Abstract
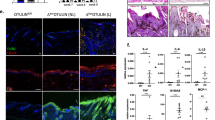
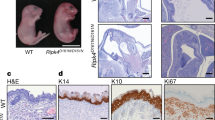
The kinase IKK1 (also known as IKKα) was previously reported to regulate epidermal development and skeletal morphogenesis by acting in keratinocytes to induce their differentiation in an NF-κB independent manner1,2,3,4,5. Here, we show that mice with epidermal keratinocyte-specific IKK1 ablation (hereafter referred to as IKK1EKO) develop a normally differentiated stratified epidermis, demonstrating that the function of IKK1 in inducing epidermal differentiation is not keratinocyte-autonomous. Despite normal epidermal stratification, the IKK1EKO mice display impaired epidermal-barrier function and increased transepidermal water loss, due to defects in stratum corneum lipid composition and in epidermal tight junctions. These defects are caused by the deregulation of retinoic acid target genes, encoding key lipid modifying enzymes and tight junction proteins, in the IKK1-deficient epidermis. Furthermore, we show that IKK1-deficient cells display impaired retinoic acid-induced gene transcription, and that IKK1 is recruited to the promoters of retinoic acid-regulated genes, suggesting that one mechanism by which IKK1 controls epidermal-barrier formation is by regulating the expression of retinoic acid receptor target genes in keratinocytes.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Hu, Y. et al. Abnormal morphogenesis but intact IKK activation in mice lacking the IKKα subunit of IκB kinase. Science 284, 316–320 (1999).
Li, Q. et al. IKK1-deficient mice exhibit abnormal development of skin and skeleton. Genes Dev. 13, 1322–1328 (1999).
Takeda, K. et al. Limb and skin abnormalities in mice lacking IKKα. Science 284, 313–316 (1999).
Hu, Y. et al. IKKα controls formation of the epidermis independently of NF-κB. Nature 410, 710–714 (2001).
Sil, A. K., Maeda, S., Sano, Y., Roop, D. R. & Karin, M. IαB kinase-α acts in the epidermis to control skeletal and craniofacial morphogenesis. Nature 428, 660–664 (2004).
Pasparakis, M. et al. TNF-mediated inflammatory skin disease in mice with epidermis-specific deletion of IKK2. Nature 417, 861–866 (2002).
Mills, A. A. et al. p63 is a p53 homologue required for limb and epidermal morphogenesis. Nature 398, 708–713 (1999).
Yang, A. et al. p63 is essential for regenerative proliferation in limb, craniofacial and epithelial development. Nature 398, 714–718 (1999).
Fisher, C. IKKα−/− mice share phenotype with pupoid fetus (pf/pf) and repeated epilation (Er/Er) mutant mice. Trends Genet. 16, 482–484 (2000).
Furuse, M. et al. Claudin-based tight junctions are crucial for the mammalian epidermal barrier: a lesson from claudin-1-deficient mice. J. Cell Biol. 156, 1099–1111 (2002).
Elias, P. M. et al. Desmoglein isoform distribution affects stratum corneum structure and function. J. Cell Biol. 153, 243–249 (2001).
Madison, K. C. Barrier function of the skin: “la raison d'etre” of the epidermis. J. Invest. Dermatol. 121, 231–241 (2003).
Zettersten, E. et al. Recessive x-linked ichthyosis: role of cholesterol-sulfate accumulation in the barrier abnormality. J. Invest. Dermatol. 111, 784–790 (1998).
Gurrieri, S. et al. Differentiation-dependent regulation of secreted phospholipases A2 in murine epidermis. J. Invest. Dermatol. 121, 156–164 (2003).
Bouwstra, J. A., Honeywell-Nguyen, P. L., Gooris, G. S. & Ponec, M. Structure of the skin barrier and its modulation by vesicular formulations. Prog. Lipid Res. 42, 1–36 (2003).
Bouwstra, J. A., Gooris, G. S., Dubbelaar, F. E., Weerheim, A. M. & Ponec, M. pH, cholesterol sulfate, and fatty acids affect the stratum corneum lipid organization. J. Investig. Dermatol. Symp. Proc. 3, 69–74 (1998).
Macheleidt, O., Kaiser, H. W. & Sandhoff, K. Deficiency of epidermal protein-bound omega-hydroxyceramides in atopic dermatitis. J. Invest. Dermatol. 119, 166–173 (2002).
Larcher, F., Murillas, R., Bolontrade, M., Conti, C. J. & Jorcano, J. L. VEGF/VPF overexpression in skin of transgenic mice induces angiogenesis, vascular hyperpermeability and accelerated tumor development. Oncogene 17, 303–311 (1998).
Li, Q., Lu, Q., Estepa, G. & Verma, I. M. Identification of 14-3-3sigma mutation causing cutaneous abnormality in repeated-epilation mutant mouse. Proc. Natl Acad. Sci. USA 102, 15977–15982 (2005).
