Abstract
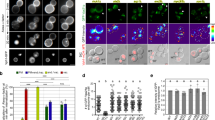
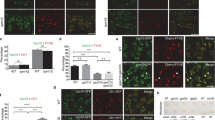
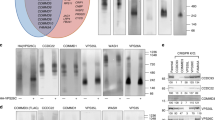
The recruitment of the small GTPase Arf6 and ARNO from cytosol to endosomal membranes is driven by V-ATPase-dependent intra-endosomal acidification. The molecular mechanism that mediates this pH-sensitive recruitment and its role are unknown. Here, we demonstrate that Arf6 interacts with the c-subunit, and ARNO with the a2-isoform of V-ATPase. The a2-isoform is targeted to early endosomes, interacts with ARNO in an intra-endosomal acidification-dependent manner, and disruption of this interaction results in reversible inhibition of endocytosis. Inhibition of endosomal acidification abrogates protein trafficking between early and late endosomal compartments. These data demonstrate the crucial role of early endosomal acidification and V-ATPase/ARNO/Arf6 interactions in the regulation of the endocytic degradative pathway. They also indicate that V-ATPase could modulate membrane trafficking by recruiting and interacting with ARNO and Arf6; characteristics that are consistent with the role of V-ATPase as an essential component of the endosomal pH-sensing machinery.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
References
Sorkin, A. & Von Zastrow, M. Signal transduction and endocytosis: close encounters of many kinds. Nature Rev. Mol. Cell. Biol. 3, 600–614 (2002).
Bonifacino, J.S. & Glick, B.S. The mechanisms of vesicle budding and fusion. Cell 116, 153–166 (2004).
Kirchhausen, T. Three ways to make a vesicle. Nature Rev. Mol. Cell. Biol. 1, 187–198 (2000).
Donaldson, J. G. Multiple roles for Arf6: Sorting, structuring, and signaling plasma membrane. J. Biol. Chem. 278, 41573–41576 (2003).
D'Souza-Schorey, C., Li, G., Colombo, M.I. & Stahl, P.D. A regulatory role for ARF6 in receptor-mediated endocytosis. Science 267, 1175–1178 (1995).
Paleotti, O. et al. The small G-protein Arf6GTP recruits the AP-2 adaptor complex to membranes. J. Biol. Chem. 280, 21661–21665 (2005).
Peters, P.J. et al. Characterization of coated vesicles that participate in endocytic recycling. Traffic 2, 885–895 (2001).
Radhakrishna, H., Klausner, R.D. & Donaldson, J.G. Aluminumfluoride stimulates surface protrusions in cells overexpressing the ARF6 GTPase. J. Cell. Biol. 134, 935–947 (1996).
Frank, S.R., Hatfield, J.C. & Casanova, J.E. Remodeling of the actin cytoskeleton is coordinately regulated by protein kinase C and the ADP-ribosylation factor nucleotide exchange factor ARNO. Mol. Biol. Cell. 9, 3133–3146 (1998).
Massenburg, D. et al. Activation of rat brain phospholipase D by ADP-ribosylation factors 1,5, and 6: separation of ADP-ribosylation factor-dependent and oleate-dependent enzymes. Proc. Natl Acad. Sci. USA 91, 11718–11722 (1994).
Santy, L.C. & Casanova, J.E. Activation of ARF6 by ARNO stimulates epithelial cell migration through downstream activation of both Rac1 and phospholipase D. J. Cell. Biol. 154, 599–610 (2001).
Mellman, I. The importance of being acid: the role of acidification in intracellular membrane traffic. J. Exp. Biol. 172, 39–45 (1992).
Mellman, I., Fuchs, R. & Helenius, A. Acidification of the endocytic and exocytic pathways. Annu. Rev. Biochem. 55, 663–700 (1986).
Trombetta, E. S., Ebersold, M., Garrett, W., Pypaert, M. & Mellman, I. Activation of lysosomal function during dendritic cell maturation. Science 299, 1400–1403 (2003).
Nishi, T. & Forgac, M. The vacuolar (H+)-ATPases-nature's most versatile proton pumps. Nature Rev. Mol. Cell. Biol. 3, 94–103 (2002).
Sun-Wada, G.H., Wada Y. & Futai, M. Lysosome and lysosome-related organelles responsible for specialized functions in higher organisms, with special emphasis on vacuolar-type proton ATPase. Cell. Struct. Funct. 28, 455–463 (2003).
