Abstract
Haematopoietic stem cells renew blood. Accumulation of DNA damage in these cells promotes their decline, while misrepair of this damage initiates malignancies. Here we describe the features and mutational landscape of DNA damage caused by acetaldehyde, an endogenous and alcohol-derived metabolite. This damage results in DNA double-stranded breaks that, despite stimulating recombination repair, also cause chromosome rearrangements. We combined transplantation of single haematopoietic stem cells with whole-genome sequencing to show that this damage occurs in stem cells, leading to deletions and rearrangements that are indicative of microhomology-mediated end-joining repair. Moreover, deletion of p53 completely rescues the survival of aldehyde-stressed and mutated haematopoietic stem cells, but does not change the pattern or the intensity of genome instability within individual stem cells. These findings characterize the mutation of the stem-cell genome by an alcohol-derived and endogenous source of DNA damage. Furthermore, we identify how the choice of DNA-repair pathway and a stringent p53 response limit the transmission of aldehyde-induced mutations in stem cells.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Accession codes
References
Roswall, N. & Weiderpass, E. Alcohol as a risk factor for cancer: existing evidence in a global perspective. J. Prev. Med. Public Health 48, 1–9 (2015)
Wang, M. et al. Identification of DNA adducts of acetaldehyde. Chem. Res. Toxicol. 13, 1149–1157 (2000)
Lai, C. L. et al. Dominance of the inactive Asian variant over activity and protein contents of mitochondrial aldehyde dehydrogenase 2 in human liver. Alcohol. Clin. Exp. Res. 38, 44–50 (2014)
Yokoyama, A. & Omori, T. Genetic polymorphisms of alcohol and aldehyde dehydrogenases and risk for esophageal and head and neck cancers. Jpn. J. Clin. Oncol. 33, 111–121 (2003)
Garaycoechea, J. I. et al. Genotoxic consequences of endogenous aldehydes on mouse haematopoietic stem cell function. Nature 489, 571–575 (2012)
Langevin, F., Crossan, G. P., Rosado, I. V., Arends, M. J. & Patel, K. J. Fancd2 counteracts the toxic effects of naturally produced aldehydes in mice. Nature 475, 53–58 (2011)
Garaycoechea, J. I. & Patel, K. J. Why does the bone marrow fail in Fanconi anemia? Blood 123, 26–34 (2014)
Hira, A. et al. Variant ALDH2 is associated with accelerated progression of bone marrow failure in Japanese Fanconi anemia patients. Blood 122, 3206–3209 (2013)
Nijnik, A. et al. DNA repair is limiting for haematopoietic stem cells during ageing. Nature 447, 686–690 (2007)
Rossi, D. J. et al. Deficiencies in DNA damage repair limit the function of haematopoietic stem cells with age. Nature 447, 725–729 (2007)
Milyavsky, M . et al. A distinctive DNA damage response in human hematopoietic stem cells reveals an apoptosis-independent role for p53 in self-renewal. Cell Stem Cell 7, 186–197 (2010)
Mohrin, M. et al. Hematopoietic stem cell quiescence promotes error-prone DNA repair and mutagenesis. Cell Stem Cell 7, 174–185 (2010)
Flach, J. et al. Replication stress is a potent driver of functional decline in ageing haematopoietic stem cells. Nature 512, 198–202 (2014)
Niedzwiedz, W. et al. The Fanconi anaemia gene FANCC promotes homologous recombination and error-prone DNA repair. Mol. Cell 15, 607–620 (2004)
Long, D. T., Räschle, M., Joukov, V. & Walter, J. C. Mechanism of RAD51-dependent DNA interstrand cross-link repair. Science 333, 84–87 (2011)
Tan, S. L. W . et al. A class of environmental and endogenous toxins induces BRCA2 haploinsufficiency and genome instability. Cell 169, 1105–1118.e15 (2017)
Bunting, S. F. et al. 53BP1 inhibits homologous recombination in Brca1-deficient cells by blocking resection of DNA breaks. Cell 141, 243–254 (2010)