Calleja, C. et al. Genetic and pharmacological evidence that a retinoic acid cannot be the RXR-activating ligand in mouse epidermis keratinocytes. Genes Dev. 20, 1525–1538 (2006).
Attar, P. S. et al. Inhibition of retinoid signaling in transgenic mice alters lipid processing and disrupts epidermal barrier function. Mol. Endocrinol. 11, 792–800 (1997).
Imakado, S. et al. Targeting expression of a dominant-negative retinoic acid receptor mutant in the epidermis of transgenic mice results in loss of barrier function. Genes Dev. 9, 317–329 (1995).
Hughes, P. J. et al. Up-regulation of steroid sulphatase activity in HL60 promyelocytic cells by retinoids and 1α,25-dihydroxyvitamin D3. Biochem. J. 355, 361–371 (2001).
Antonio, V., Janvier, B., Brouillet, A., Andreani, M. & Raymondjean, M. Oxysterol and 9-cis-retinoic acid stimulate the group IIA secretory phospholipase A2 gene in rat smooth-muscle cells. Biochem. J. 376, 351–360 (2003).
Kubota, H. et al. Retinoid X receptor α and retinoic acid receptor γ mediate expression of genes encoding tight-junction proteins and barrier function in F9 cells during visceral endodermal differentiation. Exp. Cell Res. 263, 163–172 (2001).
Humphries, J. D., Parry, E. J., Watson, R. E., Garrod, D. R. & Griffiths, C. E. All-trans retinoic acid compromises desmosome expression in human epidermis. Br. J. Dermatol. 139, 577–584 (1998).
Weninger, W., Rendl, M., Mildner, M. & Tschachler, E. Retinoids downregulate vascular endothelial growth factor/vascular permeability factor production by normal human keratinocytes. J. Invest. Dermatol. 111, 907–911 (1998).
Fisher, G. J. & Voorhees, J. J. Molecular mechanisms of retinoid actions in skin. Faseb J. 10, 1002–1013 (1996).
Giguere, V., Lyn, S., Yip, P., Siu, C. H. & Amin, S. Molecular cloning of cDNA encoding a second cellular retinoic acid-binding protein. Proc. Natl Acad. Sci USA 87, 6233–6237 (1990).
Nagpal, S. et al. Tazarotene-induced gene 1 (TIG1), a novel retinoic acid receptor-responsive gene in skin. J. Invest. Dermatol. 106, 269–274 (1996).
Park, K. J., Krishnan, V., O'Malley, B. W., Yamamoto, Y. & Gaynor, R. B. Formation of an IKKα-dependent transcription complex is required for estrogen receptor-mediated gene activation. Mol. Cell 18, 71–82 (2005).
Hoberg, J. E., Yeung, F. & Mayo, M. W. SMRT derepression by the IκB kinase α: a prerequisite to NF-κB transcription and survival. Mol. Cell 16, 245–255 (2004).
Chen, J. D. & Evans, R. M. A transcriptional co-repressor that interacts with nuclear hormone receptors. Nature 377, 454–457 (1995).
Nenci, A. et al. Skin lesion development in a mouse model of incontinentia pigmenti is triggered by NEMO deficiency in epidermal keratinocytes and requires TNF signaling. Hum. Mol. Genet. 15, 531–542 (2006).
Reichelt, J., Breiden, B., Sandhoff, K. & Magin, T. M. Loss of keratin 10 is accompanied by increased sebocyte proliferation and differentiation. Eur. J. Cell Biol. 83, 747–759 (2004).
Acknowledgements
We thank J. X. Neto for the retinoic acid reporter constructs, A. Aranda for the GST–CSMRT vector, and T. Krieg and C. Niessen for critical reading of the manuscript. The genomics core facility of the European Molecular Biology Laboratory (EMBL) assisted with the microarray experiments and data submission. This work was supported by the University of Cologne and by EMBL and by EU FP6 grant MUGEN (LSHG-CT-2005-005203) to M. P.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
Supplementary Figures S1, S2, S3, S4 and S5 (PDF 467 kb)
Rights and permissions
About this article
Cite this article
Gareus, R., Huth, M., Breiden, B. et al. Normal epidermal differentiation but impaired skin-barrier formation upon keratinocyte-restricted IKK1 ablation. Nat Cell Biol 9, 461–469 (2007). https://doi.org/10.1038/ncb1560
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ncb1560
This article is cited by
-
Deletion of IKK2 in haematopoietic cells of adult mice leads to elevated interleukin-6, neutrophilia and fatal gastrointestinal inflammation
Cell Death & Disease (2021)
-
Chondroitin 6-sulfate represses keratinocyte proliferation in mouse skin, which is associated with psoriasis
Communications Biology (2021)
-
Modes of genetic adaptations underlying functional innovations in the rumen
Science China Life Sciences (2021)
-
The RIPK4–IRF6 signalling axis safeguards epidermal differentiation and barrier function
Nature (2019)
-
IKKα is required in the intestinal epithelial cells for tumour stemness
British Journal of Cancer (2018)