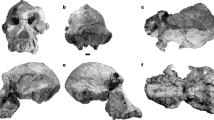
Abstract
The evolutionary history of extant hominoids (humans and apes) remains poorly understood. The African fossil record during the crucial time period, the Miocene epoch, largely comprises isolated jaws and teeth, and little is known about ape cranial evolution. Here we report on the, to our knowledge, most complete fossil ape cranium yet described, recovered from the 13 million-year-old Middle Miocene site of Napudet, Kenya. The infant specimen, KNM-NP 59050, is assigned to a new species of Nyanzapithecus on the basis of its unerupted permanent teeth, visualized by synchrotron imaging. Its ear canal has a fully ossified tubular ectotympanic, a derived feature linking the species with crown catarrhines. Although it resembles some hylobatids in aspects of its morphology and dental development, it possesses no definitive hylobatid synapomorphies. The combined evidence suggests that nyanzapithecines were stem hominoids close to the origin of extant apes, and that hylobatid-like facial features evolved multiple times during catarrhine evolution.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Fleagle, J. G. Primate Adaptation and Evolution, 3rd edn (Academic, 2013)
Begun, D. R. in Handbook of Paleoanthropology (eds Henke, W. & Tattersall, I. ) 1261–1332 (Springer, 2015)
Hill, A. & Ward, S. C. Origin of the Hominidae: the record of African large hominoid evolution between 14 My and 4 My. Yearb. Phys. Anthropol.31, 49–83 (1988)
Ward, S. C. & Duren, D. in The Primate Fossil Record (ed. Hartwig, W. C. ) 385–397 (Cambridge Univ. Press, 2002)
Bernor, R. L. New apes fill the gap. Proc. Natl Acad. Sci. USA104, 19661–19662 (2007)
Lebatard, A. E. et al. Cosmogenic nuclide dating of Sahelanthropus tchadensis and Australopithecus bahrelghazali: Mio–Pliocene hominids from Chad.Proc. Natl Acad. Sci. USA105, 3226–3231 (2008)
Tafforeau, P. et al. Applications of X-ray synchrotron microtomography for non-destructive 3D studies of paleontological specimens.Appl. Phys. Adv. Mater.83, 195–202 (2006)
Rossie, J. B. & MacLatchy, L. A new pliopithecoid genus from the early Miocene of Uganda. J. Hum. Evol.50, 568–586 (2006)
Benefit, B. R. & McCrossin, M. L. The lacrimal fossa of Cercopithecoidea, with special reference to cladistic analysis of Old World monkey relationships. Folia Primatol.60, 133–145 (1993)
Rae, T. C. Mosaic evolution in the origin of the Hominoidea.Folia Primatol.70, 125–135 (1999)
Benefit, B. R. & McCrossin, M. L. Facial anatomy of Victoriapithecus and its relevance to the ancestral cranial morphology of Old World monkeys and apes. Am. J. Phys. Anthropol.92, 329–370 (1993)
Alba, D. M . et al. Miocene small-bodied ape from Eurasia sheds light on hominoid evolution. Science350, aab2625 (2015)
Harrison, T. New fossil anthropoids from the middle Miocene of East Africa and their bearing on the origin of the Oreopithecidae. Am. J. Phys. Anthropol.71, 265–284 (1986)
Harrison, T. in The Primate Fossil Record (ed. W. C. Hartwig ) 311–338 (Cambridge Univ. Press, 2002)
Harrison, T. in Cenozoic Mammals of Africa (eds Werdelin, L. & Sanders, W. J. ) 429–469 (Univ. California Press, 2010)
Pickford, M. & Kunimatsu, Y. Catarrhines from the Middle Miocene (ca. 14.5 Ma) of Kipsaraman, Tugen Hills, Kenya. Anthropol. Sci.113, 189–224 (2005)
Tafforeau, P. & Smith, T. M. Nondestructive imaging of hominoid dental microstructure using phase contrast X-ray synchrotron microtomography.J. Hum. Evol.54, 272–278 (2008)
Smith, T. & Tafforeau, P. New visions of dental tissue research: tooth development, chemistry, and structure. Evol. Anthropol.17, 213–226 (2008)