Adamo, A. et al. Preventing nonhomologous end joining suppresses DNA repair defects of Fanconi anemia. Mol. Cell 39, 25–35 (2010)
Pace, P. et al. Ku70 corrupts DNA repair in the absence of the Fanconi anemia pathway. Science 329, 219–223 (2010)
Osawa, M., Hanada, K., Hamada, H. & Nakauchi, H. Long-term lymphohematopoietic reconstitution by a single CD34-low/negative hematopoietic stem cell. Science 273, 242–245 (1996)
Dykstra, B., Olthof, S., Schreuder, J., Ritsema, M. & de Haan, G. Clonal analysis reveals multiple functional defects of aged murine hematopoietic stem cells. J. Exp. Med. 208, 2691–2703 (2011)
Pang, W. W. et al. Human bone marrow hematopoietic stem cells are increased in frequency and myeloid-biased with age. Proc. Natl Acad. Sci. USA 108, 20012–20017 (2011)
Ghezraoui, H. et al. Chromosomal translocations in human cells are generated by canonical nonhomologous end-joining. Mol. Cell 55, 829–842 (2014)
Wyatt, D. W. et al. Essential roles for Polymerase θ-mediated end joining in the repair of chromosome breaks. Mol. Cell 63, 662–673 (2016)
García-Rubio, M. L. et al. The Fanconi anemia pathway protects genome integrity from R-loops. PLoS Genet. 11, e1005674 (2015)
Schwab, R. A. et al. The Fanconi anemia pathway maintains genome stability by coordinating replication and transcription. Mol. Cell 60, 351–361 (2015)
Jaber, S., Toufektchan, E., Lejour, V., Bardot, B. & Toledo, F. p53 downregulates the Fanconi anaemia DNA repair pathway. Nat. Commun. 7, 11091 (2016)
Oberbeck, N. et al. Maternal aldehyde elimination during pregnancy preserves the fetal genome. Mol. Cell 55, 807–817 (2014)
Kawashima, N. et al. Aldehyde dehydrogenase-2 polymorphism contributes to the progression of bone marrow failure in children with idiopathic aplastic anaemia. Br. J. Haematol. 168, 460–463 (2015)
Gu, Y. et al. Growth retardation and leaky SCID phenotype of Ku70-deficient mice. Immunity 7, 653–665 (1997)
Farley, F. W., Soriano, P., Steffen, L. S. & Dymecki, S. M. Widespread recombinase expression using FLPeR (flipper) mice. Genesis 28, 106–110 (2000)
de Boer, J. et al. Transgenic mice with hematopoietic and lymphoid specific expression of Cre. Eur. J. Immunol. 33, 314–325 (2003)
Donehower, L. A . et al. Mice deficient for p53 are developmentally normal but susceptible to spontaneous tumours. Nature 356, 215–221 (1992)
Giri, S. D. & Chatterjee, A. Modulation of mitomycin C-induced sister chromatid exchanges and cell cycle delay by buthionine sulfoximine and reduced glutathione in mouse bone marrow cells in vivo. Mutat. Res. 413, 227–234 (1998)
Orsburn, B. et al. Differential requirement for H2AX and 53BP1 in organismal development and genome maintenance in the absence of poly(ADP)ribosyl polymerase 1. Mol. Cell. Biol. 30, 2341–2352 (2010)
Geigl, J. B., Uhrig, S. & Speicher, M. R. Multiplex-fluorescence in situ hybridization for chromosome karyotyping. Nat. Protoc. 1, 1172–1184 (2006)
Gribble, S. M. et al. Massively parallel sequencing reveals the complex structure of an irradiated human chromosome on a mouse background in the Tc1 model of Down syndrome. PLoS ONE 8, e60482 (2013)
Reinholdt, L., Ashley, T., Schimenti, J. & Shima, N. Forward genetic screens for meiotic and mitotic recombination-defective mutants in mice. Methods Mol. Biol. 262, 87–107 (2004)
Kent, D ., Dykstra, B. & Eaves, C. Isolation and assessment of long-term reconstituting hematopoietic stem cells from adult mouse bone marrow. Curr. Protoc. Stem Cell Biol. (2007)
Oguro, H., Ding, L. & Morrison, S. J. SLAM family markers resolve functionally distinct subpopulations of hematopoietic stem cells and multipotent progenitors. Cell Stem Cell 13, 102–116 (2013)
Behjati, S. et al. Genome sequencing of normal cells reveals developmental lineages and mutational processes. Nature 513, 422–425 (2014)
Nik-Zainal, S. et al. The genome as a record of environmental exposure. Mutagenesis 30, 763–770 (2015)
