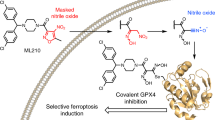
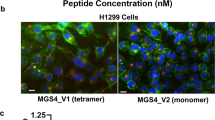
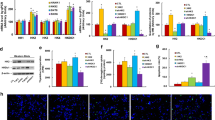
Abstract
Several nonmammalian members of the RNase A superfamily exhibit anticancer activity that appears to correlate with resistance to the cytosolic ribonuclease inhibitor (RI). We mutated two human ribonucle- ases—pancreatic RNase (hRNAse) and eosinophil-derived neurotoxin (EDN)—to incorporate cysteine residues at putative sites of close contact to RI, but distant from the catalytic sites. Coupling of Cys89 of RNase and Cys87 of EDN to proteins at these sites via a thioether bond produced enzymatically active conjugates that were resistant to RI. To elicit cellular targeting as well as to block RI binding, transferrin was conjugated to a mutant human RNase, rhRNase(Gly89→Cys) and a mutant EDN (Thr87→Cys). The transferrin–rhRNase(Gly89→Cys) thioether conjugate was 5000-fold more toxic to U251 cells than recombinant wild-type hRNase. In addition, transferrin-targeted EDN exhibited tumor cell toxicities similar to those of hRNase. Thus, we endowed two human RI-sensitive RNases with greater cytotoxicity by increasing their resistance to RI. This strategy has the potential to generate a novel set of recombinant human proteins useful for targeted therapy of cancer.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
D'Alessio, G. & Riordan, J.F. Ribonucleases: Structures and Functions (Academic Press, New York, 1997).
Domachowske, J.B. & Rosenberg, H.F. Eosinophils inhibit retroviral transduction of human target cells by a ribonuclease-dependent mechanism. J. Leukoc. Biol. 62, 363– 368 (1997).
Lehrer, R.I. et al. Antibacterial properties of eosinophil major basic protein and eosinophil cationic protein. J. Immunol. 142, 4428– 4434 (1989).
Rosenberg, H.F. Recombinant human eosinophil cationic protein. Ribonuclease activity is not essential for cytotoxicity. J. Biol. Chem. 270, 7876–7881 (1995).
Durack, D.T., Ackerman, S.J., Loegering, D.A. & Gleich, G.J. Purification of human eosinophil-derived neurotoxin. Proc. Natl. Acad. Sci. USA 78, 5165–5169 (1981).
Newton, D.L. et al. Toxicity of an antitumor ribonuclease to Purkinje neurons. J. Neurosci. 14, 538–544 (1994).
Nitta, K. et al. Inhibition of cell proliferation by Rana catesbeiana and Rana japonica lectins belonging to the ribonuclease superfamily. Cancer Res. 54, 920–927. ( 1994).
Mikulski, S.M., Grossman, A.M., Carter, P.W., Shogen, K. & Costanzi, J.J. Phase I human clinical trial of ONCONASE (P-30 protein) administered intravenously on a weekly schedule in cancer patient with solid tumors. Int. J. Oncol. 3, 57–64 (1993).
Vescia, S., Tramontano, D., Augusti-Tocco, G. & D'Alessio, G. In vitro studies on selective inhibition of tumor cell growth by seminal ribonuclease. Cancer Res. 40, 3740–3744 (1980).
Laccetti, P. et al. In vivo and in vitro growth-inhibitory effect of bovine seminal ribonuclease on a system of rat thyroid epithelial transformed cells and tumors. Cancer Res. 52, 4582–4586 (1992).
Wu, Y., Mikulski, S.M., Ardelt, W., Rybak, S.M. & Youle, R.J. A cytotoxic ribonuclease. Study of the mechanism of onconase cytotoxicity. J. Biol. Chem. 268, 10686–10693 (1993).
Murthy, B.S. & Sirdeshmukh, R. Sensitivity of monomeric and dimeric forms of bovine seminal ribonuclease to human placental ribonuclease inhibitor. Biochem J. 281, 343– 348 (1992).
Saxena, S.K. et al. Comparison of RNases and toxins upon injection into Xenopus oocytes. J. Biol. Chem. 266, 21208– 21214 (1991).
Rybak, S.M., Saxena, S.K., Ackerman, E.J. & Youle, R.J. Cytotoxic potential of ribonuclease and ribonuclease hybrid proteins. J. Biol. Chem. 266, 21202–21207 (1991).