Bromage, T. G. et al. Lamellar bone is an incremental tissue reconciling enamel rhythms, body size, and organismal life history. Calcif. Tissue Int.84, 388–404 (2009)
Martin, L. Significance of enamel thickness in hominoid evolution.Nature314, 260–263 (1985)
Smith, T. M. et al. Dental evidence for ontogenetic differences between modern humans and Neanderthals. Proc. Natl Acad. Sci. USA107, 20923–20928 (2010)
Olejniczak, A. J. Micro-Computed Tomography of Primate Molars. PhD thesis, Stony Brook Univ. (2006)
Dirks, W. & Bowman, J. E. Life history theory and dental development in four species of catarrhine primates. J. Hum. Evol.53, 309–320 (2007)
Gonzales, L. A., Benefit, B. R., McCrossin, M. L. & Spoor, F. Cerebral complexity preceded enlarged brain size and reduced olfactory bulbs in Old World monkeys. Nat. Commun.6, 7580 (2015)
Spoor, F. & Zonneveld, F. Comparative review of the human bony labyrinth. Am. J. Phys. Anthropol.107 (Suppl. 27), 211–251 (1998)
Ryan, T. M . et al. Evolution of locomotion in Anthropoidea: the semicircular canal evidence. Proc. R. Soc. B279, 3467–3475 (2012)
Stevens, N. J. et al. Palaeontological evidence for an Oligocene divergence between Old World monkeys and apes. Nature497, 611–614 (2013)
Harrison, T. & Rook, L. in Function, Phylogeny, and Fossils: Miocene Hominoid Evolution and Adaptations (eds Begun, D. R., Ward, C. V. & Rose, M. D. ) 327–362 (Plenum, 1997)
Begun, D. R., Ward, C. V. & Rose, M. D. (eds) in Function, Phylogeny, and Fossils: Miocene Hominoid Evolution and Adaptations 389–415 (Plenum, 1997)
Begun, D. R. in Hominoid Evolution and Climate Change in Europe Vol. 2 (eds De Bonis, L., Koufos, G. & Andrews, P. ) 231–253 (Cambridge Univ. Press, 2001)
Begun, D. R., Nargolwalla, M. C. & Kordos, L. European Miocene hominids and the origin of the African ape and human clade. Evol. Anthropol.21, 10–23 (2012)
McCrossin, M. L. An oreopithecid proximal humerus from the middle Miocene of Maboko Island, Kenya. Int. J. Primatol.13, 659–677 (1992)
Kelley, J., Ward, S., Brown, B., Hill, A. & Duren, D. L. Dental remains of Equatorius africanus from Kipsaramon, Tugen Hills, Baringo District, Kenya. J. Hum. Evol.42, 39–62 (2002)
Behrensmeyer, A. K., Deino, A. L., Hill, A., Kingston, J. D. & Saunders, J. J. Geology and geochronology of the middle Miocene Kipsaramon site complex, Muruyur Beds, Tugen Hills, Kenya. J. Hum. Evol.42, 11–38 (2002)
Gilbert, C. C., Goble, E. D. & Hill, A. Miocene Cercopithecoidea from the Tugen Hills, Kenya. J. Hum. Evol.59, 465–483 (2010)
Dodson, R. G. Geology of the Area South of Lodwar, Degree Sheet 18, N.E. Quarter. Report No. 87 (Geological Survey of Kenya, 1971)
Morley, C. K. et al. in Geoscience of Rift Systems - Evolution of East Africa. AAPG Studies in Geology Vol.44 (ed. Morley, C. K. ) 19–54 (American Association of Petroleum Geologists, 1999)
Hilgen, F. J., Lourens, L. J. & Van Dam, J. A. in The Geologic Time Scale (eds Gradstein, F. M., Ogg, J. G., Schmitz, M. D. & Ogg, G. M. ) 923–978 (Elsevier, 2012)
Kuiper, K. F. et al. Synchronizing rock clocks of Earth history.Science320, 500–504 (2008)
Steiger, R. H. & Jäger, E. Subcommission on geochronology: convention on the use of decay constants in geo- and cosmochronology. Earth Planet. Sci. Lett36, 359–362 (1977)
Dalrymple, G. B ., Alexander, E. C. Jr, Lanphere, M. A. & Kraker, G. P. Irradiation of Samples for 40Ar/39Ar Dating Using The Geological Survey TRIGA Reactor. US Geological Survey Professional Paper No. 1176 (US Geological Survey, 1981)
