Abstract
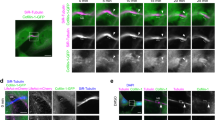
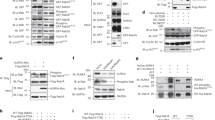
Abscission is the least understood step of cytokinesis. It consists of the final cut of the intercellular bridge connecting the sister cells at the end of mitosis, and is thought to involve membrane trafficking as well as lipid and cytoskeleton remodelling1,2,3,4,5,6. We previously identified the Rab35 GTPase as a regulator of a fast recycling endocytic pathway that is essential for post-furrowing cytokinesis stages7. Here, we report that the phosphatidylinositol-4,5-bisphosphate (PtdIns(4,5)P2) 5-phosphatase OCRL, which is mutated in Lowe syndrome patients8,9, is an effector of the Rab35 GTPase in cytokinesis abscission. GTP-bound (active) Rab35 directly interacts with OCRL and controls its localization at the intercellular bridge. Depletion of Rab35 or OCRL inhibits cytokinesis abscission and is associated with local abnormal PtdIns(4,5)P2 and F-actin accumulation in the intercellular bridge. These division defects are also found in cell lines derived from Lowe patients and can be corrected by the addition of low doses of F-actin depolymerization drugs. Our data demonstrate that PtdIns(4,5)P2 hydrolysis is important for normal cytokinesis abscission to locally remodel the F-actin cytoskeleton in the intercellular bridge. They also reveal an unexpected role for the phosphatase OCRL in cell division and shed new light on the pleiotropic phenotypes associated with Lowe disease.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Glotzer, M. The molecular requirements for cytokinesis. Science 307, 1735–1739 (2005).
Eggert, U. S., Mitchison, T. J. & Field, C. M. Animal cytokinesis: from parts list to mechanisms. Annu. Rev. Biochem. 75, 543–566 (2006).
Barr, F. A. & Gruneberg, U. Cytokinesis: placing and making the final cut. Cell 131, 847–860 (2007).
Echard, A. Membrane traffic and polarization of lipid domains during cytokinesis. Biochem. Soc. Trans. 36, 395–399 (2008).
Steigemann, P. & Gerlich, D. W. Cytokinetic abscission: cellular dynamics at the midbody. Trends Cell Biol. 19, 606–616 (2009).
Montagnac, G., Echard, A. & Chavrier, P. Endocytic traffic in animal cell cytokinesis. Curr. Opin. Cell Biol. 20, 454–461 (2008).
Kouranti, I., Sachse, M., Arouche, N., Goud, B. & Echard, A. Rab35 regulates an endocytic recycling pathway essential for the terminal steps of cytokinesis. Curr. Biol. 16, 1719–1725 (2006).
Lowe, M. Structure and function of the Lowe syndrome protein OCRL1. Traffic 6, 711–719 (2005).
Vicinanza, M., D’Angelo, G., Di Campli, A. & De Matteis, M. A. Function and dysfunction of the PI system in membrane trafficking. EMBO J. 27, 2457–2470 (2008).
Echard, A., Hickson, G. R., Foley, E. & O’Farrell, P. H. Terminal cytokinesis events uncovered after an RNAi screen. Curr. Biol. 14, 1685–1693 (2004).
Eggert, U. S. et al. Parallel chemical genetic and genome-wide RNAi screens identify cytokinesis inhibitors and targets. PLoS Biol. 2, e379 (2004).
Skop, A. R., Liu, H., Yates, J., III, Meyer, B. J. & Heald, R. Dissection of the mammalian midbody proteome reveals conserved cytokinesis mechanisms. Science 305, 61–66 (2004).
Lowe, C. U., Terrey, M. & Mac, L. E. Organic-aciduria, decreased renal ammonia production, hydrophthalmos, and mental retardation; a clinical entity. Am. J. Dis. Child 83, 164–184 (1952).
Attree, O. et al. The Lowe’s oculocerebrorenal syndrome gene encodes a protein highly homologous to inositol polyphosphate-5-phosphatase. Nature 358, 239–242 (1992).
Hyvola, N. et al. Membrane targeting and activation of the Lowe syndrome protein OCRL1 by rab GTPases. EMBO J. 25, 3750–3761 (2006).
Fukuda, M., Kanno, E., Ishibashi, K. & Itoh, T. Large scale screening for novel rab effectors reveals unexpected broad Rab binding specificity. Mol. Cell Proteomics 7, 1031–1042 (2008).
Hou, X. et al. A structural basis for Lowe syndrome caused by mutations in the Rab-binding domain of OCRL1. EMBO J. 30, 1659–1670 (2011).