Newton, D.L. et al. 1992.Cytotoxic ribonuclease chimeras. Targeted tumoricidal activity in vitro and in vivo. J. Biol. Chem. 267, 19572–19578.
Newton, D.L., Nicholls, P.J., Rybak, S.M. & Youle, R.J. Expression and characterization of recombinant human eosinophil-derived neurotoxin and eosinophil-derived neurotoxin-anti-transferrin receptor sFv. J. Biol. Chem. 269, 26739–26745 (1994).
Lee, F.S., Shapiro, R. & Vallee, B.L. Tight-binding inhibition of angiogenin and ribonuclease A by placental ribonuclease inhibitor. Biochemistry 28, 225–230 (1989).
Kobe, B. & Deisenhofer, J. Mechanism of ribonuclease inhibition by ribonuclease inhibitor protein based on the crystal structure of its complex with ribonuclease A. J. Mol. Biol. 264, 1028–1043 (1996).
Shapiro, R. & Vallee, B.L. Interaction of human placental ribonuclease with placental ribonuclease inhibitor. Biochemistry 30, 2246–2255 ( 1991).
Papageorgiou, A.C., Shapiro, R. & Acharya, K.R. Molecular recognition of human angiogenin by placental ribonuclease inhibitor—an x-ray crystallographic study at 2.0 Å resolution. Embo J. 16, 5162– 5177 (1997).
Piccoli, R. et al. The dual-mode quaternary structure of seminal RNase. Proc. Natl. Acad. Sci. USA 89, 1870– 1874 (1992).
Kim, J.S., Soucek, J., Matousek, J. & Raines, R.T. Mechanism of ribonuclease cytotoxicity. J. Biol. Chem. 270, 31097–31102 (1995).
Laske, D.W., Youle, R.J. & Oldfield, E.H. 1997.Tumor regression with regional distribution of the targeted toxin TF- CRM107 in patients with malignant brain tumors. Nat. Med. 3, 1362– 1368.
Recht, L., Torres, C.O., Smith, T.W., Raso, V. & Griffin, T.W. Transferrin receptor in normal and neoplastic brain tissue: implications for brain-tumor immunotherapy. J. Neurosurg. 72, 941–945 (1990).
Jain, R.K. Barriers to drug delivery in solid tumors. Sci. Am. 271, 58–65 (1994).
Boix, E. et al. Role of the N terminus in RNase A homologues: differences in catalytic activity, ribonuclease inhibitor interaction and cytotoxicity. J. Mol. Biol. 257, 992–1007. ( 1996).
Ho, S.N., Hunt, H.D., Horton, R.M., Pullen, J.K. & Pease, L.R. Site-directed mutagenesis by overlap extension using the polymerase chain reaction. Gene 77, 51–59 (1989).
Yakovlev, G., Moiseyev, G.P., Sorrentino, S., De Prisco, R. & Libonati, M. Single-strand-preferring RNases degrade double-stranded RNAs by destabilizing its secondary structure. J. Biomol. Struct. Dyn. 15, 243–250 (1997).
Johnson, V.G., Wilson, D., Greenfield, L. & Youle, R.J. The role of the diphtheria toxin receptor in cytosol translocation. J. Neurosurg. 68, 1295–1300 (1988).
Wu, Y. et al. A study of the intracellular routing of cytotoxic ribonucleases. J. Biol. Chem. 270, 17476–17481 (1995).
Leland, P.A., Schultz, L.W., Kim, B.M., & Raines, R.T. Ribonuclease A variants with potent cytotoxic activity. Proc. Natl. Acad. Sci. USA 95, 10407–10412 ( 1998).
Acknowledgements
We thank Pat Johnson and Joan Barrick for technical assistance, and JoAnn Castelli for reading the manuscript. Certain commercial equipment, instruments, and materials are identified in this paper in order to specify the experimental procedure. Such identification does not imply recommendation or endorsement by the National Institute of Standards and Technology, nor does it imply that the materials or equipment are necessarily the best available for the purpose.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Suzuki, M., Saxena, S., Boix, E. et al. Engineering receptor-mediated cytotoxicity into human ribonucleases by steric blockade of inhibitor interaction. Nat Biotechnol 17, 265–270 (1999). https://doi.org/10.1038/7010
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/7010
This article is cited by
-
Improving the efficacy of antibody-based cancer therapies
Nature Reviews Cancer (2001)
-
Bovine seminal RNase induces apoptosis in normal proliferating lymphocytes
International Journal of Clinical & Laboratory Research (2000)
-
Smartbombs and cloaking devices
Nature Biotechnology (1999)