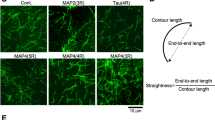
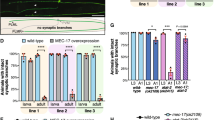
Abstract
Orchestrated remodelling of the cytoskeketon is prominent during neurite extension. In contrast with the extensive characterization of actin filament regulation, little is known about the dynamics of microtubules during neurite extension. Here we identify an atypical protein kinase C (aPKC)–Aurora A–NDEL1 pathway that is crucial for the regulation of microtubule organization during neurite extension. aPKC phosphorylates Aurora A at Thr 287 (T287), which augments interaction with TPX2 and facilitates activation of Aurora A at the neurite hillock, followed by phosphorylation of NDEL1 at S251 and recruitment. Suppression of aPKC, Aurora A or TPX2, or disruption of Ndel1, results in severe impairment of neurite extension. Analysis of microtubule dynamics with a microtubule plus-end marker revealed that suppression of the aPKC–Aurora A–NDEL1 pathway resulted in a significant decrease in the frequency of microtubule emanation from the microtubule organizing centre (MTOC), suggesting that Aurora A acts downstream of aPKC. These findings demonstrate a surprising role of aPKC–Aurora A–NDEL1 pathway in microtubule remodelling during neurite extension.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
References
Bentley, D. & O'Connor, T. P. Cytoskeletal events in growth cone steering. Curr. Opin. Neurobiol. 4, 43–48 (1994).
Tanaka, E. & Sabry, J. Making the connection: cytoskeletal rearrangements during growth cone guidance. Cell 83, 171–176 (1995).
Gallo, G. & Letourneau, P. C. Regulation of growth cone actin filaments by guidance cues. J. Neurobiol. 58, 92–102 (2004).
Dent, E. W. et al. Filopodia are required for cortical neurite initiation. Nature Cell Biol. 9, 1347–1359 (2007).
Kwiatkowski, A. V. et al. Ena/VASP is required for neuritogenesis in the developing cortex. Neuron 56, 441–455 (2007).
Lewis, A. K. & Bridgman, P. C. Nerve growth cone lamellipodia contain two populations of actin filaments that differ in organization and polarity. J. Cell Biol. 119, 1219–1243 (1992).
Svitkina, T. M. et al. Mechanism of filopodia initiation by reorganization of a dendritic network. J. Cell Biol. 160, 409–421 (2003).
Mallavarapu, A. & Mitchison, T. Regulated actin cytoskeleton assembly at filopodium tips controls their extension and retraction. J. Cell Biol. 146, 1097–1106 (1999).
Ridley, A. J. Rho GTPases and actin dynamics in membrane protrusions and vesicle trafficking. Trends Cell Biol. 16, 522–529 (2006).
Nobes, C. D. & Hall, A. Rho, rac, and cdc42 GTPases regulate the assembly of multimolecular focal complexes associated with actin stress fibers, lamellipodia, and filopodia. Cell 81, 53–62 (1995).
Dent, E. W. & Gertler, F. B. Cytoskeletal dynamics and transport in growth cone motility and axon guidance. Neuron 40, 209–227 (2003).
Gundersen, G. G. Evolutionary conservation of microtubule-capture mechanisms. Nature Rev. Mol. Cell Biol. 3, 296–304 (2002).
Fukata, M. et al. Rac1 and Cdc42 capture microtubules through IQGAP1 and CLIP-170. Cell 109, 873–885 (2002).
Krylyshkina, O. et al. Nanometer targeting of microtubules to focal adhesions. J. Cell Biol. 161, 853–859 (2003).
Fukata, Y. et al. CRMP-2 binds to tubulin heterodimers to promote microtubule assembly. Nature Cell Biol. 4, 583–591 (2002).
Zakharenko, S. & Popov, S. Dynamics of axonal microtubules regulate the topology of new membrane insertion into the growing neurites. J. Cell Biol. 143, 1077–1086 (1998).
Dobyns, W. B. The neurogenetics of lissencephaly. Neurol. Clin. 7, 89–105 (1989).
Dobyns, W. B., Reiner, O., Carrozzo, R. & Ledbetter, D. H. Lissencephaly. A human brain malformation associated with deletion of the LIS1 gene located at chromosome 17p13. J. Am. Med. Assoc. 270, 2838–2842 (1993).