Cabezas-Wallscheid, N. et al. Identification of regulatory networks in HSCs and their immediate progeny via integrated proteome, transcriptome, and DNA methylome analysis. Cell Stem Cell 15, 507–522 (2014)
Shen, Z. et al. MPprimer: a program for reliable multiplex PCR primer design. BMC Bioinformatics 11, 143 (2010)
Quail, M. A. et al. A large genome center’s improvements to the Illumina sequencing system. Nat. Methods 5, 1005–1010 (2008)
Krishna, G., Fiedler, R. & Theiss, J. C. Simultaneous evaluation of clastogenicity, aneugenicity and toxicity in the mouse micronucleus assay using immunofluorescence. Mutat. Res. 282, 159–167 (1992)
Acknowledgements
We thank D. Kent for technical advice with single-HSC transplants; R. Berks, A. Middleton, J. Wiles, C. Knox, X. Gong, J. Roe, J. Willems, the ARES staff and Biomed for their help with mouse work; M. Daly, F. Zhang, V. Romashova and M. Balmont for help with flow cytometry; and J. Sale, C. Rada, M. Taylor, Y. L. Wu and members of the Patel laboratory for critical reading of the manuscript. The Human Research Tissue Bank (supported by the NIHR Cambridge Biomedical Research Centre) processed histology. K.J.P. is supported by the MRC and the Jeffrey Cheah Foundation. G.P.C. and L.M. were supported by CRUK. J.I.G. is supported by the Wellcome Trust and King’s College, Cambridge.
Author information
Authors and Affiliations
Contributions
J.I.G., G.P.C. and K.J.P. conceived the study and wrote the manuscript. J.I.G. conducted the majority of the experiments and analysed the data. G.P.C. assisted in the characterization of genomic instability in Aldh2−/−Fancd2−/− mice and single HSC transplantation. F.L. analysed the survival of chicken DT40 cells, performed western blotting and assisted with the analysis of micronucleus samples. L.M. assisted with single cell transplantation and performed the BigBlue in vivo point-mutation analysis. S.L. and F.Y. performed the M-FISH karyotyping of mouse metaphases. N.P. performed validations of indels by targeted deep sequencing. G.G., S.R., S.N.-Z. and M.R.S. provided assistance with the analysis and interpretation of sequencing data.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data figures and tables
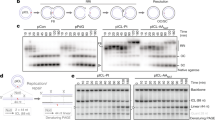
Extended Data Figure 1 Ethanol-induced genomic instability.
a, Left, representative images of bone marrow metaphase spreads from wild-type mice treated with mitomycin C (MMC); n shows the number of SCE events per metaphase. Right, comparison between number of SCEs in the bone marrow of wild-type and Aldh2−/− mice treated with ethanol (5.8 g kg−1) or MMC (1 mg kg−1). Triplicate experiments, 25 metaphases per mouse, n = 75; P calculated by two-sided Mann–Whitney test; data shown as mean and s.e.m. Ethanol causes a strong homologous recombination response in Aldh2−/− mice, comparable to that observed in wild-type mice exposed to MMC. b, Left, representative images of bone marrow metaphase spreads from wild-type and Fanca−/− mice; n shows the number of SCE events per metaphase. Right, quantification of SCEs (duplicate experiments, 25 metaphases per mouse, n = 50; P calculated by two-sided Mann–Whitney test; data shown as mean and s.e.m.). Mice deficient in cross-link repair (Fanca−/−, or Fancd2−/− in Fig. 1a) show a small but significant increase in the number of spontaneous SCE events, indicating that a homologous recombination repair response occurs in the absence of the Fanconi anaemia pathway. c, Scheme depicting the formation of micronucleated erythrocytes. Micronuclei (Mn) generated by fragmentation or mis-segregation of chromosomes during erythrocyte maturation remain in the erythrocyte after extrusion of the main nucleus. These fragments can be detected by a DNA stain (PI+). During maturation, red-cell progenitors lose CD71 expression. Therefore, peripheral