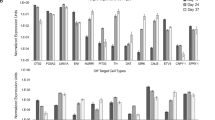
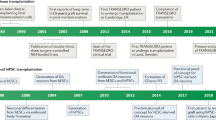
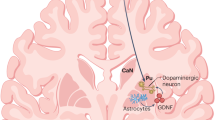
Abstract
Multipotent neural stem cells, capable of giving rise to both neurons and glia, line the cerebral ventricles of all adult animals, including humans. In addition, distinct populations of nominally glial progenitor cells, which also have the capacity to generate several cell types, are dispersed throughout the subcortical white matter and cortex. A number of approaches have evolved for using neural progenitor cells in cell therapy. Four strategies are especially attractive for clinical translation: first, transplantation of oligodendrocyte progenitor cells as a means of treating the disorders of myelin; second, transplantation of phenotypically restricted neuronal progenitor cells to treat diseases of discrete loss of a single neuronal phenotype, such as Parkinson disease; third, implantation of mixed progenitor pools to treat diseases characterized by the loss of several discrete phenotypes, such as spinal cord injury; and fourth, mobilization of endogenous neural progenitor cells to restore neurons lost as a result of neurodegenerative diseases, in particular Huntington disease. Together, these may present the most compelling strategies and near-term disease targets for cell-based neurological therapy.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Katie Ris

Katie Ris


Katie Ris
Similar content being viewed by others
References
Goldman, S. Adult neurogenesis: from canaries to the clinic. J. Neurobiol. 36, 267–286 (1998).
Gage, F.H. Neurogenesis in the adult brain. J. Neurosci. 22, 612–613 (2002).
Kirschenbaum, B. et al. In vitro neuronal production and differentiation by precursor cells derived from the adult human forebrain. Cereb. Cortex 4, 576–589 (1994).
Pincus, D.W. et al. Fibroblast growth factor-2/brain-derived neurotrophic factor-associated maturation of new neurons generated from adult human subependymal cells. Ann. Neurol. 43, 576–585 (1998).
Roy, N.S. et al. In vitro neurogenesis by progenitor cells isolated from the adult human hippocampus. Nat. Med. 6, 271–277 (2000).
Sanai, N. et al. Unique astrocyte ribbon in adult human brain contains neural stem cells but lacks chain migration. Nature 427, 740–744 (2004).
Lindvall, O., Kokaia, Z. & Martinez-Serrano, A. Stem cell therapy for human neurodegenerative disorders. Nat. Med. 10 suppl., S42–S50 (2004).
Goldman, S.A. Directed mobilization of endogenous neural progenitor cells: the intersection of stem cell biology and gene therapy. Curr. Opin. Mol. Ther. 6, 466–472 (2004).
Gage, F. Mammalian neural stem cells. Science 287, 1433–1438 (2000).
Alvarez-Buylla, A. & Garcia-Verdugo, J.M. Neurogenesis in adult subventricular zone. J. Neurosci. 22, 629–634 (2002).
Morshead, C.M. et al. Neural stem cells in the adult mammalian forebrain: a relatively quiescent subpopulation of subependymal cells. Neuron 13, 1071–1082 (1994).
Weiss, S. et al. Multipotent CNS stem cells are present in the adult mammalian spinal cord and ventricular neuroaxis. J. Neurosci. 16, 7599–7609 (1996).
Luskin, M.B. Restricted proliferation and migration of postnatally generated neurons derived from the forebrain subventricular zone. Neuron 11, 173–189 (1993).
Lois, C. & Alvarez-Buylla, A. Proliferating subventricular zone cells in the adult mammalian forebrain can differentiate into neurons and glia. Proc. Natl. Acad. Sci. USA 90, 2074–2077 (1993).
Levine, J.M., Reynolds, R. & Fawcett, J.W. The oligodendrocyte precursor cell in health and disease. Trends Neurosci. 24, 39–47 (2001).
Palmer, T.D., Markakis, E.A., Willhoite, A.R., Safar, F. & Gage, F.H. Fibroblast growth factor-2 activates a latent neurogenic program in neural stem cells from diverse regions of the adult CNS. J. Neurosci. 19, 8487–8497 (1999).
Roy, N.S. et al. Identification, isolation, and promoter-defined separation of mitotic oligodendrocyte progenitor cells from the adult human subcortical white matter. J. Neurosci. 19, 9986–9995 (1999).