Sun-Wada, G.H., Wada Y. & Futai, M. Diverse and essential roles of mammalian vacuolar-type proton pump ATPase: toward the physiological understanding of inside acidic compartments. Biochim. Biophys. Acta 1658, 106–114 (2004).
Toyomura, T., Oka, T., Yamaguchi, C., Wada, Y. & Futai, M. Three subunit a isoforms of mouse vacuolar H(+)-ATPase. Preferential expression of the a3 isoform during osteoclast differentiation. J. Biol. Chem. 275, 8760–8765 (2000).
Oka, T. et al. a4, a unique kidney-specific isoform of mouse vacuolar H+ATPase subunit a. J. Biol. Chem. 276, 40050–40054 (2001).
Toyomura, T. et al. From lysosomes to the plasma membrane: localization of vacuolar-type H+ATPase with the a3 isoform during osteoclast differentiation. J. Biol. Chem. 278, 22023–22030 (2003).
Marshansky, V., Fleser, A., Noel, J., Bourgoin, S. & Vinay, P. Isolation of heavy endosomes from dog proximal tubules in suspension. J. Membr. Biol. 153, 59–73 (1996).
Marshansky, V. & Vinay, P. Proton gradient formation in early endosomes from proximal tubules. Biochim. Biophys. Acta 1284, 171–180 (1996).
Marshansky, V. et al. Identification of ADP-ribosylation factor-6 in brush-border membrane and early endosomes of human kidney proximal tubules. Electrophoresis 18, 538–547 (1997).
Marshansky, V. et al. Receptor-mediated endocytosis in kidney proximal tubules: recent advances and hypothesis. Electrophoresis 18, 2661–2676 (1997).
Maranda, B. et al. Intra-endosomal pH-sensitive recruitment of the Arf-nucleotide exchange factor ARNO and Arf6 from cytoplasm to proximal tubule endosomes. J. Biol. Chem. 276, 18540–18550 (2001).
El Annan, J. et al. Differential expression and targeting of endogenous Arf1 and Arf6 small GTPases in kidney epithelial cell in situ. Am. J. Physiol. (Cell Physiol) 286, C768–C778 (2004).
Marshansky, V., Ausiello, D. A. & Brown, D. Physiological importance of endosomal acidification: potential role in proximal tubulopathies. Curr. Opin. Nephrol. Hypertens. 11, 527–537 (2002).
Brown, D., & Marshansky, V. in Handbook of ATPases. (eds Futai, M., Wada, Y. & Kaplan, J.H.) 413–442 (Wiley-VCH, New York, 2004).
Zeuzem, S., Zimmermann, P. & Schulz, I. Association of a 19- and a 21-kDa GTP-binding protein to pancreatic microsomal vesicles is regulated by the intravesicular pH established by a vacuolar-type H(+)-ATPase. J. Membr. Biol. 125, 231–241 (1992).
Zeuzem, S. et al. Intravesicular acidification correlates with binding of ADP-ribosylation factor to microsomal membranes. Proc. Natl Acad. Sci. USA 89, 6619–6623 (1992).
Aniento, F., Gu, F., Parton, R. G. & Gruenberg, J. An endosomal beta COP is involved in the pH-dependent formation of transport vesicles destined for late endosomes. J. Cell Biol. 133, 29–41 (1996).
Gu, F. & Gruenberg, J. Biogenesis of transport intermediates in the endocytic pathway. FEBS Lett. 452, 61–66 (1999).
Gu, F. & Gruenberg, J. ARF1 regulates pH-dependent COP functions in the early endocytic pathway. J. Biol. Chem. 275, 8154–8160 (2000).
Chardin, P. et al. A human exchange factor for ARF contains Sec7- and pleckstrin-homology domains. Nature 384, 481–484 (1996).
Mansour, M., Lee, S. Y. & Pohajdak, B. The N-terminal coiled coil domain of the cytohesin/ARNO family of guanine nucleotide exchange factors interacts with the scaffolding protein CASP. J. Biol. Chem. 277, 32302–32309 (2002).
Santy, L. C., Frank, S. R., Hatfield, J. C. & Casanova, J. E. Regulation of ARNO nucleotide exchange by a PH domain electrostatic switch. Curr. Biol. 9, 1173–1176 (1999).
Sun-Wada, G.H. et al. A proton pump ATPase with testis-specific E1-subunit isoform required for acrosome acidification. J. Biol. Chem. 277, 18098–18105 (2002).
