Abstract
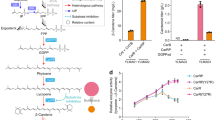
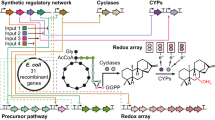
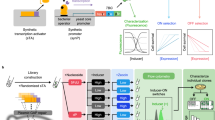
The burgeoning demand for complex, biologically active molecules for medicine, materials science, consumer products, and agrochemicals is driving efforts to engineer new biosynthetic pathways into microorganisms and plants. We have applied principles of breeding, including mixing genes and modifying catalytic functions by in vitro evolution, to create new metabolic pathways for biosynthesis of natural products in Escherichia coli. We expressed shuffled phytoene desaturases in the context of a carotenoid biosynthetic pathway assembled from different bacterial species and screened the resulting library for novel carotenoids. One desaturase chimera efficiently introduced six rather than four double bonds into phytoene, to favor production of the fully conjugated carotenoid, 3,4,3′,4′-tetradehydrolycopene. This new pathway was extended with a second library of shuffled lycopene cyclases to produce a variety of colored products. One of the new pathways generates the cyclic carotenoid torulene, for the first time, in E. coli. This combined approach of rational pathway assembly and molecular breeding may allow the discovery and production, in simple laboratory organisms, of new compounds that are essentially inaccessible from natural sources or by synthetic chemistry.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Altreuter, D.H. & Clark, D.S. Combinatorial biocatalysis: taking the lead from Nature. Curr. Opin. Biotechnol. 10, 130–136 (1999).
Reynolds, K.A. Combinatorial biosynthesis: lesson learned from nature. Proc. Natl. Acad. Sci. USA 95, 12744–12746 (1998).
Cane, D.E., Walsh, C.T. & Khosla, C. Biochemistry—harnessing the biosynthetic code: combinations, permutations, and mutations. Science 282, 63–68 (1998).
Lau, J., Fu, H., Cane, D.E. & Khosla, C. Dissecting the role of acyltransferase domains of modular polyketide synthases in the choice and stereochemical fate of extender units. Biochemistry 38, 1643–1651 (1999).
Stemmer, W.P.C. Rapid evolution of a protein in-vitro by DNA-shuffling. Nature 370, 389–391 (1994).
Crameri, A., Raillard, S.A., Bermudez, E. & Stemmer, W.P.C. DNA shuffling of a family of genes from diverse species accelerates directed evolution. Nature 391, 288–291 (1998).
Joo, H., Lin, Z.L. & Arnold, F.H. Laboratory evolution of peroxide-mediated cytochrome P450 hydroxylation. Nature 399, 670–673 (1999).
Arnold, F.H. & Volkov, A.A. Directed evolution of biocatalysts. Curr. Opin. Chem. Biol. 3, 54–59 (1999).
Bertram, J.S. Carotenoids and gene regulation. Nutr. Rev. 57, 182–191 (1999).
Singh, D.K. & Lippman, S.M. Cancer chemoprevention—Part 1: Retinoids and carotenoids and other classic antioxidants. Oncology NY 12, 1643–1653 (1998).
Rock, C.L. Carotenoids: biology and treatment. Pharmacol. Ther. 75, 185–197 (1997).
Edge, R., McGarvey, D.J. & Truscott, T.G. The carotenoids as antioxidants—a review. J. Photochem. Photobiol. 41, 189–200 (1997).
Johnson, E.A. & Schroeder, W.A. Microbial carotenoids. Adv. Biochem. Eng. Biotechnol. 53, 119–178 (1995).
Misawa, N. & Shimada, H. Metabolic engineering for the production of carotenoids in noncarotenogenic bacteria and yeasts. J. Biotechnol. 59, 169–181 (1998).
Hirschberg, J. In Carotenoids: biosynthesis and metabolism, Vol. 3 (eds Britton, G., Liaaen-Jensen, S. & Pfander, H.) 148–194 (Birkhäuser Verlag, Basel; 1998).
Sandmann, G., Albrecht, M., Schnurr, G., Knorzer, O. & Boger, P. The biotechnological potential and design of novel carotenoids by gene combination in Escherichia coli. Trends Biotechnol. 17, 233–237 (1999).
Britton, G. In Carotenoids: biosynthesis and metabolism, Vol. 3 (eds Britton, G., Liaaen-Jensen, S. & Pfander, H.) 13–147 (Birkhäuser Verlag, Basel; 1998).