Kondo, T. & Raff, M. Oligodendrocyte precursor cells reprogrammed to become multipotential CNS stem cells. Science 289, 1754–1757 (2000).
Nunes, M.C. et al. Identification and isolation of multipotential neural progenitor cells from the subcortical white matter of the adult human brain. Nat. Med. 9, 439–447 (2003).
Belachew, S. et al. Postnatal NG2 proteoglycan-expressing progenitor cells are intrinsically multipotent and generate functional neurons. J. Cell Biol. 161, 169–186 (2003).
Pincus, D.W. et al. In vitro neurogenesis by adult human epileptic temporal neocortex. Clin. Neurosurg. 44, 17–25 (1997).
Kukekov, V. et al. Multipotent stem/progenitor cells with similar properties arise from two neurogenic regions of adult human brain. Exp. Neurol. 156, 333–344 (1999).
Roy, N.S. et al. Promoter-targeted selection and isolation of neural progenitor cells from the adult human ventricular zone. J. Neurosci. Res. 59, 321–331 (2000).
Arsenijevic, Y. et al. Isolation of multipotent neural precursors residing in the cortex of the adult human brain. Exp. Neurol. 170, 48–62 (2001).
Palmer, T. et al. Progenitor cells from human brain after death. Nature 411, 42–43 (2001).
Goldman, S. Glia as neural progenitor cells. Trends Neurosci. 26, 590–596 (2003).
Goldman, S. & Sim, F. Neural progenitor cells of the adult forebrain. in Stem Cells: Nuclear Programming and Therapeutic Applications. Novartis Foundation Symposium 265, 66–92 (John Wiley, London, 2005).
Watt, F.M. Stem cell fate and patterning in mammalian epidermis. Curr. Opin. Genet. Dev. 11, 410–417 (2001).
Niemann, C. & Watt, F.M. Designer skin: lineage commitment in postnatal epidermis. Trends Cell Biol. 12, 185–192 (2002).
Loeffler, M. & Potten, C.S. Stem cells and cellular pedigrees. in Stem Cells (ed. Potten, C.S.) 1–28 (Academic Press, San Diego, 1997).
Potten, C.S. & Loeffler, M. Stem cells: attributes, cycles, spirals, pitfalls and uncertainties. Lessons for and from the crypt. Development 110, 1001–1020 (1990).
Garcia-Verdugo, J., Doetsch, F., Wichterle, H. & Alvarez-Buylla, A. Architecture and cell types of the adult subventricular zone: in search of the stem cells. J. Neurobiol. 36, 234–248 (1998).
Doetsch, F., Petreanu, L., Caille, I., Garcia-Verdugo, J. & Alvarez-Buylla, A. EGF converts transit-amplifying neurogenic precursors in the adult brain into multipotent stem cells. Neuron 36, 1021–1034 (2002).
Menezes, J.R., Smith, C.M., Nelson, K.C. & Luskin, M.B. The division of neuronal progenitor cells during migration in the neonatal mammalian forebrain. Mol. Cell. Neurosci. 6, 496–508 (1995).
Palmer, T.D., Takahashi, J. & Gage, F.H. The adult rat hippocampus contains primordial neural stem cells. Mol. Cell. Neurosci. 8, 389–404 (1997).
Shihabuddin, L. et al. Intracerebral transplantation of adult mouse neural progenitor cells into the Niemann-Pick-A mouse leads to a marked decrease in lysosomal storage pathology. J. Neurosci. 24, 10642–10651 (2004).
Vescovi, A.L., Reynolds, B.A., Fraser, D.D. & Weiss, S. bFGF regulates the proliferative fate of unipotent (neuronal) and bipotent (neuronal/astroglial) EGF-generated CNS progenitor cells. Neuron 11, 951–966 (1993).
Palmer, T.D., Ray, J. & Gage, F.H. FGF-2-responsive neuronal progenitors reside in proliferative and quiescent regions of the adult rodent brain. Mol. Cell. Neurosci. 6, 474–486 (1995).
Gritti, A. et al. Multipotential stem cells from the adult mouse brain proliferate and self-renew in response to basic fibroblast growth factor. J. Neurosci. 16, 1091–1100 (1996).
Flax, J. et al. Engraftable human neural stem cells respond to developmental cues, replace neurons, and express foreign genes. Nat. Biotechnol. 16, 1033–1039 (1998).
