Abstract
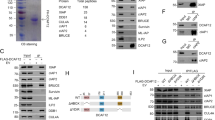
Members of the Inhibitor of Apoptosis Protein (IAP) family block activation of the intrinsic cell death machinery by binding to and neutralizing the activity of pro-apoptotic caspases. In Drosophila melanogaster, the pro-apoptotic proteins Reaper (Rpr), Grim and Hid (head involution defective) all induce cell death by antagonizing the anti-apoptotic activity of Drosophila IAP1 (DIAP1), thereby liberating caspases. Here, we show that in vivo, the RING finger of DIAP1 is essential for the regulation of apoptosis induced by Rpr, Hid and Dronc. Furthermore, we show that the RING finger of DIAP1 promotes the ubiquitination of both itself and of Dronc. Disruption of the DIAP1 RING finger does not inhibit its binding to Rpr, Hid or Dronc, but completely abrogates ubiquitination of Dronc. Our data suggest that IAPs suppress apoptosis by binding to and targeting caspases for ubiquitination.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Meier, P., Finch, A. & Evan, G. Nature 407, 796–801 (2000).
Thornberry, N. A. & Lazebnik, Y. Science 281, 1312–1316 (1998).
Alnemri, E. S. J. Cell. Biochem. 64, 33–42 (1997).
Hengartner, M. O. Nature 407, 770–776 (2000).
Uren, A. G., Coulson, E. J. & Vaux, D. L. Trends Biochem. Sci. 23, 159–162 (1998).
Deveraux, Q. L. & Reed, J. C. Genes Dev. 13, 239–252 (1999).
Wang, S. L., Hawkins, C. J., Yoo, S. J., Muller, H. A. & Hay, B. A. Cell 98, 453–463 (1999).
Lisi, S., Mazzon, I. & White, K. Genetics 154, 669–678 (2000).
Goyal, L., McCall, K., Agapite, J., Hartwieg, E. & Steller, H. EMBO J. 19, 589–597 (2000).
Goyal, L. Cell 104, 805–808 (2001).
Joazeiro, C. A. & Weissman, A. M. Cell 102, 549–552 (2000).
Grether, M. E., Abrams, J. M., Agapite, J., White, K. & Steller, H. Genes Dev. 9, 1694–1708 (1995).
Hay, B. A., Wassarman, D. A. & Rubin, G. M. Cell 83, 1253–1262 (1995).
White, K., Tahaoglu, E. & Steller, H. Science 271, 805–807 (1996).
Bergmann, A., Agapite, J., McCall, K. & Steller, H. Cell 95, 331–341 (1998).
Rigaut, G. et al. Nature Biotechnol. 17, 1030–1032 (1999).
Vucic, D., Kaiser, W. J. & Miller, L. K. Mol. Cell. Biol. 18, 3300–3309 (1998).
Hawkins, C. J. et al. J. Biol. Chem. 275, 27084–27093 (2000).
Meier, P., Silke, J., Leevers, S. J. & Evan, G. I. EMBO J. 19, 598–611 (2000).
Quinn, L. M. et al. J. Biol. Chem. 275, 40416–40424 (2000).
Fenteany, G. & Schreiber, S. L. J. Biol. Chem. 273, 8545–8548 (1998).
Yang, Y., Fang, S., Jensen, J. P., Weissman, A. M. & Ashwell, J. D. Science 288, 874–877 (2000).
Suzuki, Y., Nakabayashi, Y. & Takahashi, R. Proc. Natl Acad. Sci. USA 98, 8662–8667 (2001).
Huang, H. et al. J. Biol. Chem. 275, 26661–26664 (2000).
Pickart, C. M. Mol. Cell 8, 499–504 (2001).
Holley et al. Nature Cell Biol. DOI: 10.1038/ncb798.
Ryoo et al. Nature Cell Biol. DOI: 10.1038/ncb795.
Wing et al. Nature Cell Biol. DOI: 10.1038/ncb800.
Hays et al. Nature Cell Biol. DOI: 10.1038/ncb794.
Yoo et al. Nature Cell Biol. DOI: 10.1038/ncb793.
Acknowledgements
We wish to thank K. White for the generous gift of anti-DIAP1 antibody, D. Bohmann for the HA–ubiquitin construct and B. Seraphin for the TAP-construct. We thank T. Tenev for technical advice and graphical support and members of the laboratory for discussions. Furthermore, we thank members of the Evan and Downward laboratories for helpful discussions and support. We also thank S. Schneider for critical reading of the manuscript. We apologize to the scientists whose work we could not cite because of space limitations. H.S. is an investigator of the Howard Hughes Medical Institute. Part of this work was supported by National Institutes of Health grant RO1GM60124.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary figures and table
Figure S1. Characterization of diap1 mutant alleles in S2 cells. (PDF 172 kb)
Figure S2. The DRONC eye phenotype is severely enhanced in flies heterozygous for mutations in the DIAP1 RING finger or mutations that impair DIAP1 binding to DRONC.
Supplementary Table 1. The amino acid changes deduced from the nucleotide sequence of the diap1 gene of homozygous mutant embryos and their genetic properties. for GMR-hid.
Rights and permissions
About this article
Cite this article
Wilson, R., Goyal, L., Ditzel, M. et al. The DIAP1 RING finger mediates ubiquitination of Dronc and is indispensable for regulating apoptosis. Nat Cell Biol 4, 445–450 (2002). https://doi.org/10.1038/ncb799
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ncb799
This article is cited by
-
Wg/Wnt1 and Erasp link ER stress to proapoptotic signaling in an autosomal dominant retinitis pigmentosa model
Experimental & Molecular Medicine (2023)
-
The Hh pathway promotes cell apoptosis through Ci-Rdx-Diap1 axis
Cell Death Discovery (2021)
-
Non-apoptotic enteroblast-specific role of the initiator caspase Dronc for development and homeostasis of the Drosophila intestine
Scientific Reports (2021)
-
Toll signaling promotes JNK-dependent apoptosis in Drosophila
Cell Division (2020)
-
Ecdysone controlled cell and tissue deletion
Cell Death & Differentiation (2020)