CD71+ red cells represent immature, short-lived reticulocytes (Ret) and CD71− cells represent mature, long-lived normochromic erythrocytes (NCEs). d, Proof-of-principle experiment showing the induction of micronucleated reticulocytes 48 h after MMC treatment (1 mg kg−1). P calculated by two-sided Mann–Whitney test; data shown as mean and s.e.m.; n = 29, 8, 20 and 9 mice, left to right. e, Treatment of Aldh2−/− mice with ethanol (5.8 g kg−1) leads to potent micronucleus formation. This induction is comparable to that observed in wild-type mice that were treated with the aneugen vincristine (Vcn, 0.2 mg kg−1, 48 h) or clastogenic γ-irradiation (IR, 400 rad, 48 h)46. P calculated by two-sided Mann–Whitney test; data shown as mean and s.e.m.; n = 29, 15, 10, 11, 25 and 15 mice. f, List of chromosomal aberrations observed in the bone marrow of 8-to-12-week-old untreated Aldh2−/−Fancd2−/− and control mice. g, List of chromosomal aberrations observed in the bone marrow of 8-to-12-week-old Aldh2−/−Fancd2−/− and control mice 48 h after ethanol treatment (5.8 g kg−1, injected intraperitoneally, IP). In f and g, three mice and 30 metaphases per mouse were analysed per condition, and the numbers represent the fraction of abnormal metaphases per mouse. h, Bar chart classifying the type of aberrations for each genotype (90 metaphases per condition). i, Examples of different types of chromosomal aberrations.
Extended Data Figure 2 A single dose of ethanol precipitates bone-marrow failure in Aldh2−/−Fancd2−/− mice.
a, A single dose of ethanol (5.8 g kg−1, injected intraperitoneally) leads to anaemia in Aldh2−/−Fancd2−/− mice one to two months after treatment (P calculated by Mantel-Cox test; n = number of mice). b, Haematoxylin and eosin staining of bone marrow sections 30 days after ethanol treatment (original magnification, ×100). c, Full blood-count analysis for Aldh2−/−Fancd2−/− and control mice, before injection and terminal bleeds after ethanol treatment (P calculated by paired t-test; data shown as mean and s.e.m.; n = number of mice, as in a).
Extended Data Figure 3 Generation of a conditional Fanca allele.
a, Mice carrying the previously reported Fanca− allele (Fancatm1a(EUCOMM)Wtsi) were crossed with mice carrying the FLP recombinase, yielding the Fancafl allele (Fancatm1c(EUCOMM)Wtsi). This allele restores FANCA expression as shown by western blot (Fig. 3). Cre-mediated recombination of Fancafl yields the FancaΔ allele (Fancatm1d(EUCOMM)Wtsi), which lacks exon 3 and leads to loss of FANCA protein (Fig. 3). b, Genotyping PCRs for the wild-type, Fanca− and Fancafl alleles with primers FL033, FL040 and En2A; showing bands of the expected sizes. c, Western blot (single experiment) showing complete absence of FANCA protein in the spleens of Fanca−/− and Fancafl/− Vav1-iCre mice. For gel source data, see Supplementary Fig. 1. d, Determination of the number of exon 3 copies by quantitative PCR. Wild-type, Fanca+/Δ and FancaΔ/Δ mice carry 2, 1 and 0 copies, respectively. Fancafl Vav1-iCre mice show tissue-specific deletion of exon 3 in white blood cells (WBCs) and bone marrow (n = 4 technical replicates; bars: mean, s.d.). e, Microscopic analysis of haematoxylin and eosin-stained sections of testes (original magnification, ×50) from wild-type, Fanca−/−, Fancafl/fl and FancaΔ/Δ males at 12 weeks, showing impaired spermatogenesis in testes of Fanca−/− and FancaΔ/Δ mice (one experiment). f, Sensitivity assay of transformed mouse-embryonic fibroblasts (MEFs) derived from Fanca−/−, Fancafl/fl and FancaΔ/Δ embryos, showing hypersensitivity of both Fanca−/− and FancaΔ/Δ cells to the cross-linking agent mitomycin C (n = number of experiments, each carried out in quadruplicate; bars: mean, s.e.m.).
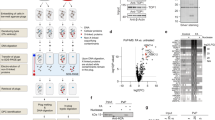
Extended Data Figure 4 Endogenous aldehydes mutate the HSC genome.