D'Souza-Schorey, C. & Stahl, P.D. Myristoylation is required for the intracellular localization and endocytic function of ARF6. Exp. Cell. Res. 221, 153–159 (1995).
Christensen, E. I. & Birn, H. Megalin and cubilin: multifunctional endocytotic receptors. Nature Rev. Mol. Cell. Biol. 3, 258–268 (2002).
Wagner, C. A. et al. Renal vacuolar H+-ATPase. Physiol. Rev. 84, 1263–1314 (2004).
Jentsch, T. J., Hubner, C. A. & Fuhrmann, J. C. Ion channels: Function unraveled by dysfunction. Nature Cell. Biol. 6, 1039–1047 (2004).
Baravalle, G. et al. Transferrin recycling and dextran transport to lysosomes is differentially affected by bafilomycin, nocodazole, and low temperature. Cell. Tissue Res. 320, 99–113 (2005).
Donaldson, J.G., Honda, A. & Weigert, R. Multiple activities for Arf1 at the Golgi complex. Biochim. Biophys. Acta 1744, 364–373 (2005).
Shao, E. & Forgac, M. Involvement of the nonhomologous region of subunit A of the yeast V-ATPase in coupling and in vivo dissociation. J. Biol. Chem. 279, 48663–48670 (2004).
Radhakrishna, H. & Donaldson, J.G. ADP-ribosylation factor 6 regulates a novel plasma membrane recycling pathway. J. Cell. Biol. 139, 49–61 (1997).
Skinner, M.A. & Wildeman, A.G. Suppression of tumor-related glycosylation of cell surface receptors by the 16-kDa membrane subunit of vacuolar H+ATPase. J. Biol. Chem. 276, 48451–48457 (2001).
Vinay, P., Gougoux, A. & Lemieux, G. Isolation of a pure suspension of rat proximal tubules. Am. J. Physiol. 241, F403–F411 (1981).
Sun-Wada, G. H. et al. Mouse proton pump ATPase C subunit isoforms (C2-a and C2-b) specifically expressed in kidney and lung. J. Biol. Chem. 278, 44843–44851 (2003).
Zhai X.Y. et al. Cubilin- and megalin-mediated uptake of albumin in cultured proximal tubule cells of opossum kidney. Kidney Int. 58, 1523–1533 (2000).
Hryciw, D.H. et al. Cofilin interacts with ClC-5 and regulates albumin uptake in proximal tubule cell lines. J. Biol. Chem. 278, 40169–40176 (2003).
Acknowledgements
We would like to thank C. Reinecker for generously providing Rab7–EGFP and Rab11–EGFP constructs and for advice on Volocity software. We would like to thank J. Donaldson for generously providing the Arf6–HA construct and M.A. Billeter for kindly providing the HEK cell line expressing T7-polymerase. We also wish to thank H. Huang for expert technical assistance on FACS analysis. We are grateful to V. Hsu for critical discussion and reading of the manuscript. M.F. and G.-H.S.-W. were supported by CREST, the Japan Science and Technology Agency. This work was supported by a National Institutes of Health grants (DK38452 and DK42956). The Microscopy Core facility of the MGH Program in Membrane Biology receives additional support from the Boston Area Diabetes and Endocrinology Research Center (DK57521) and the Center for the Study of Inflammatory Bowel Disease (DK43341).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
Supplementary methods, references, figures S1 and S2, movie legends (PDF 146 kb)
Rights and permissions
About this article
Cite this article
Hurtado-Lorenzo, A., Skinner, M., Annan, J. et al. V-ATPase interacts with ARNO and Arf6 in early endosomes and regulates the protein degradative pathway. Nat Cell Biol 8, 124–136 (2006). https://doi.org/10.1038/ncb1348
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ncb1348
This article is cited by
-
The pathophysiology of distal renal tubular acidosis
Nature Reviews Nephrology (2023)
-
The lysosomal V-ATPase a3 subunit is involved in localization of Mon1-Ccz1, the GEF for Rab7, to secretory lysosomes in osteoclasts
Scientific Reports (2022)
-
The a subunit isoforms of vacuolar-type proton ATPase exhibit differential distribution in mouse perigastrulation embryos
Scientific Reports (2022)
-
Genetic architecture and phenotypic landscape of deafness and onychodystrophy syndromes
Human Genetics (2022)
-
Vacuolar-type proton ATPase is required for maintenance of apicobasal polarity of embryonic visceral endoderm
Scientific Reports (2021)