Reiner, O. et al. Isolation of a Miller–Dieker lissencephaly gene containing G protein beta-subunit-like repeats. Nature 364, 717–721 (1993).
Wynshaw-Boris, A. Lissencephaly and LIS1: insights into the molecular mechanisms of neuronal migration and development. Clin. Genet. 72, 296–304 (2007).
Gupta, A., Tsai, L. H. & Wynshaw-Boris, A. Life is a journey: a genetic look at neocortical development. Nature Rev. Genet. 3, 342–355 (2002).
Niethammer, M. et al. NUDEL is a novel Cdk5 substrate that associates with LIS1 and cytoplasmic dynein. Neuron 28, 697–711 (2000).
Sasaki, S. et al. A LIS1/NUDEL/cytoplasmic dynein heavy chain complex in the developing and adult nervous system. Neuron 28, 681–696 (2000).
Mori, D. et al. NDEL1 phosphorylation by Aurora-A kinase is essential for centrosomal maturation, separation, and TACC3 recruitment. Mol. Cell. Biol. 27, 352–367 (2007).
Lindsay, R. M. Nerve growth factors (NGF, BDNF) enhance axonal regeneration but are not required for survival of adult sensory neurons. J. Neurosci. 8, 2394–2405 (1988).
Walter, A. O., Seghezzi, W., Korver, W., Sheung, J. & Lees, E. The mitotic serine/threonine kinase Aurora2/AIK is regulated by phosphorylation and degradation. Oncogene 19, 4906–4916 (2000).
Pasquale, E. B. Eph receptor signalling casts a wide net on cell behaviour. Nature Rev. Mol. Cell Biol. 6, 462–475 (2005).
Murai, K. K. & Pasquale, E. B. New exchanges in eph-dependent growth cone dynamics. Neuron 46, 161–163 (2005).
Nikolic, M. The molecular mystery of neuronal migration: FAK and Cdk5. Trends Cell Biol. 14, 1–5 (2004).
Zhang, X. et al. Dishevelled promotes axon differentiation by regulating atypical protein kinase C. Nature Cell Biol. 9, 743–754 (2007).
Chen, Y. M. et al. Microtubule affinity-regulating kinase 2 functions downstream of the PAR-3/PAR-6/atypical PKC complex in regulating hippocampal neuronal polarity. Proc. Natl Acad. Sci. USA 103, 8534–8539 (2006).
Xie, Z. et al. Activation of protein kinase C zeta by peroxynitrite regulates LKB1-dependent AMP-activated protein kinase in cultured endothelial cells. J. Biol. Chem. 281, 6366–6375 (2006).
Krystyniak, A., Garcia-Echeverria, C., Prigent, C. & Ferrari, S. Inhibition of Aurora A in response to DNA damage. Oncogene 25, 338–348 (2006).
Kufer, T. A. et al. Human TPX2 is required for targeting Aurora-A kinase to the spindle. J. Cell Biol. 158, 617–623 (2002).
Bayliss, R., Sardon, T., Vernos, I. & Conti, E. Structural basis of Aurora-A activation by TPX2 at the mitotic spindle. Mol. Cell 12, 851–862 (2003).
Ozlu, N. et al. An essential function of the C. elegans ortholog of TPX2 is to localize activated Aurora A kinase to mitotic spindles. Dev. Cell 9, 237–248 (2005).
Wittmann, T., Wilm, M., Karsenti, E. & Vernos, I. TPX2, A novel Xenopus MAP involved in spindle pole organization. J. Cell Biol. 149, 1405–1418 (2000).
Akimoto, K. et al. EGF or PDGF receptors activate atypical PKCλ through phosphatidylinositol 3-kinase. EMBO J. 15, 788–798 (1996).
Kotani, K. et al. Requirement of atypical protein kinase Cλ for insulin stimulation of glucose uptake but not for Akt activation in 3T3-L1 adipocytes. Mol. Cell. Biol. 18, 6971–6982 (1998).
Wirtz-Peitz, F., Nishimura, T. & Knoblich, J. A. Linking cell cycle to asymmetric division: Aurora-A phosphorylates the Par complex to regulate Numb localization. Cell 135, 161–173 (2008).
Mimori-Kiyosue, Y., Shiina, N. & Tsukita, S. The dynamic behavior of the APC-binding protein EB1 on the distal ends of microtubules. Curr. Biol. 10, 865–868 (2000).
de Anda, F. C. et al. Centrosome localization determines neuronal polarity. Nature 436, 704–708 (2005).