Circos plots showing the mutations observed in all sequenced HSC clones (wild type, n = 3; Aldh2−/−, n = 3; Fancd2−/−, n = 4; and Aldh2−/−Fancd2−/−, n = 5 HSC genomes). Substitutions, indels and rearrangements are plotted.
Extended Data Figure 5 Detection of point mutations in mice with the BigBlue reporter system.
a, Chromosome 4 of the BigBlue reporter mouse harbours a λ-phage transgene that contains the mutational target. The phage DNA can be recovered from mouse tissues, packaged into phage and used to infect bacteria. Phage cII mutants can be detected by the ability of these phage to form plaques at 24 °C. b, Quantification of the frequency of cII− -mutant phage recovered from the bone marrow of young Aldh2−/−Fancd2−/− and control mice carrying the BigBlue transgene. ENU-treated mice serve as positive controls for the assay (P calculated by two-sided Mann–Whitney test; data shown as mean and s.e.m.; n = 7, 7, 6, 7 and 6 mice, left to right). c, Relative contribution of the indicated mutation classes to the point-mutation spectra of cII−-mutant phage isolated from the bone marrow. The ENU-mutation spectrum is characterized by T to A transversions and T to C transitions. n is the number of sequenced cII− mutant phage.
Extended Data Figure 6 Aldehyde-induced stress elicits a p53 response.
a, Representative flow cytometry plots for the quantification of p53+ LKS cells from 8-to-12-week-old Aldh2−/−Fancd2−/− and control mice. Cells were collected from wild-type and Trp53−/− mice 2 h after 10 Gy irradiation as positive and negative controls, respectively, for the assay. b, Quantification of the frequency of p53+ cells in different bone-marrow populations. c, Quantification of the frequency of cleaved-caspase-3+ cells in different bone marrow populations by flow cytometry. In b and c, irradiated wild-type and Trp53−/− mice were used as controls. Owing to the low numbers of LKS CD48− CD150+ cells in Aldh2−/−Fancd2−/− mice, the number of p53+ or cleaved-caspase-3+ HSCs could not be determined (data shown as mean and s.e.m.; n = number of mice). d, e, Survival of B cells and myeloid progenitors (CFU-GM) following exposure to acetaldehyde in vitro. Cells were obtained from Fancd2−/−Trp53−/− and control mice. Each point represents the mean of three independent experiments, each carried out in quadruplicate; data shown as mean and s.e.m. f, Frequency of CFU-S12 in the bone marrow of Aldh2−/−Fancd2−/−Trp53−/− and control mice. Each point represents the number of CFU-S12 in the spleen of a single recipient (P calculated by two-sided Mann–Whitney test; data shown as mean and s.e.m.; n = 10–15 mice).
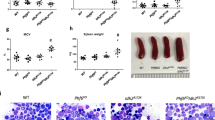
Extended Data Figure 7 p53 deficiency suppresses peripheral-blood cytopenias and ethanol-induced bone-marrow failure in Aldh2−/−Fancd2−/− mice.
a, Full blood count analysis of Aldh2−/−Fancd2−/−Trp53−/− and control mice (8-to-12 weeks old, on a C57BL/6 × 129S4S6/Sv F1 background). A significant increase in the number of white blood cells, red blood cells, platelets and haematocrit was observed in Aldh2−/−Fancd2−/−Trp53−/− mice compared to Aldh2−/−Fancd2−/− mice (P calculated by two-sided Mann–Whitney test; data shown as mean and s.e.m.; n = 17, 16, 21, 14, 18, 12, 18 and 12 mice, left to right). b, Aldh2−/−Fancd2−/−, Aldh2−/−Fancd2−/−Trp53−/− and control mice were treated with ethanol in their drinking water for 10 days as described previously6. Full blood-count analyses were carried out after 10 days of ethanol treatment. c, Bone marrow cellularity after 10 days of ethanol treatment. In b, c, P calculated by two-sided Mann–Whitney test; data shown as mean and s.e.m.; n = 5, 6, 8, 6, 6, 4, 6 and 5 mice, left to right. d, Haematoxylin and eosin staining of bone-marrow sections 10 days after ethanol treatment (original magnification, ×100).