Keyoung, H.M. et al. High-yield selection and extraction of two promoter-defined phenotypes of neural stem cells from the fetal human brain. Nat. Biotechnol. 19, 843–850 (2001).
Vescovi, A. et al. Isolation and cloning of multipotential stem cells from the embryonic human CNS and establishment of transplantable human stem cells lines by epigenetic stimulation. Exp. Neurol. 156, 71–83 (1999).
Carpenter, M. et al. In vitro expansion of a multipotent population of human neural progenitor cells. Exp. Neurol. 158, 265–278 (1999).
Svendsen, C., Caldwell, M. & Ostenfeld, T. Human neural stem cells: isolation, expansion and transplantation. Brain Pathol. 9, 499–513 (1999).
Brustle, O. et al. Chimeric brains generated by intraventricular transplantation of fetal human brain cells into embryonic rats. Nat. Biotechnol. 16, 1040–1044 (1998).
Fricker, R. et al. Site-specific migration and neuronal differentiation of human neural progenitor cells after transplantation in the adult rat brain. J. Neurosci. 19, 5990–6005 (1999).
Uchida, N. et al. Direct isolation of human central nervous system stem cells. Proc. Natl. Acad. Sci. USA 97, 14720–14725 (2000).
Lee, A. et al. Isolation of neural stem cells from the postnatal cerebellum. Nat. Neurosci. 8, 723–729 (2005).
Rietze, R. et al. Purification of a pluripotent neural stem cell from the adult mouse brain. Nature 412, 736–739 (2001).
Capela, A. & Temple, S. LeX/ssea-1 is expressed by adult mouse CNS stem cells, identifying them as nonependymal. Neuron 35, 865–875 (2002).
Wang, S. et al. Isolation of neuronal precursors by sorting embryonic forebrain transfected with GFP regulated by the T alpha 1 tubulin promoter. Nat. Biotechnol. 16, 196–201 (1998).
Chalfie, M., Tu, Y., Euskirchen, G., Ward, W. & Prasher, D. Green fluorescent protein as a marker for gene expression. Science 263, 802–805 (1994).
Gloster, A. et al. The Tα1-tubulin promoter specifies gene expression as a function of neuronal growth and regeneration in transgenic mice. J. Neurosci. 14, 7319–7330 (1994).
Kawaguchi, A. et al. Nestin-GFP transgenic mice: Visualization of the self-renewal and multipotency of CNS stem cells. Mol. Cell. Neurosci. 17, 259–273 (2001).
Wang, S., Roy, N.S., Benraiss, A. & Goldman, S.A. Promoter-based isolation and fluorescence-activated sorting of mitotic neuronal progenitor cells from the adult mammalian ependymal/subependymal zone. Dev. Neurosci. 22, 167–176 (2000).
Mignone, J., Kukekov, V., Chiang, A., Steindler, D. & Enikolopov, G. Neural stem and progenitor cells in nestin-GFP transgenic mice. J. Comp. Neurol. 469, 311–324 (2004).
Scolding, N. et al. Oligodendrocyte progenitors are present in the normal adult human CNS and in the lesions of multiple sclerosis. Brain [see comments] 121, 2221–2228 (1998).
Kaye, E. Update on genetic disorders affecting white matter. Pediatr. Neurol. 24, 11–24 (2001).
Powers, J. The leukodystrophies: overview and classification. in Myelin Biology and Disorders, vol. 2 (ed. Lazzarini, R.A.) 663–690 (Elsevier Academic Press, San Diego, 2004).
Back, S. & Rivkees, S. Emerging concepts in periventricular white matter injury. Semin. Perinatol. 6, 405–414 (2004).
Follett, P. et al. Glutamate receptor-mediated oligodendrocyte toxicity in periventricular leukomalacia: a protective role for topiramate. J. Neurosci. Res. 24, 4412–4420 (2004).
Robinson, S. et al. Developmental changes induced by graded prenatal systemic hypoxic-ischemic insults in rats. Neurobiol. Dis. 18, 568–581 (2005).
Windrem, M.S. et al. Progenitor cells derived from the adult human subcortical white matter disperse and differentiate as oligodendrocytes within demyelinated lesions of the rat brain. J. Neurosci. Res. 69, 966–975 (2002).