Dalrymple, G. B. & Ryder, G. 40Ar/39Ar age spectra of Apollo 15 impact melt rocks by laser heating and their bearing on the history of lunar basin formation. J. Geophys. Res.98 (E7), 13085–13095 (1993)
Spoor, C. F. The Comparative Morphology and Phylogeny of The Human Bony Labyrinth. PhD thesis, Utrecht Univ. (1993)
Spoor, F. et al. The primate semicircular canal system and locomotion. Proc. Natl Acad. Sci. USA104, 10808–10812 (2007)
Smith, R. J. & Jungers, W. L. Body mass in comparative primatology. J. Hum. Evol.32, 523–559 (1997)
Chamberlain, A. T. A Taxonomic Review and Phylogenetic Analysis of Homo habilis. PhD thesis, Univ. Liverpool (1987)
Rafferty, K. L., Walker, A., Ruff, C. B., Rose, M. D. & Andrews, P. J. Postcranial estimates of body weight in Proconsul, with a note on a distal tibia of P. major from Napak, Uganda. Am. J. Phys. Anthropol.97, 391–402 (1995)
Hammer, Ø., Harper, D. A. T. & Ryan, P. D. PAST: paleontological statistics software package for education and data analysis.Palaeontol. Electronica4, 4 (2001)
Moyà-Solà, S. & Kohler, M. New partial cranium of Dryopithecus Lartet, 1863 (Hominoidea, Primates) from the Upper Miocene of Can-Llobateres, Barcelona, Spain. J. Hum. Evol.29, 101–139 (1995)
Cameron, D. W. A revised systematic scheme for the Eurasian Miocene fossil Hominidae. J. Hum. Evol.33, 449–477 (1997)
Benefit, B. R., Gitau, S. N., McCrossin, M. L. & Palmer, A. K. A mandible of Mabokopithecus clarki sheds new light on oreopithecid evolution. Am. J. Phys. Anthropol.105 (Suppl. 26), 109 (1998)
Wiens, J. J. (ed.) in Phylogenetic Analysis of Morphological Data. Comparative Evolutionary Series 115–145 (Smithsonian Institution Press, 2000)
Gilbert, C. C., Stanley, W. T., Olson, L. E., Davenport, T. R. B. & Sargis, E. J. Morphological systematics of the kipunji (Rungwecebus kipunji) and the ontogenetic development of phylogenetically informative characters in the Papionini. J. Hum. Evol.60, 731–745 (2011)
Isler, K. et al. Endocranial volumes of primate species: scaling analyses using a comprehensive and reliable data set. J. Hum. Evol.55, 967–978 (2008)
Acknowledgements
We thank the Government of Kenya for permission to do this research, the National Museums of Kenya, the American Museum of Natural History, Harvard Museum of Comparative Zoology, the Musée des Confluences of Lyon, and the Digital Morphology Museum (KUPRI) for access to specimens in their care, and the Turkana Basin Institute for field support. The Leakey Foundation and trustee G. Getty, the Foothill-De Anza Foundation, De Anza College Design and Manufacturing Technologies Department, the National Geographic Society, the European Synchrotron Radiation Facility (beamlines ID 19 and BM5), and the Max Planck Society funded fieldwork or laboratory studies. We are grateful to M. Kibunjia and F. Manthi for facilitating permission to transport the specimen to Grenoble for scanning, and to D. Berthet, E. Delson, M. Hill, J.-J. Hublin, D. Plotzki, and H. Temming for facilitating computed tomography scans of comparative specimens. We are grateful to R. David, M. Gilleland, L. Gonzales, T. Harrison, J. Kelley, Y. Kunimatsu, L. Martin, M. Leakey, K. O’Maley, M. Pickford, C. Rendiger, B. Sokhi, and A. Stoessel for help with this research. We thank the field crew, A. Ekes, B. Ewoi, J. Ekusi, A. Ekuon, C. Nyete, and T. Ngundo, for their tireless efforts in the field that led to the discovery of the specimen, C. Kiarie for preparing the fossil, and B. Monroy for preparing some of the graphics. We particularly thank J. Ekusi for discovering KNM-NP 59050.
