Abstract
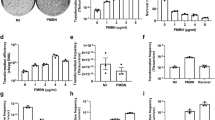
We report high–frequency transformation of penicillin–producing strains of Penicillium chrysogenum using plasmid vectors carrying the pyr4 gene of Neurospora crassa. Transformation to stable uracil independence is associated with acquisition of transforming DNA in high molecular forms that are not simple multimers of the vector. A transformant strain showed a fivefold higher orotidine–5′–monophosphate decarboxylase activity (coded by the pyr4 gene) than the wild type strain, indicating that the genes carried in the transforming vector are efficiently expressed in Penicillium.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Demain, A.L. 1983. Biosynthesis of β-lactam antibiotics, p. 189–228 In: Antibiotics containing the β-lactam structure I. A. L. Demain and N. A. Solomon (eds.). Springer-Verlag International, New York.
Martín, J.F. and Liras, P. 1985. Biosynthesis of β-lactam antibiotics: Design and construction of overproducing strains. Trends Biotechnol. 3: 39–44.
Queener, S.W., Ingolia, T.D., Skatrud, P.L., Chapman, J.L., and Kaster, K.R. 1985. A system for genetic transformation of Cephalosporium acremonium, p.268–472 In: Microbiology. D. Schlessinger (ed.). American Society for Microbiology, Washington D.C.
Skatrud, P.L., Samson, S., Carr, L., Ingolia, T.D., and Queener, S.W. 1986. Molecular Biology of Cephalosporium acremonium and Penicillium chrysogenum. In: Proceedings of the Genetics of Industrial Microorganisms. M. Alacevick (ed.). Yugoslavia. In press.
Samson, S.M., Belagaje, R., and Blankenship, D.T., et al. Isolation, sequence determination and expression in Escherichia coli of the isopenicillin N synthetase gene from Cephalosporium acremonium. Nature 318: 191–194.
Tilburn, J., Schazzocchio, C., Taylor, G.G., Zabicky-Zissman, J.H., Lockington, R.A., and Davies, R.W. 1984. Transformation by integration in Aspergillus nidulans. Gene 26: 205–221.
Ballance, D.J., and Turner, G. 1985. Gene cloning in Aspergillus nidulans: Isolation of the isocitrate lyase gene (acuD). Mol. Gen. Genet. 202: 271–275.
Yelton, M.M., Hamer, J.E., and Timberlake, W.E. 1984. Transformation of Aspergillus nidulans by using a trp plasmid. Proc. Natl. Acad. Sci. USA. 81: 1470–1474.
Buxton, F.P., and Radford, A. 1983. Cloning of the structural gene for orotidine-5′-phosphate carboxylase of Neurospora crassa by expression in Escherichia coli. Mol. Gen. Genet. 190: 403–405.
Ballance, D.J., and Turner, G. 1985. Development of a high-frequency transforming vector for Aspergillus nidulans. Gene 36: 321–331.
Díez, B., Alvarez, E., Cantoral, J.M., Barredo, J.L., and Martín, J.F. 1987. Isolation and characterization of pyrG mutants of Penicillium chrysogenum by resistance to 5′-fluoroorotic acid. Current Genet. Submitted.
Rose, M., Grisafi, P., and Botstein, D. 1984. Structure and function of the yeast URA3 gene: Expression in E. coli. Gene 29: 113–124.
Botstein, D., and Davis, R.W. 1982. Principles and practice of recombinant DNA research with yeast, p. 607–635. In: Molecular Biology of the yeast Saccharomyces: Metabolism and Gene Expression. J. N. Strathern, E. W. Jones and J. R. Broach (eds.). Cold Spring Harbor Laboratory, Cold Spring Harbor, New York.
Maniatis, T., Fritsch, E.F., Sambrook, J. 1982. Molecular cloning. A laboratory manual. Cold Spring Harbor Laboratory, Cold Spring Harbor, New York.
Johnstone, I.L. 1985. Transformation of Aspergillus nidulans. Microbiol. Sci. 2: 307–311.
Ballance, D.J., Buxton, F.P., and Turner, G. 1983. Transformation of Aspergillus nidulans by the orotidine-5′-phosphate decarboxylase gene of Neurospora crassa. Biochem. Biophys. Res. Commun. 112: 284–289.
John, M.A., and Peberdy, J.F. 1984. Transformation of Aspergillus nidulans using the argB gene. Enzyme Microbiol. Technol. 6: 386–389.
Wernars, K., Goosen, T., Wennekes, L.M.J., Visser, J., Bos, C.J., Van Der Broek, H.W.J., Van Gorcom, R.F.M., Van Den Hondel, C.A.M.J.J., and Pouwels, P.H. 1985. Gene amplification in Aspergillus nidulans by transformation with vectors containing the amdS gene. Curr. Genet. 9: 361–368.
Hinnen, A., Hicks, J.B., and Fink, G.R. 1978. Transformation of yeast. Proc. Natl. Acad. Sci. USA 75: 1929–1933.
Stinchcomb, D.T., Thomas, M., Kelly, J., Selber, E.R., and Davis, R.W. 1980. Eucaryotic DNA segments capable of autonomous replication in yeast. Proc. Natl. Acad. Sci. USA. 77: 4559–4563.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Cantoral, J., Díez, B., Barredo, J. et al. High–Frequency Transformation of Penicillium Chrysogenum. Nat Biotechnol 5, 494–497 (1987). https://doi.org/10.1038/nbt0587-494
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nbt0587-494
This article is cited by
-
Role of AtYap1 in the reactive oxygen species regulation of lovastatin production in Aspergillus terreus
Applied Microbiology and Biotechnology (2023)
-
bZIP transcription factors PcYap1 and PcRsmA link oxidative stress response to secondary metabolism and development in Penicillium chrysogenum
Microbial Cell Factories (2022)
-
New Antimicrobial Potential and Structural Properties of PAFB: A Cationic, Cysteine-Rich Protein from Penicillium chrysogenum Q176
Scientific Reports (2018)
-
A Penicillium chrysogenum-based expression system for the production of small, cysteine-rich antifungal proteins for structural and functional analyses
Microbial Cell Factories (2016)
-
Direct involvement of the CreA transcription factor in penicillin biosynthesis and expression of the pcbAB gene in Penicillium chrysogenum
Applied Microbiology and Biotechnology (2014)