Windrem, M.S., Roy, N., Nunes, M. & Goldman, S.A. Identification, selection, and use of human oligodendrocyte progenitor cells. in Neural Stem Cells for Brain Repair (eds. Zigova, T., Snyder, E.) 69–88 (Humana, NY, 2003).
Windrem, M.S. et al. Fetal and adult human oligodendrocyte progenitor cell isolates myelinate the congenitally dysmyelinated brain. Nat. Med. 10, 93–97 (2004).
Yandava, B.D., Billinghurst, L.L. & Snyder, E.Y. “Global” cell replacement is feasible via neural stem cell transplantation: evidence from the dysmyelinated shiverer mouse brain. Proc. Natl. Acad. Sci. USA 96, 7029–7034 (1999).
Snyder, E.Y., Taylor, R.M. & Wolfe, J.H. Neural progenitor cell engraftment corrects lysosomal storage throughout the MPS VII mouse brain. Nature 374, 367–370 (1995).
Urayama, A., Grubb, J., Sly, W. & Banks, W. Developmentally regulated mannose 6-phophate receptor-mediated transport of a lysosomal enzyme across the blood-brain barrier. Proc. Natl. Acad. Sci. USA 101, 12658–12663 (2004).
Pluchino, S. et al. Injection of adult neurospheres induces recovery in a chronic model of multiple sclerosis. Nature 422, 688–694 (2003).
Pluchino, S., Furlan, R. & Martino, G. Cell-based remyelinating therapies in multiple sclerosis: evidence from experimental studies. Curr. Opin. Neurol. 17, 247–255 (2004).
Franklin, R.J. Why does remyelination fail in multiple sclerosis? Nat. Rev. Neurosci. 3, 705–714 (2002).
Jenner, P. & Olanow, C.W. Understanding cell death in Parkinson's disease. Ann. Neurol. 44, S72–S84 (1998).
Olanow, C., Kordower, J. & Freeman, T. Fetal nigral transplantation as a therapy for Parkinson's disease. Trends Neurosci. 19, 102–109 (1996).
Lindvall, O. Cerebral implantation in movement disorders: state of the art. Mov. Disord. 14, 201–205 (1999).
Bjorklund, A. & Lindvall, O. Cell replacement therapies for central nervous system disorders. Nat. Neurosci. 3, 537–544 (2000).
Hagell, P. et al. Dyskinesias following neural transplantation in Parkinson's disease. Nat. Neurosci. 5, 627–628 (2002).
Studer, L., Tabar, V. & McKay, R. Transplantation of expanded mesencephalic precursors leads to recovery in Parkinsonian rats. Nat. Neurosci. 1, 290–295 (1998).
Sawamoto, K. et al. Generation of dopaminergic neurons in the adult brain from mesencephalic precursor cells labeled with a nestin-GFP transgene. J. Neurosci. 21, 3895–3903 (2001).
Ye, W., Shimamura, K., Rubenstein, J., Hynes, M. & Rosenthal, A. FGF and Shh signals control dopaminergic and serotinergic cell fate in the anterior neural plate. Cell 93, 755–766 (1998).
Lee, S-H., Lumelsky, N., Studer, L., Auerbach, J. & McKay, R. Efficient generation of midbrain and hindbrain neurons from mouse embryonic stem cells. Nat. Biotechnol. 18, 675–679 (2000).
Studer, L. et al. Enhanced proliferation, survival and dopaminergic differentiation of CNS precursors in lowered oxygen. J. Neurosci. 20, 7377–7383 (2000).
Kim, J. et al. Dopamine neurons derived from embryonic stem cells function in an animal model of Parkinson's disease. Nature 418, 50–56 (2002).
Takagi, Y. et al. Dopaminergic neurons generated from monkey embryonic stem cells function in a Parkinson primate model. J. Clin. Invest. 115, 102–108 (2005).
Goridis, C. & Rohrer, H. Specification of catecholaminergic and serotonergic neurons. Nat. Neurosci. 3, 531–541 (2002).
Bjorklund, L. et al. Embryonic stem cells develop into functional dopaminergic neurons after transplantation in a Parkinson rat model. Proc. Natl. Acad. Sci. USA 99, 2344–2349 (2002).
Nistor, G., Totoiu, M., Haque, N.S., Carpenter, M. & Kierstead, H. Human embryonic stem cells differentiate into oligodendrocytes in high purity and myelinate after spinal cord contusion. Glia 49, 385–396 (2005).
