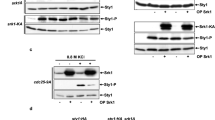
Abstract
In Saccharomyces cerevisiae cells, high external osmolarity leads to the activation of a p38-related mitogen-activated protein (MAP) kinase though Pbs2. Pbs2 tagged with green fluorescent protein (Pbs2–GFP) is evenly distributed in the cytoplasm but excluded from the nucleus before and after exposure to stress. Here we show that a catalytically inactive form of Pbs2 attains a highly polarised localization during osmostress. This phenomenon depends of the osmosensor Sho1 and on a functional Cdc42 GTPase. Cdc42, but not the actin cytoskeleton, influences Sho1-dependent activation of the MAP kinase. Sho1 itself accumulates at sites of polar growth, but independently of stress conditions and Cdc42. These observations allow us to define the sequence of events that occurs during propogation of osmostress signals.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Toone, W. M. & Jones, N. Stress-activated signalling pathways in yeast. Genes Cells 3, 485– 498 (1998).
Posas, F., Takekawa, M. & Saito, H. Signal transduction by MAP kinase cascades in budding yeast. Curr. Opin. Microbiol. 1, 175– 182 (1998).
Kyriakis, J. M. Making the connection: coupling of stress-activated ERK/MAPK (extracellular-signal-regulated kinase/mitogen-activated protein kinase) core signalling modules to extracellular stimuli and biological responses. Biochem. Soc. Symp. 64, 29– 48 (1999).
Maeda, T., Wurgler-Murphy, S. M. & Saito, H. A two-component system that regulates an osmosensing MAP kinase cascade in yeast. Nature 369, 242– 245 (1994).
Maeda, T., Takekawa, M. & Saito, H. Activation of yeast PBS2 MAPKK by MAPKKKs or by binding of an SH3-containing osmosensor. Science 269, 554– 558 (1995).
Posas, F. & Saito, H. Osmotic activation of the HOG MAPK pathway via Ste11p MAPKKK: scaffold role of Pbs2p MAPKK. Science 276, 1702– 1705 (1997).
Roberts, R. & Fink, G. R. Elements of a single MAP kinase cascade in Saccharomyces cerevisiae mediate two developmental programs in the same cell type: mating and invasive growth. Genes Dev. 15, 2974– 2985 (1994).
Whitmarsh, A. J. & Davis, R. J. Structural organization of MAP-kinase signaling modules by scaffold proteins in yeast and mammals. Trends Biochem. Sci. 23, 481– 485 (1998).
Garrington, T. P. & Johnson, G. L. Organization and regulation of mitogen-activated protein kinase signalling pathways. Curr. Opin. Cell Biol. 11, 211– 218 (1999)
Kjoller, L. & Hall, A. Signalling to Rho GTPases. Exp. Cell Res. 253, 166– 179 (1999).
Simon, M. N. et al. Role for the Rho-family GTPase Cdc42 in yeast mating-pheromone signal pathway. Nature 376, 702– 705 (1995).
Zhao, Z. S., Leung, T., Manser, E. & Lim, L. Pheromone signalling in Saccharomyces cerevisiae requires the small GTP-binding protein Cdc42p and its activator CDC24. Mol. Cell Biol. 15, 5246– 5257 (1995).
Mosch, H. U., Roberts, R. L. & Fink, G. R. Ras2 signals via the Cdc42/Ste20/mitogen-activated protein kinase module to induce filamentous growth in Saccharomyces cerevisiae. Proc. Natl Acad. Sci. USA 28, 5352– 5356 (1996).
Pryciak, P. M. & Huntress, F. A. Membrane recruitment of the kinase cascade scaffold protein Ste5 by the Gβγ complex underlies activation of the yeast pheromone response pathway. Genes Dev. 12, 2684– 2697 (1998).
Leberer, E. et al. Functional characterization of the Cdc42p binding domain of yeast Ste20p protein kinase. EMBO J. 16, 83– 97 (1997).
Peter, M., Neiman, A. M., Park, H. O., van Lohuizen, M. & Herskowitz, I. Functional analysis of the interaction between the small GTP binding protein Cdc42 and the Ste20 protein kinase in yeast. EMBO J. 24, 7046– 7059 (1996).
Oehlen, L. J. W. M. & Cross, F. R. The role of Cdc42 in signal transduction and mating of the budding yeast Saccharomyces cerevisiae. J. Biol. Chem. 273, 8556– 8559 (1998).
Ferrigno, P., Posas, F., Koepp, D., Saito, H. & Silver, P. A. Regulated nucleo/cytoplasmic exchange of HOG1 MAPK requires the importin beta homologs NMD5 and XPO1. EMBO J. 17, 5606– 5614 (1998).
Reiser, V., Ruis, H. & Ammerer, G. Kinase activity-dependent nuclear export opposes stress-induced nuclear accumulation and retention of Hog1 mitogen-activated protein kinase in the budding yeast Saccharomyces cerevisiae. Mol. Biol. Cell 10, 1147– 1161 (1999).