Extended Data Figure 8 Genomic instability in Aldh2−/−Fancd2−/−Trp53−/− mice.
a, Quantification of micronucleated NCEs in the blood of Aldh2−/−Fancd2−/−Trp53−/− and control mice (P calculated by two-sided Mann–Whitney test; data shown as mean and s.e.m.; n = 8 mice). b, List of chromosomal aberrations observed in the bone marrow of 8-to-12 week-old untreated Aldh2−/−Fancd2−/−Trp53−/− and control mice. Three mice and 30 metaphases per mouse were analysed per genotype; the numbers represent the fraction of abnormal metaphases per mouse. c, Bar chart classifying the types of aberrations for each genotype (90 metaphases per condition). d, Examples of two metaphases from an Aldh2−/−Fancd2−/−Trp53−/− mouse.
Extended Data Figure 9 Validation of indels by targeted deep sequencing.
a, Scheme depicting the generation of HSC clones by transplantation of single stem cells, subsequent whole-genome sequencing and validation of indel calls by amplicon deep sequencing. On the basis of the indel location from 20× whole-genome sequencing, we designed multiplex PCRs and deep sequenced the PCR products to higher coverage (100–100,000×) to confirm that the calls were not sequencing artefacts. In addition, we attempted to detect indels in DNA samples of bone-marrow cells from the mice that provided the transplanted HSCs. b, Coverage depth and VAF of the filtered set of indel calls from whole-genome sequencing (n = 342 indels; box plot shows the mean, box edges represent the first and third quartiles, whiskers extend over 10–90% of data). c, Coverage depth and VAF of the indel calls from deep sequencing validation (n = 159 locations; box plot shows the mean, box edges represent the first and third quartiles, whiskers extend over 10–90% of data). One hundred and fifty-nine locations had coverage greater than 100× and were used for the analysis. We could validate the presence of 91.2% of the initial calls; 14/159 (8.8%) calls had VAF <0.1 and were deemed false positives (indicated by grey shading). Note that the VAF distribution is centred tightly around 0.5, confirming the clonal nature of most indels. d, We used targeted deep sequencing to look for indel calls in bone-marrow samples from the mice that provided the transplanted HSCs. In most cases, the calls were below the detection limit of the assay (VAF <0.0001). However, we could detect indels from two Aldh2−/−Fancd2−/− HSCs, indicative of ‘clonal haematopoiesis’ in these mice (accounting for 0.7 and 21.4% of blood production, respectively). Data shown as mean and s.e.m.; n = 13 and 7 indels.
Extended Data Figure 10 Validation of rearrangements by PCR.
a, Scheme depicting the generation of HSC clones by transplantation of single stem cells, subsequent whole-genome sequencing and validation of rearrangement calls by PCR. We designed primers for nested PCRs flanking the breakpoints calculated by the BRASS algorithm, and the identity of the products was confirmed by Sanger sequencing. In addition, we attempted to detect the rearrangements in DNA samples of bone-marrow cells from the mice that provided the transplanted HSCs, demonstrating that these changes did not arise during clonal expansion and were present in the stem cell at the time of transplantation. b, Agarose gels (one experiment) showing presence of specific PCR amplification from DNA of HSC clones, absence in matched germline samples from the tail of the same mouse and, in some cases, detection in bone-marrow tissue that predates the transplants. PCR amplification in these samples is dependent on the contribution of the transplanted HSC to blood production, and the sensitivity of each PCR. Gel source data is shown in Supplementary Fig. 1. c, List summarizing the rearrangements found in 28 HSC clones and the results from b. All 27 rearrangements could be detected by PCR and confirmed by Sanger sequencing. 16/27 (59%) rearrangements could be detected before transplantation.
Extended Data Figure 11 Mechanisms to maintain genetic integrity and suppress mutagenesis by endogenous aldehydes in HSCs.
a, Aldehyde catabolism and Fanconi anaemia (FA)-pathway-mediated DNA repair constitute two distinct tiers of protection against aldehyde damage. Loss of this protection leads to the accumulation of DNA damage and mutagenesis. Passage of mutated genetic information is prevented by the activation of p53, leading to HSC loss. b, In the absence of a functional Fanconi anaemia pathway, aldehyde lesions degenerate into DNA DSBs that can be repaired through error-free recombination. However, this mechanism is not sufficient to fully compensate for Fanconi anaemia inactivation, leading to the engagement of both classical and alternative end-joining, and subsequent mutagenic repair.
