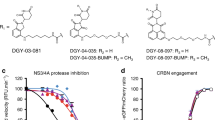
Abstract
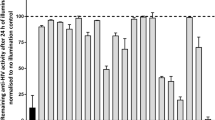
The HIV-1 protein Vif, essential for in vivo viral replication1,2,3,4, targets the human DNA-editing enzyme, APOBEC3G (A3G)5, which inhibits replication of retroviruses and hepatitis B virus6,7. As Vif has no known cellular homologs, it is an attractive, yet unrealized, target for antiviral intervention. Although zinc chelation inhibits Vif and enhances viral sensitivity to A3G8, this effect is unrelated to the interaction of Vif with A3G. We identify a small molecule, RN-18, that antagonizes Vif function and inhibits HIV-1 replication only in the presence of A3G. RN-18 increases cellular A3G levels in a Vif-dependent manner and increases A3G incorporation into virions without inhibiting general proteasome-mediated protein degradation. RN-18 enhances Vif degradation only in the presence of A3G, reduces viral infectivity by increasing A3G incorporation into virions and enhances cytidine deamination of the viral genome. These results demonstrate that the HIV-1 Vif-A3G axis is a valid target for developing small molecule–based new therapies for HIV infection or for enhancing innate immunity against viruses.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Gabuzda, D.H. et al. Role of vif in replication of human immunodeficiency virus type 1 in CD4+ T lymphocytes. J. Virol. 66, 6489–6495 (1992).
Madani, N. & Kabat, D. Cellular and viral specificities of human immunodeficiency virus type 1 vif protein. J. Virol. 74, 5982–5987 (2000).
Strebel, K. et al. The HIV 'A' (sor) gene product is essential for virus infectivity. Nature 328, 728–730 (1987).
von Schwedler, U., Song, J., Aiken, C. & Trono, D. Vif is crucial for human immunodeficiency virus type 1 proviral DNA synthesis in infected cells. J. Virol. 67, 4945–4955 (1993).
Sheehy, A.M., Gaddis, N.C., Choi, J.D. & Malim, M.H. Isolation of a human gene that inhibits HIV-1 infection and is suppressed by the viral Vif protein. Nature 418, 646–650 (2002).
Cullen, B.R. Role and mechanism of action of the APOBEC3 family of antiretroviral resistance factors. J. Virol. 80, 1067–1076 (2006).
Turelli, P. & Trono, D. Editing at the crossroad of innate and adaptive immunity. Science 307, 1061–1065 (2005).
Xiao, Z., Ehrlich, E., Luo, K., Xiong, Y. & Yu, X.F. Zinc chelation inhibits HIV Vif activity and liberates antiviral function of the cytidine deaminase APOBEC3G. FASEB J. 21, 217–222 (2007).
Mehle, A., Goncalves, J., Santa-Marta, M., McPike, M. & Gabuzda, D. Phosphorylation of a novel SOCS-box regulates assembly of the HIV-1 Vif-Cul5 complex that promotes APOBEC3G degradation. Genes Dev. 18, 2861–2866 (2004).
Jarmuz, A. et al. An anthropoid-specific locus of orphan C to U RNA-editing enzymes on chromosome 22. Genomics 79, 285–296 (2002).
Doehle, B.P., Schafer, A. & Cullen, B.R. Human APOBEC3B is a potent inhibitor of HIV-1 infectivity and is resistant to HIV-1 Vif. Virology 339, 281–288 (2005).
Langlois, M.A., Beale, R.C., Conticello, S.G. & Neuberger, M.S. Mutational comparison of the single-domained APOBEC3C and double-domained APOBEC3F/G anti-retroviral cytidine deaminases provides insight into their DNA target site specificities. Nucleic Acids Res. 33, 1913–1923 (2005).
Mariani, R. et al. Species-specific exclusion of APOBEC3G from HIV-1 virions by Vif. Cell 114, 21–31 (2003).
Marin, M., Rose, K.M., Kozak, S.L. & Kabat, D. HIV-1 Vif protein binds the editing enzyme APOBEC3G and induces its degradation. Nat. Med. 9, 1398–1403 (2003).