Roy, N. et al. Enhancer-specified GFP-based FACS purification of human spinal motor neurons from embryonic stem cells. Exp. Neurol. (in the press) (2005).
Li, M., Pevny, L., Lovell-Badge, R. & Smith, A. Generation of purified neural precursors from embryonic stem cells by lineage selection. Curr. Biol. 8, 971–974 (1998).
Hu, F. & Strittmatter, S.M. Regulating axon growth within the postnatal central nervous system. Semin. Perinatol. 28, 371–378 (2004).
Gao, Y. et al. Activated CREB is sufficient to overcome inhibitors in myelin and promote spinal axon regeneration in vivo. Neuron 44, 609–621 (2004).
Nikulina, E., Tidwell, J.L., Dai, H.N., Bregman, B.S. & Filbin, M.T. The phosphodiesterase inhibitor rolipram delivered after a spinal cord lesion promotes axonal regeneration and functional recovery. Proc. Natl. Acad. Sci. USA 101, 8786–8790 (2004).
Hofstetter, C. et al. Allodynia limits the usefulness of intraspinal neural stem cell grafts; directed differentaition improves outcome. Nat. Neurosci. 8, 346–353 (2005).
Ogawa, Y. et al. Transplantation of in vitro-expanded fetal neural progenitor cells results in neurogenesis and functional recovery after spinal cord contusion injury in adult rats. J. Neurosci. Res. 69, 925–933 (2002).
Ourednik, J., Ourednik, V., Lynch, W.P., Schachner, M. & Snyder, E. Neural stem cells display an inherent mechanism for rescuing dysfunctional neuons. Nat. Biotechnol. 20, 1103–1110 (2002).
Han, S.W., Liu, Y., Tyler-Polsz, C., Rao, M.S. & Fischer, I. Transplantation of glial-restricted precursor cells into the adult spinal cord. Glia 45, 1–16 (2004).
Brustle, O. et al. Embryonic stem cell-derived glial precursors: a source of myelinating transplants. Science 285, 754–756 (1999).
Wang, X. et al. P2X7 receptor inhibition improves recovery after spinal cord injury. Nat. Med. 10, 821–827 (2004).
Pearse, D. et al. cAMP and Schwann cells promote axonal growth and functional recovery after spinal cord injury. Nat. Med. 10, 610–616 (2004).
Kerr, D.A. et al. Human embryonic germ cell derivatives facilitate motor recovery of rats with diffuse motor neuron injury. J. Neurosci. 23, 5131–5140 (2003).
Li, X. et al. Specification of motoneurons from human embryonic stem cells. Nat. Biotechnol. 23, 215–221 (2005).
Wichterle, H., Lieberam, I., Porter, J.A. & Jessell, T.M. Directed differentiation of embryonic stem cells into motor neurons. Cell 110, 385–397 (2002).
Roy, N. et al. Telomerase-immortalization of neuronally restricted progenitor cells derived from the human fetal spinal cord. Nat. Biotechnol. 22, 297–305 (2004).
Arber, S. et al. Requirement for the homeobox gene Hb9 in the consolidation of motor neuron identity. Neuron 23, 659–674 (1999).
Nakano, T., Windrem, M., Zappavigna, V. & Goldman, S.A. Identification of a conserved 125 base-pair Hb9 enhancer that specifies gene expression to spinal motor neurons. Dev. Biol. (in the press) (2005).
Arvidsson, A., Collin, T., Kirik, D., Kokaia, Z. & Lindvall, O. Neuronal replacement from endogenous precursors in the adult brain after stroke. Nat. Med. 8, 963–970 (2002).
Parent, J., Vexler, Z., Gong, C., Derugin, N. & Ferriero, D. Rat forebrain neurogenesis and striatal neuron replacement after focal stroke. Ann. Neurol. 52, 802–813 (2002).
Jin, K. et al. Directed migration of neuronal precursors into the ischemic cerebral cortex and striatum. Mol. Cell. Neurosci. 24, 171–189 (2003).
Nakatomi, H. et al. Regeneration of hippocampal pyramidal neurons after ischemic brain injury by recruitment of endogenous progenitors. Cell 110, 429–441 (2002).
Curtis, M.A. et al. Increased cell proliferation and neurogenesis in the adult human Huntington's disease brain. Proc. Natl. Acad. Sci. USA 100, 9023–9027 (2003).