Wilson, G. M. et al. The FIP3-Rab11 protein complex regulates recycling endosome targeting to the cleavage furrow during late cytokinesis. Mol. Biol. Cell 16, 849–860 (2005).
Choudhury, R. et al. Lowe syndrome protein OCRL1 interacts with clathrin and regulates protein trafficking between endosomes and the trans-Golgi network. Mol. Biol. Cell 16, 3467–3479 (2005).
Faucherre, A. et al. Lowe syndrome protein Ocrl1 is translocated to membrane ruffles upon Rac GTPase activation: a new perspective on Lowe syndrome pathophysiology. Hum. Mol. Genet. 14, 1441–1448 (2005).
Erdmann, K. S. et al. A role of the Lowe syndrome protein OCRL in early steps of the endocytic pathway. Dev. Cell 13, 377–390 (2007).
Choudhury, R., Noakes, C. J., McKenzie, E., Kox, C. & Lowe, M. Differential clathrin binding and subcellular localization of OCRL1 splice isoforms. J. Biol. Chem. 284, 9965–9973 (2009).
Sato, M. et al. Regulation of endocytic recycling by C. elegans Rab35 and its regulator RME-4, a coated-pit protein. EMBO J. 27, 1183–1196 (2008).
Zhang, J., Fonovic, M., Suyama, K., Bogyo, M. & Scott, M. P. Rab35 controls actin bundling by recruiting fascin as an effector protein. Science 325, 1250–1254 (2009).
Allaire, P. D. et al. The Connecdenn DENN domain: a GEF for Rab35 mediating cargo-specific exit from early endosomes. Mol. Cell 37, 370–382 (2010).
Hichri, H. et al. From Lowe syndrome to Dent disease: correlations between mutations of the OCRL1 gene and clinical and biochemical phenotypes. Hum. Mutat. 32, 379–388 (2011).
Zhang, X., Jefferson, A. B., Auethavekiat, V. & Majerus, P. W. The protein deficient in Lowe syndrome is a phosphatidylinositol-4,5-bisphosphate 5-phosphatase. Proc. Natl Acad. Sci. USA 92, 4853–4856 (1995).
Schmid, A. C., Wise, H. M., Mitchell, C. A., Nussbaum, R. & Woscholski, R. Type II phosphoinositide 5-phosphatases have unique sensitivities towards fatty acid composition and head group phosphorylation. FEBS Lett. 576, 9–13 (2004).
Hammond, G. R., Schiavo, G. & Irvine, R. F. Immunocytochemical techniques reveal multiple, distinct cellular pools of PtdIns4P and PtdIns(4,5)P(2). Biochem. J. 422, 23–35 (2009).
Begle, A., Tryoen-Toth, P., de Barry, J., Bader, M. F. & Vitale, N. ARF6 regulates the synthesis of fusogenic lipids for calcium-regulated exocytosis in neuroendocrine cells. J. Biol. Chem. 284, 4836–4845 (2009).
Di Paolo, G. & De Camilli, P. Phosphoinositides in cell regulation and membrane dynamics. Nature 443, 651–657 (2006).
Miserey-Lenkei, S. et al. Rab and actomyosin-dependent fission of transport vesicles at the Golgi complex. Nat. Cell Biol. 12, 645–654 (2010).
Ma, L., Cantley, L. C., Janmey, P. A. & Kirschner, M. W. Corequirement of specific phosphoinositides and small GTP-binding protein Cdc42 in inducing actin assembly in Xenopus egg extracts. J. Cell Biol. 140, 1125–1136 (1998).
Wong, R. et al. PIP2 hydrolysis and calcium release are required for cytokinesis in Drosophila spermatocytes. Curr. Biol. 15, 1401–1406 (2005).
Carlton, J. G. & Martin-Serrano, J. Parallels between cytokinesis and retroviral budding: a role for the ESCRT machinery. Science 316, 1908–1912 (2007).
Lee, H. H., Elia, N., Ghirlando, R., Lippincott-Schwartz, J. & Hurley, J. H. Midbody targeting of the ESCRT machinery by a noncanonical coiled coil in CEP55. Science 322, 576–580 (2008).
Guizetti, J. et al. Cortical constriction during abscission involves helices of ESCRT-III-dependent filaments. Science 331, 1616–1620 (2011).
Sagona, A. P. et al. PtdIns(3)P controls cytokinesis through KIF13A-mediated recruitment of FYVE-CENT to the midbody. Nat. Cell Biol. 12, 362–371 (2010).
Suchy, S. F. & Nussbaum, R. L. The deficiency of PIP2 5-phosphatase in Lowe syndrome affects actin polymerization. Am. J. Hum. Genet. 71, 1420–1427 (2002).