Mehle, A. et al. Vif overcomes the innate antiviral activity of APOBEC3G by promoting its degradation in the ubiquitin-proteasome pathway. J. Biol. Chem. 279, 7792–7798 (2004).
Sheehy, A.M., Gaddis, N.C. & Malim, M.H. The antiretroviral enzyme APOBEC3G is degraded by the proteasome in response to HIV-1 Vif. Nat. Med. 9, 1404–1407 (2003).
Stopak, K., de Noronha, C., Yonemoto, W. & Greene, W.C. HIV-1 Vif blocks the antiviral activity of APOBEC3G by impairing both its translation and intracellular stability. Mol. Cell 12, 591–601 (2003).
Zhang, H. et al. The cytidine deaminase CEM15 induces hypermutation in newly synthesized HIV-1 DNA. Nature 424, 94–98 (2003).
Conticello, S.G., Harris, R.S. & Neuberger, M.S. The Vif protein of HIV triggers degradation of the human antiretroviral DNA deaminase APOBEC3G. Curr. Biol. 13, 2009–2013 (2003).
Kobayashi, M., Takaori-Kondo, A., Miyauchi, Y., Iwai, K. & Uchiyama, T. Ubiquitination of APOBEC3G by an HIV-1 Vif-Cullin5-Elongin B-Elongin C complex is essential for Vif function. J. Biol. Chem. 280, 18573–18578 (2005).
Yu, X. et al. Induction of APOBEC3G ubiquitination and degradation by an HIV-1 Vif-Cul5-SCF complex. Science 302, 1056–1060 (2003).
Yu, Y., Xiao, Z., Ehrlich, E.S., Yu, X. & Yu, X.F. Selective assembly of HIV-1 Vif-Cul5-ElonginB-ElonginC E3 ubiquitin ligase complex through a novel SOCS box and upstream cysteines. Genes Dev. 18, 2867–2872 (2004).
Zavrski, I. et al. Proteasome: an emerging target for cancer therapy. Anticancer Drugs 16, 475–481 (2005).
Wichroski, M.J., Ichiyama, K. & Rana, T.M. Analysis of HIV-1 viral infectivity factor-mediated proteasome-dependent depletion of APOBEC3G: correlating function and subcellular localization. J. Biol. Chem. 280, 8387–8396 (2005).
Wichroski, M.J., Robb, G.B. & Rana, T.M. Human retroviral host restriction factors APOBEC3G and APOBEC3F localize to mRNA processing bodies. PLoS Pathog. 2, e41 (2006).
Acknowledgements
The HIV-1 subgenomic proviral vector pNL-A1 harboring HXB2 strain Vif, and the corresponding pNL-A1Δvif were generous gifts of Klaus Strebel. HIV-1 luciferase reporter constructs pNL4-3LucR−E− and pNL4-3ΔVif LucR−E− were provided by Nathaniel Landau through the NIH AIDS Research and Reference Reagent Program, Division of AIDS, NIAID, NIH. The plasmid APOBEC3F-HA was a gift from Michael Malim, and plasmids APOBEC3B-HA and APOBEC3C-HA were generous gifts from Bryan Cullen. We also thank Rana laboratory members for helpful discussions and support and the University of Massachusetts Center for AIDS Research (CFAR) for virology support. This work was supported in part by an NIH grant to T.M.R. and M.S. and by a Developmental award from the UMASS CFAR.
Author information
Authors and Affiliations
Corresponding authors
Supplementary information
Supplementary Text and Figures
Figures 1–10 (PDF 1002 kb)
Rights and permissions
About this article
Cite this article
Nathans, R., Cao, H., Sharova, N. et al. Small-molecule inhibition of HIV-1 Vif. Nat Biotechnol 26, 1187–1192 (2008). https://doi.org/10.1038/nbt.1496
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nbt.1496
This article is cited by
-
Identification of small molecule compounds targeting the interaction of HIV-1 Vif and human APOBEC3G by virtual screening and biological evaluation
Scientific Reports (2018)
-
ASK1 restores the antiviral activity of APOBEC3G by disrupting HIV-1 Vif-mediated counteraction
Nature Communications (2015)
-
Small molecules that inhibit Vif-induced degradation of APOBEC3G
Virology Journal (2014)