Jin, K. et al. Increased hippocampal neurogenesis in Alzheimer's disease. Proc. Natl. Acad. Sci. USA 101, 343–347 (2004).
Magavi, S., Leavitt, B. & Macklis, J. Induction of neurogenesis in the neocortex of adult mice. Nature 405, 951–955 (2000).
Chen, J., Magavi, S. & Macklis, J. Neurogenesis of corticospinal motor neurons extending spinal projections in adult mice. Proc. Natl. Acad. Sci. USA 101, 16357–16362 (2004).
Kuhn, H.G., Winkler, J., Kempermann, G., Thal, L.J. & Gage, F.H. Epidermal growth factor and fibroblast growth factor-2 have different effects on neural progenitors in the adult rat brain. J. Neurosci. 17, 5820–5829 (1997).
Fallon, J. et al. In vivo induction of massive proliferation, directed migration, and differentiation of neural cells in the adult mammalian brain. Proc. Natl. Acad. Sci. USA 97, 14686–14691 (2000).
Jin, K. et al. Vascular endothelial growth factor (VEGF) stimulates neurogenesis in vitro and in vivo. Proc. Natl. Acad. Sci. USA 99, 11946–11950 (2002).
Cao, L. et al. VEGF links hippocampal activity with neurogenesis, learning and memory. Nat. Genet. 36, 827–835 (2004).
Jin, K., Mao, X.O., Sun, Y., Xie, L. & Greenberg, D.A. Stem cell factor stimulates neurogenesis in vitro and in vivo. J. Clin. Invest. 110, 311–319 (2002).
Packer, M. et al. Nitric oxide negatively regulates mammalian adult neurogenesis. Proc. Natl Acad. Sci. USA 100, 9566–9571 (2003).
Cameron, H.A., Hazel, T.G. & McKay, R.D. Regulation of neurogenesis by growth factors and neurotransmitters. J. Neurobiol. 36, 287–306 (1998).
Aberg, M., Aberg, D., Hedbacker, H., Oscarsson, J. & Eriksson, P. Peripheral infusion of IGF-1 selectively induces neurogenesis in the adult rat hippocampus. J. Neurosci. 20, 2896–2903 (2000).
Malberg, J., Eisch, A., Nestler, E. & Duman, R. Chronic antidepressant treatment increases neurogenesis in adult rat hippocampus. J. Neurosci. 20, 9104–9110 (2000).
Kirschenbaum, B. & Goldman, S.A. Brain-derived neurotrophic factor promotes the survival of neurons arising from the adult rat forebrain subependymal zone. Proc. Natl. Acad. Sci. USA 92, 210–214 (1995).
Goldman, S., Kirschenbaum, B., Harrison-Restelli, C. & Thaler, H. Neuronal precursor cells of the adult rat ventricular zone persist into senescence, with no change in spatial extent or BDNF response. J. Neurobiol. 32, 554–566 (1997).
Louissaint, A., Rao, S., Leventhal, G. & Goldman, S.A. Coordinated ineraction of neurogenesis and angiogenesis in the adult songbird brain. Neuron 34, 945–960 (2002).
Ahmed, S., Reynolds, B.A. & Weiss, S. BDNF enhances the differentiation but not the survival of CNS stem cell-derived neuronal precursors. J. Neurosci. 15, 5765–5778 (1995).
Benraiss, A., Chmielnicki, E., Lerner, K., Roh, D. & Goldman, S.A. Adenoviral brain-derived neurotrophic factor induces both neostriatal and olfactory neuronal recruitment from endogenous progenitor cells in the adult forebrain. J. Neurosci. 21, 6718–6731 (2001).
Pencea, V., Bingaman, K.D., Wiegand, S.J. & Luskin, M.B. Infusion of brain-derived neurotrophic factor into the lateral ventricle of the adult rat leads to new neurons in the parenchyma of the striatum, septum, thalamus, and hypothalamus. J. Neurosci. 21, 6706–6717 (2001).
Ivkovic, S. & Ehrlich, M. Expression of the striatal DARPP-32/ARPP-21 phenotype in GABAergic neurons requires neurotrophins in vivo and in vitro. J. Neurosci. 19, 5409–5419 (1999).
Gross, R.E. et al. Bone morphogenetic proteins promote astroglial lineage commitment by mammalian subventricular zone progenitor cells. Neuron 17, 595–606 (1996).
