. Mechanism of strand passage by Escherichia coli topoisomerase I. The role of the required nick in catenation and knotting of duplex DNA. J. Biol. Chem. 260, 4984–4994 (1985).
Takusagawa, F. & Kamitori, S. A real knot in protein. J. Am. Chem. Soc. 118, 8945–8946 (1996).
Rosengren, K. J. et al. Microcin J25 has a threaded sidechain-to-backbone ring structure and not a head-to-tail cyclized backbone. J. Am. Chem. Soc. 125, 12464–12474 (2003).
Duda, R. L. Protein chainmail: catenated protein in viral capsids. Cell 94, 55–60 (1998).
Quake, S. R. Topological effects of knots in polymers. Phys. Rev. Lett. 73, 3317–3320 (1994).
Saitta, A. M., Soper, P. D., Wasserman, E. & Klein, M. L. Influence of a knot on the strength of a polymer strand. Nature 399, 46–48 (1999).
Sauvage, J. P. Interlacing molecular threads on transition-metals—catenands, catenates, and knots. Acc. Chem. Res. 23, 319–327 (1990).
Ashton, P. R. et al. A [2]catenane made to order. Angew. Chem. Int. Ed. Engl. 28, 1396–1399 (1989).
Hunter, C. A. Synthesis and structure elucidation of a new [2]-catenane. J. Am. Chem. Soc. 114, 5303–5311 (1992).
Nierengarten, J. F., Dietrichbuchecker, C. O. & Sauvage, J. P. Synthesis of a doubly interlocked [2]-catenane. J. Am. Chem. Soc. 116, 375–376 (1994).
Mohr, B., Weck, M., Sauvage, J. P. & Grubbs, R. H. High-yield synthesis of [2]catenanes by intramolecular ring-closing metathesis. Angew. Chem. Int. Ed. Engl. 36, 1308–1310 (1997).
Lincheneau, C., Jean-Denis, B. & Gunnlaugsson, T. Self-assembly formation of mechanically interlocked [2]- and [3]catenanes using lanthanide ion [Eu(III)] templation and ring closing metathesis reactions. Chem. Commun. 50, 2857–2860 (2014).
Leigh, D. A., Pritchard, R. G. & Stephens, A. J. A star of David catenane. Nature Chem. 6, 978–982 (2014).
Chichak, K. S. et al. Molecular Borromean rings. Science 304, 1308–1312 (2004).
Ayme, J. F. et al. A synthetic molecular pentafoil knot. Nature Chem. 4, 15–20 (2012).
Hasenknopf, B., Lehn, J. M., Kneisel, B. O., Baum, G. & Fenske, D. Self-assembly of a circular double helicate. Angew. Chem. Int. Ed. Engl. 35, 1838–1840 (1996).
Allen, K. E. et al. Head-to-tail and heteroleptic pentanuclear circular helicates. Angew. Chem. Int. Ed. 49, 6655–6658 (2010).
Safont-Sempere, M. M., Fernandez, G. & Wurthner, F. Self-sorting phenomena in complex supramolecular systems. Chem. Rev. 111, 5784–5814 (2011).
Ayme, J. F., Beves, J. E., Campbell, C. J. & Leigh, D. A. The self-sorting behavior of circular helicates and molecular knots and links. Angew. Chem. Int. Ed. 53, 7823–7827 (2014).
Kramer, R., Lehn, J. M. & Marquis-Rigault, A. Self-recognition in helicate self-assembly: spontaneous formation of helical metal complexes from mixtures of ligands and metal ions. Proc. Natl Acad. Sci. USA 90, 5394–5398 (1993).
Riddell, I. A. et al. Five discrete multinuclear metal–organic assemblies from one ligand: deciphering the effects of different templates. J. Am. Chem. Soc. 135, 2723–2733 (2013).
Sarma, R. J. & Nitschke, J. R. Self-assembly in systems of subcomponents: simple rules, subtle consequences. Angew. Chem. Int. Ed. 47, 377–380 (2008).