Yamada, M. et al. LIS1 and NDEL1 coordinate the plus-end-directed transport of cytoplasmic dynein. EMBO J. 27, 2471–2483 (2008).
Jan, Y. N. & Jan, L. Y. Asymmetric cell division in the Drosophila nervous system. Nature Rev. Neurosci. 2, 772–779 (2001).
Ohno, S. Intercellular junctions and cellular polarity: the PAR-aPKC complex, a conserved core cassette playing fundamental roles in cell polarity. Curr. Opin. Cell Biol. 13, 641–648 (2001).
Shi, S. H., Jan, L. Y. & Jan, Y. N. Hippocampal neuronal polarity specified by spatially localized mPar3/mPar6 and PI 3-kinase activity. Cell 112, 63–75 (2003).
Lee, C. Y. et al. Drosophila Aurora-A kinase inhibits neuroblast self-renewal by regulating aPKC/Numb cortical polarity and spindle orientation. Genes Dev. 20, 3464–3474 (2006).
Wang, H. et al. Aurora-A acts as a tumor suppressor and regulates self-renewal of Drosophila neuroblasts. Genes Dev. 20, 3453–3463 (2006).
Pugacheva, E. N., Jablonski, S. A., Hartman, T. R., Henske, E. P. & Golemis, E. A. HEF1-dependent Aurora A activation induces disassembly of the primary cilium. Cell 129, 1351–1363 (2007).
Kim, A. H. et al. A centrosomal Cdc20-APC pathway controls dendrite morphogenesis in postmitotic neurons. Cell 136, 322–336 (2009).
Brewer, G. J., Torricelli, J. R., Evege, E. K. & Price, P. J. Optimized survival of hippocampal neurons in B27-supplemented Neurobasal, a new serum-free medium combination. J. Neurosci. Res. 35, 567–576 (1993).
Acknowledgements
We thank Yukimi Kira, Yoriko Yabunaka, Gaku Kuwabara, Toshiyuki Kawashima and Takako Takitho for technical support, and Hiromichi Nishimura and Keiko Fujimoto for mouse breeding. This work was also supported by the Osaka Community Foundation to D.M. This work was supported by a Grant-in-Aid for Scientific Research from the Ministry of Education, Science, Sports and Culture of Japan to S.H. This work was also supported by the Cell Science Research Foundation, the Japan Spina Bifida and Hydrocephalus Research Foundation, the Takeda Science Foundation and the Hoh-ansha Foundation to S.H, and National Institutes of Health grants NS41030 and HD47380 to A.W.-B.
Author information
Authors and Affiliations
Contributions
D.M., M.Y., Y.S. and A.S. performed the experimental work, Y.M.-K., S.O. and H.S. conducted data analysis, and A.W.-B. and S.H. performed project planning and wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
Supplementary Information (PDF 1345 kb)
Supplementary Information
Supplementary Movie 1 (MOV 1037 kb)
Supplementary Information
Supplementary Movie 2 (MOV 1091 kb)
Supplementary Information
Supplementary Movie 3 (MOV 1022 kb)
Supplementary Information
Supplementary Movie 4 (MOV 1078 kb)
Supplementary Information
Supplementary Movie 5 (MOV 616 kb)
Rights and permissions
About this article
Cite this article
Mori, D., Yamada, M., Mimori-Kiyosue, Y. et al. An essential role of the aPKC–Aurora A–NDEL1 pathway in neurite elongation by modulation of microtubule dynamics. Nat Cell Biol 11, 1057–1068 (2009). https://doi.org/10.1038/ncb1919
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ncb1919
This article is cited by
-
Neurokinin-1 receptor drives PKCɑ-AURKA/N-Myc signaling to facilitate the neuroendocrine progression of prostate cancer
Cell Death & Disease (2023)
-
Targeting AURKA in Cancer: molecular mechanisms and opportunities for Cancer therapy
Molecular Cancer (2021)
-
High-throughput kinase inhibitor screening reveals roles for Aurora and Nuak kinases in neurite initiation and dendritic branching
Scientific Reports (2021)
-
Insights into the non-mitotic functions of Aurora kinase A: more than just cell division
Cellular and Molecular Life Sciences (2020)
-
Protein kinases: master regulators of neuritogenesis and therapeutic targets for axon regeneration
Cellular and Molecular Life Sciences (2020)