Gomes, W.A., Mehler, M.F. & Kessler, J.A. Transgenic overexpression of BMP4 increases astroglial and decreases oligodendroglial lineage commitment. Dev. Biol. 255, 164–177 (2003).
Mabie, P.C. et al. Bone morphogenetic proteins induce astroglial differentiation of oligodendroglial-astroglial progenitor cells. J. Neurosci. 17, 4112–4120 (1997).
Zimmerman, L.B., De Jesus-Escobar, J.M. & Harland, R.M. The Spemann organizer signal noggin binds and inactivates bone morphogenetic protein 4. Cell 86, 599–606 (1996).
Lim, D. et al. Noggin antagonizes BMP signaling to create a niche for adult neurogenesis. Neuron 28, 713–726 (2000).
Chmielnicki, E. & Goldman, S.A. Induced neurogenesis by endogenous progenitor cells in the adult mammalian brain. Prog. Brain Res. 138, 451–464 (2002).
Chmielnicki, E., Benraiss, A., Economides, A.N. & Goldman, S.A. Adenovirally expressed noggin and brain-derived neurotrophic factor cooperate to induce new medium spiny neurons from resident progenitor cells in the adult striatal ventricular zone. J. Neurosci. 24, 2133–2142 (2004).
Mangiarini, L. et al. Exon 1 of the HD gene with an expanded CAG repeat is sufficient to cause a progressive neurological phenotype in transgenic mice. Cell 87, 493–506 (1996).
Cho, S.R., Chmielnicki, E. & Goldman, S.A. Adenoviral co-delivery of BDNF and noggin induces striatal neuronal replacement and delays motor impairment in a transgenic model of Huntington's Disease. Mol. Ther. 9, S86–S87 (2004).
Singh, S. et al. Identification of a cancer stem cell in human brain tumors. Cancer Res. 63, 5821–5828 (2003).
Singh, S. et al. Identification of human brain tumor initiating cells. Nature 432, 396–401 (2004).
Craig, C.G. et al. In vivo growth factor expansion of endogenous subependymal neural precursor cell populations in the adult mouse brain. J. Neurosci. 16, 2649–2658 (1996).
Notlebohm, F. The neural basis of birdsong. PLoS Biology 3, e164 (2005).
Svendsen, C.N., Bhattacharyya, A. & Tai, Y.T. Neurons from stem cells: preventing an identity crisis. Nat. Rev. Neurosci. 2, 831–834 (2001).
Acknowledgements
I thank M. Nedergaard, N. Roy, M. Windrem, F. Sim, A. Benraiss, S. Wang, E. Chmielnicki and S.-R. Cho for their contributions to the studies discussed, and apologize to the many authors whose relevant work I have not been able to cite in this short review. My thanks to F. Sim for expert help in illustration. Supported by the US National Institutes of Health–National Institute of Neurological Disorders and Stroke, the National Multiple Sclerosis Society, the New York Spinal Cord Injury Research Program, The A–T Children's Project and the CNS foundation, Berlex Bioscience and Merck Research Laboratories.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
S.G. receives laboratory support from Merck Research Laboratories and Berlex Bioscience, and is a consultant to both Merck and Q Therapeutics.
Rights and permissions
About this article
Cite this article
Goldman, S. Stem and progenitor cell–based therapy of the human central nervous system. Nat Biotechnol 23, 862–871 (2005). https://doi.org/10.1038/nbt1119
Published:
Issue Date:
DOI: https://doi.org/10.1038/nbt1119
This article is cited by
-
Rapid differentiation of hiPSCs into functional oligodendrocytes using an OLIG2 synthetic modified messenger RNA
Communications Biology (2022)
-
Molecularly cleavable bioinks facilitate high-performance digital light processing-based bioprinting of functional volumetric soft tissues
Nature Communications (2022)
-
Promising Role of Oral Cavity Mesenchymal Stem Cell-Derived Extracellular Vesicles in Neurodegenerative Diseases
Molecular Neurobiology (2022)
-
Intracerebroventricular Administration of hNSCs Improves Neurological Recovery after Cardiac Arrest in Rats
Stem Cell Reviews and Reports (2021)
-
Placenta-derived multipotent mesenchymal stromal cells: a promising potential cell-based therapy for canine inflammatory brain disease
Stem Cell Research & Therapy (2020)