Supplementary information
Supplementary Figure 1
This file contains source gels. a, Fanca genotyping, related to Extended data Fig. 3b. b, Western blots showing lack of Fanca protein, related to Extended data Fig. 3c. c, Agarose gels for the validation of rearrangements by PCR, related to Extended data Fig. 10. (PDF 8806 kb)
Supplementary Figure 2
This file contains flow cytometry gating strategies. a, Gating strategy to quantify the frequency of Mn-NCE and Mn-Ret relating to Fig. 2b and g, Fig. 3f and Extended data Fig. 8a. b, Gating strategy for the quantification of LKS and HSCs relating to Fig. 3b-d and Fig. 6b. c, Gating strategy for the sorting and transplantation of single HSCs relating to Fig. 4b and 6d. d, Gating strategy to assess the chimaerism in total peripheral blood, B, T and myeloid cells relating to Fig. 4c. (PDF 2452 kb)
Supplementary Table 1
This file contains generation of Fanca-/-Ku70-/- and FancaF/-Ku70-/- Vav1-iCre mice. a, Fanca+/- crosses in a C57BL/6 background showing that Fanca-/- mice are genotyped at sub-Mendelian ratios 2-3 weeks after birth (13.3% instead of the expected 25%, Fisher’s exact test: P<0.0001). b, Ku70+/- crosses in a C57BL/6 background showing that Ku70-/- mice are genotyped at sub-Mendelian ratios 2-3 weeks after birth (8.5% instead of the expected 25%, Fisher’s exact test: P<0.0001). c, Fanca+/- Ku70+/- crosses were set up to generate Fanca-/-Ku70-/- mice. Although Mendelian segregation predicts 6.25% of the pups to be Fanca-/-Ku70-/-, the actual expected ratio is 1.12% if the sub-Mendelian ratios of both Fanca-/- and Ku70-/- are taken into account. However, no Fanca-/-Ku70-/- pups were found from 463 pups genotyped (Fisher’s exact test: P<0.0307). d, FancaF/F Ku70+/- x Fanca+/- Ku70+/- Vav1-iCre crosses were set up to generate FancaF/-Ku70-/- Vav1-iCre mice in a C57BL/6 background. These mice were born at the expected ratio, which takes into account the sub-Mendelian frequency of Ku70-/- mice (1.6% observed and 2.1% expected, Fisher’s exact test: P<0.6). (PDF 613 kb)
Supplementary Table 2
This file contains p53 deficiency does not suppress the embryonic lethality of Aldh2-/-Fancd2-/- mice in Aldh2-/- mothers. a, b, In agreement with a previous report 6, Aldh2-/-Fancd2-/- mice on a C57BL/6 x 129S4S6/Sv F1 background could be obtained from Aldh2+/- 129S4S6/Sv mothers but could not be generated from Aldh2-/- 129S4S6/Sv mothers (c). d, Similar crosses were set up to obtain Aldh2-/-Fancd2-/-p53-/- mice on a C57BL/6 x 129S4S6/Sv F1 background, triple mutant mice were born at expected ratios from Aldh2+/- 129S4S6/Sv mothers. e, No Aldh2-/-Fancd2-/-p53-/- mice could be obtained from Aldh2-/- 129S4S6/Sv mothers, showing that lack of p53 is not sufficient to prevent loss of these embryos (P: Fisher’s exact test comparing the number of observed and expected double or triple mutants). (PDF 658 kb)
Rights and permissions
About this article
Cite this article
Garaycoechea, J., Crossan, G., Langevin, F. et al. Alcohol and endogenous aldehydes damage chromosomes and mutate stem cells. Nature 553, 171–177 (2018). https://doi.org/10.1038/nature25154
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature25154
This article is cited by
-
p53 regulates diverse tissue-specific outcomes to endogenous DNA damage in mice
Nature Communications (2024)
-
Microplastics dampen the self-renewal of hematopoietic stem cells by disrupting the gut microbiota-hypoxanthine-Wnt axis
Cell Discovery (2024)
-
FAAP100 is required for the resolution of transcription-replication conflicts in primordial germ cells
BMC Biology (2023)
-
Cancers make their own luck: theories of cancer origins
Nature Reviews Cancer (2023)
-
Aged hematopoietic stem cells entrap regulatory T cells to create a prosurvival microenvironment
Cellular & Molecular Immunology (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.