Kim, J. et al. Functional genomic screen for modulators of ciliogenesis and cilium length. Nature 464, 1048–1051 (2010).
Echard, A. et al. Interaction of a Golgi-associated kinesin-like protein with Rab6. Science 279, 580–585 (1998).
Nizak, C. et al. Recombinant antibodies against subcellular fractions used to track endogenous Golgi protein dynamics in vivo . Traffic 4, 739–753 (2003).
Racusen, L. C., Wilson, P. D., Hartz, P. A., Fivush, B. A. & Burrow, C. R. Renal proximal tubular epithelium from patients with nephropathic cystinosis: immortalized cell lines as in vitro model systems. Kidney Int. 48, 536–543 (1995).
Monnier, N., Satre, V., Lerouge, E., Berthoin, F. & Lunardi, J. OCRL1 mutation analysis in French Lowe syndrome patients: implications for molecular diagnosis strategy and genetic counseling. Hum. Mutat. 16, 157–165 (2000).
Uzan-Gafsou, S. et al. Rab11A controls the biogenesis of Birbeck granules by regulating Langerin recycling and stability. Mol. Biol. Cell 18, 3169–3179 (2007).
Oorschot, V., de Wit, H., Annaert, W. G. & Klumperman, J. A novel flat-embedding method to prepare ultrathin cryosections from cultured cells in their in situ orientation. J. Histochem. Cytochem. 50, 1067–1080 (2002).
Slot, J. W. & Geuze, H. J. Cryosectioning and immunolabeling. Nat. Protoc. 2, 2480–2491 (2007).
Acknowledgements
We thank R. Woscholski (Imperial College, London, UK), O. Dorseuil (Institut Cochin, Paris, France), A. Gautreau (CNRS Lebs, Gif, France), A. Alcover (Institut Pasteur, Paris), S. Miserey-Lenkei, P. Benaroch and F. Nagano (Institut Curie, Paris, France) for providing reagents and plasmids; V. Chauvet, A. Gautreau, A. Houdusse, B. Payrastre, N. Vitale and R. Weil for helpful discussions; S. Miserey-Lenkei and E. Crowell for critical reading of the manuscript; F. Legendre (Hôpital Necker, Paris, France) for the establishment of patients cell lines; and J. Lunardi (CHU Grenoble, France) and G. Baujat for the pediatric network. We thank the ‘Association du Syndrome de Lowe’, patients and parents. We thank the Plate-Forme d’Imagerie Dynamique (PFID) and Imagopole, Institut Pasteur, for microscopes and assistance. The authors thank V. Fraisier, L. Sengmanivong, J-B. Sibarita and J. Salamero for support in microscopy, and acknowledge the Nikon Imaging Centre at Institut Curie-CNRS. This work has been supported by the Institut PASTEUR (G5 program), the Institut CURIE, the CNRS, the INSERM, the Agence Nationale pour la Recherche (grants ANR-Maladies Rares, GIS-Maladies Rares to B.G. and R.S., and ANR 07-JCJC-0089 to A.E.) and the Schlumberger Foundation for Education and Research—FSER (A.E.). D.D. and M.M. have been supported by the Ministère de la Recherche et de l’Enseignement Supérieur, and D.D. and L.C. have been supported by the Association pour la Recherche sur le Cancer.
Author information
Authors and Affiliations
Contributions
D.D., M.M., L.C., M.S. and A.E. designed and analysed the experiments; D.D., M.M., L.C., M.S., M.R., A.E.M., E.F. and A.E. did the experimental work; R.S. and B.G. provided reagents; D.D., M.M., L.C., M.S., B.G. and A.E. wrote or edited the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
Supplementary Information (PDF 902 kb)
Rights and permissions
About this article
Cite this article
Dambournet, D., Machicoane, M., Chesneau, L. et al. Rab35 GTPase and OCRL phosphatase remodel lipids and F-actin for successful cytokinesis. Nat Cell Biol 13, 981–988 (2011). https://doi.org/10.1038/ncb2279
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ncb2279
This article is cited by
-
Cytokinetic abscission requires actin-dependent microtubule severing
Nature Communications (2024)
-
Vesicle-mediated transport of ALIX and ESCRT-III to the intercellular bridge during cytokinesis
Cellular and Molecular Life Sciences (2023)
-
Transcellular propagation of fibrillar α-synuclein from enteroendocrine to neuronal cells requires cell-to-cell contact and is Rab35-dependent
Scientific Reports (2022)
-
Rab35 governs apicobasal polarity through regulation of actin dynamics during sprouting angiogenesis
Nature Communications (2022)
-
Fine-tuning cell organelle dynamics during mitosis by small GTPases
Frontiers of Medicine (2022)