Armstrong, G.A. & Hearst, J.E. Carotenoids. 2. Genetics and molecular biology of carotenoid pigment biosynthesis. FASEB J. 10, 228–237 (1996).
Sandmann, G. Carotenoid biosynthesis in microorganisms and plants. Eur. J. Biochem. 223, 7–24 (1994).
Garcia-Asua, G., Lang, H.P., Cogdell, R.J. & Hunter, C.N. Carotenoid diversity: a modular role for the phytoene desaturase step. Trends Plant Sci. 3, 445–449 (1998).
Bartley, G.E., Schmidhauser, T.J., Yanofsky, C. & Scolnik, P.A. Carotenoid desaturases from Rhodobacter capsulatus and Neurospora crassa are structurally and functionally conserved and contain domains homologous to flavoprotein disulfide oxidoreductase. J. Biol. Chem. 265, 16020–16024 (1990).
Fraser, P.D. et al. Expression in Escherichia coli, purification, and reactivation of the recombinant Erwinia uredovora phytoene desaturase. J. Biol. Chem. 267, 19891–19895 (1992).
Linden, H. et al. Functional complementation in Escherichia coli of different phytoene desaturase genes and analysis of accumulated carotenes. Z. Naturforsch, 46c, 1045–1051 (1991).
Armstrong, G.A., Alberti, M., Leach, F. & Hearst, J.E. Nucleotide sequence, organization, and nature of the protein products of the carotenoid biosynthesis gene cluster of Rhodobacter capsulatus. Mol. Gen. Genetics 216, 254–268 (1989).
Wierenga, R.K., Terpstra, P. & Hol, W.G.J. Prediction of the occurrence of the ADP-binding beta-alpha-beta fold in proteins, using an amino-acid fingerprint. J. Mol. Biol. 187, 101–107 (1986).
Ausich, R.L. Production of carotenoids by recombinant DNA-technology. Pure Appl. Chem. 66, 1057–1062 (1994).
Hunter, C.N. et al. Introduction of new carotenoids into the bacterial photosynthetic apparatus by combining the carotenoid biosynthetic pahtways of Erwinia herbicola and Rhodobacter sphaeroides. J. Bacteriol. 176, 3692–3697 (1994).
Takaichi, S. et al. The carotenoid 7,8-dihydro-psi end group can be cyclized by the lycopene cyclases from the bacterium Erwinia uredovora and the higher plant Capsicum annuum. Eur. J. Biochem. 241, 291–296 (1996).
Albrecht, M. et al. Synthesis of atypical cyclic and acyclic hydroxy carotenoids in Escherichia coli transformants. J. Biotechnol. 58, 177–185 (1997).
Cunningham, F.X. et al. Functional analysis of the beta and epsilon lycopene cyclase enzymes of Arabidopsis reveals a mechanism for control of cyclic carotenoid formation. Plant Cell 8, 1613–1626 (1996).
Hugueney, P. et al. Metabolism of cyclic carotenoids—a model for the alteration of this biosynthetic pathway in Capsicum annuum chromoplasts. Plant J. 8, 417–424 (1995).
An, G.-H., Cho, M.-H. & Johnson, E.A. Monocyclic carotenoid biosynthetic pathway in the yeast Phaffia rhodozyma (Xanthophyllomyces dendrorhous). J. Biosci. Bioeng. 88, 189–193 (1999).
Acknowledgements
We are grateful to Dr. Peter G. Green (Environmental Analysis Center, Caltech) for technical support and helpful discussions on mass analysis. C.S.-D. was supported by the Deutsche Forschungsgemeinschaft (DFG) and D.U. by the Japan Society for the Promotion of Science (JSPS).
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Schmidt-Dannert, C., Umeno, D. & Arnold, F. Molecular breeding of carotenoid biosynthetic pathways. Nat Biotechnol 18, 750–753 (2000). https://doi.org/10.1038/77319
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/77319
This article is cited by
-
Genome-wide identification of CCO gene family in cucumber (Cucumis sativus) and its comparative analysis with A. thaliana
BMC Plant Biology (2023)
-
Elucidation of the whole carotenoid biosynthetic pathway of aphids at the gene level and arthropodal food chain involving aphids and the red dragonfly
BMC Zoology (2021)
-
Evolution-aided engineering of plant specialized metabolism
aBIOTECH (2021)
-
Yeast carotenoids: production and activity as antimicrobial biomolecule
Archives of Microbiology (2021)
-
Use of response surface methodology to enhance carotenoid pigment production from Cellulosimicrobium strain AZ
SN Applied Sciences (2020)