De, S., Mahata, K. & Schmittel, M. Metal-coordination-driven dynamic heteroleptic architectures. Chem. Soc. Rev. 39, 1555–1575 (2010).
Schultz, D. & Nitschke, J. R. Designing multistep transformations using the Hammett equation: imine exchange on a copper(I) template. J. Am. Chem. Soc. 128, 9887–9892 (2006).
Tung, S. T., Lai, C. C., Liu, Y. H., Peng, S. M. & Chiu, S. H. Synthesis of a [2]catenane from the sodium ion templated orthogonal arrangement of two diethylene glycol chains. Angew. Chem. Int. Ed. 52, 13269–13272 (2013).
Peters, A. J., Chichak, K. S., Cantrill, S. J. & Stoddart, J. F. Nanoscale Borromean links for real. Chem. Commun. 3394–3396 (2005).
Chung, M.-K., Severin, K., Lee, S. J., Waters, M. L. & Gagné, M. R. Constitutionally selective amplification of multicomponent 84-membered macrocyclic hosts for (−)-cytidine·H+. Chem. Sci. 2, 744–747 (2011).
Dietrich-Buchecker, C., Leize, E., Nierengarten, J.-F., Sauvage, J.-P. & Van Dorsselaer, A. Singly and doubly interlocked [2]-catenanes: influence of the degree of entanglement on chemical stability as estimated by fast atom bombardment (FAB) and electrospray ionization (ESI) mass spectrometries (MS). J. Chem. Soc. Chem. Commun. 2257–2258 (1994).
Zhu, R., Lübben, J., Dittrich, B. & Clever, G. H. Stepwise halide-triggered double and triple catenation of self-assembled coordination cages. Angew. Chem. Int. Ed. 54, 2796–2800 (2015).
Acknowledgements
This work was supported by the UK Engineering and Physical Sciences Research Council (EPSRC) and a Marie Curie fellowship for J.J.H. (ITN-2010–264645). The authors thank the Diamond Light Source (UK) for synchrotron beamtime on I19 (MT7984 and MT8464).
Author information
Authors and Affiliations
Contributions
J.R.N. and C.S.W. conceived and designed the experiments. C.S.W. performed the experiments and analysed the data. T.K.R. and J.J.H. collected the X-ray data and refined the structures. A.M.B. and C.S.W. carried out the HPLC analysis. All authors discussed the results and edited the manuscript
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary information
Supplementary information (PDF 2284 kb)
Supplementary information
Crystallographic data for compound [1Cl•;Br6]•5ClO4•0.5FeBr4•10CH3CN•H2O. (CIF 14026 kb)
Supplementary information
Crystallographic data for compound [1Me•Cl6]•6ClO4•5CH3CN. (CIF 118 kb)
Supplementary information
Crystallographic data for compound [1OMe•(SCN)6]•4PF6. (CIF 5600 kb)
Supplementary information
Crystallographic data for compound [2]•6Br•0.75KBr•6.25H2O. (CIF 17958 kb)
Rights and permissions
About this article
Cite this article
Wood, C., Ronson, T., Belenguer, A. et al. Two-stage directed self-assembly of a cyclic [3]catenane. Nature Chem 7, 354–358 (2015). https://doi.org/10.1038/nchem.2205
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nchem.2205
This article is cited by
-
Cross-catenation between position-isomeric metallacages
Nature Communications (2024)
-
Mechanically axially chiral catenanes and noncanonical mechanically axially chiral rotaxanes
Nature Chemistry (2022)
-
Self-assembly of a layered two-dimensional molecularly woven fabric
Nature (2020)
-
A cyclic bis[2]catenane metallacage
Nature Communications (2020)
-
Metal–peptide rings form highly entangled topologically inequivalent frameworks with the same ring- and crossing-numbers
Nature Communications (2019)