Ayscough, K. R. et al. High rates of actin filament turnover in budding yeast and roles for actin in establishment and maintenance of cell polarity revealed using the actin inhibitor latrunculin-A. J. Cell Biol. 137, 399– 416 (1997).
Palmieri, S. J. & Haarer, B. K. Polarity and division site specification in yeast. Curr. Opin. Microbiol. 6, 678– 686 (1998).
Johnson, D. I. Cdc42: an essential Rho-type GTPase controlling eukaryotic cell polarity. Microbiol. Mol. Biol. Rev. 63, 54– 105 (1999).
Tjandra, H., Compton, J. & Kellogg, D. Control of mitotic events by the Cdc42 GTPase, the Clb2 cyclin and a member of the PAK kinase family. Curr. Biol. 10, 991– 1000 (1998).
O'Rourke, S. M. & Herskowitz, I. The Hog1 MAPK prevents cross talk between the HOG and pheromone response MAPK pathways in Saccharomyces cerevisiae. Genes Dev. 12, 2874– 2886 (1998).
Posas, F., Witten, E. A. & Saito, H. Requirement of STE50 for osmostress-induced activation of the STE11 mitogen-activated protein kinase kinase kinase in the high-osmolarity glycerol response pathway. Mol. Cell. Biol. 18, 5788– 5796 (1998).
Miller, P. J. & Johnson, D. I. Characterization of the Saccharomyces cerevisiae cdc42-1 ts allele and new temperature-conditional-lethal cdc42 alleles. Yeast 13, 561– 572 (1997).
Kozminski, K. G., Chen, A. J., Rodal, A. A., & Drubin, D. G. Functions and functional domains of the GTPase Cdc42p. Mol. Biol. Cell 11, 339– 354 (2000).
Cook, J. G., Bardwell, L. & Thorner, J. Inhibitory and activating functions for MAPK Kss1 in the S. cerevisiae filamentous-growth signalling pathway. Nature 390, 85– 88 (1997).
Madhani, H. D., Styles, C. A. & Fink, G. R. MAP kinases with distinct inhibitory functions impart signaling specificity during yeast differentiation. Cell 91, 673– 684 (1997).
Rose, M. D., Winston, F. & Hieter, P. Methods in Yeast Genetics: A Laboratory Course Manual (Cold Spring Harbor Laboratory Press, New York, 1990).
Knop, M. et al. Epitope tagging of yeast genes using a PCR-based strategy: more tags and improved practical routines. Yeast 15, 963– 972 (1999).
Sambrook, J., Fritsch, E. F. & Maniatis, T. Molecular Cloning: A Laboratory Manual 2nd ed. {AU: chapter/page no?} (Cold Spring Harbor Laboratory Press, New York, 1989).
Higuchi, R. in PCR Protocols, A Guide to Methods and Applications (eds Innis, M. A., Gelfand, D. H. & Sninsky, J. J.) 177– 183 (Academic, New York, 1990).
Gietz, R. D. & Sugino, A. New yeast–Escherichia coli shuttle vectors constructed with in vitro mutagenized yeast genes lacking six-base pair restriction sites. Gene 74, 527– 534 (1988).
Acknowledgements
We thank D. Drubin, K. Nasmyth, M. Ramezani Rad, S. O'Rourke and H. Saito for strains and plasmids, H. Ruis and all the members of the Ruis and Ammerer laboratories for discussions and material support, and L. Huber and P. Kovarik for comments on the manuscript. We are greatful to S. Reipert for help with the laser-scanning microscope. Latrunculin A was kindly supplied by M. Sanders. This work was partly supported by TMR Network grant ERBFMRX-CT96-0041 (to G.A.) and by student program grant W001 from the Austrian Fonds zur Förderung Wissenschaftlicher Forschung.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Reiser, V., Salah, S. & Ammerer, G. Polarized localization of yeast Pbs2 depends on osmostress, the membrane protein Sho1 and Cdc42. Nat Cell Biol 2, 620–627 (2000). https://doi.org/10.1038/35023568
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/35023568
This article is cited by
-
A framework for mapping, visualisation and automatic model creation of signal‐transduction networks
Molecular Systems Biology (2012)
-
The mitogen-activated protein kinase kinase kinase BcOs4 is required for vegetative differentiation and pathogenicity in Botrytis cinerea
Applied Microbiology and Biotechnology (2012)
-
The Role of sho1 in Polarized Growth of Aspergillus fumigatus
Mycopathologia (2011)
-
Integrated cellular network of transcription regulations and protein-protein interactions
BMC Systems Biology (2010)
-
Transmembrane mucins Hkr1 and Msb2 are putative osmosensors in the SHO1 branch of yeast HOG pathway
The EMBO Journal (2007)