Author information
Authors and Affiliations
Contributions
I.N. and F.S. designed the study. I.N., C.F., E.R.M., and D.L.F. did field research. J.G.F., C.C.G., F.S., and K.D.P. collected and analysed the comparative data on gross dental and cranial morphology, and J.G.F. and C.C.G. prepared the diagnosis. P.T., C.B., and F.S. did the image analyses and made virtual reconstructions, P.T. and C.B. performed the dental development study, C.F., S.M., D.L.F., J.F., and Z.E. studied the geological context, and C.C.G. and K.D.P. did the phylogenetic analyses. F.S., I.N., and E.R.M. wrote the paper with contributions from C.C.G., P.T., and J.G.F.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Additional information
Reviewer InformationNature thanks B. Benefit and the other anonymous reviewer(s) for their contribution to the peer review of this work.
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data figures and tables
Extended Data Figure 1 KNM-NP 50950 and Napudet.
a–f, Specimen as preserved in left lateral view (a), anterior view (b), right lateral view (c), superior view (d), posterior view (e), and inferior view (f), visualized using three-dimensional X-ray microtomography. g, The Napudet locality on the western side of Lake Turkana, Kenya.
Extended Data Figure 2 Stratigraphy and dating.
a, Stratigraphic sections at localities CSF 2015-1, -2 and -3, and placement of KNM-NP 59050 (orange star). See Supplementary Table 2 for associated GPS coordinates. b, Incremental release spectra for sample 15-NPD-03, corrected for the trapped 40Ar–36Ar component. The plateau obtained provided the most representative age for this sample (see Methods for details).
Extended Data Figure 3 Cranial comparison of KNM-NP 59050 with juvenile catarrhines.
a, KNM-NP 59050 as preserved.b, KNM-NP 59050 as retrodistorted.c, Hoolock sp. d, G. gorilla. e,P. troglodytes. f, P. pygmaeus. g, P. ursinus. Each specimen is shown, from left to right, in posterior, left lateral, anterior, superior, and inferior views. Rendering size is standardized on the basis of overall cranial size. Scale bars, 2 cm.
Extended Data Figure 4 Juvenile and adult facial size.
a–d, Box plots of relative snout size expressed by orbitale inferior to prosthion (a, b) and maxillary height from orbitale inferior to the alveolar margin (c, d). Values are size-adjusted on the basis of a geometric mean of cranial measurements for juveniles (a, c) and the square root of M1 area for adults (b, d). Hylobatids have significantly smaller snouts than extant great apes (analysis of variance (ANOVA) with Bonferroni correction, P < 0.01), both in juveniles and in adults. KNM-NP 59050 falls in the range of extant hylobatids. e, Orbit size (square root of maximum height × breadth in millimetres) compared to cranial size (geometric mean in millimetres) for KNM-NP 59050 (star), as well as for juvenile hylobatids (circles), Pongo (triangles), and African great apes (squares). Box and whiskers as in Fig. 3, and samples are given in Supplementary Data 2.
Extended Data Figure 5 Juvenile and adult cranial features.
a, b, Box plots of interorbital breadth in juveniles (a) and adults (b), size-adjusted as in Extended Data Fig. 4a–d. Hylobatids and African great apes have significantly greater interorbital breadth than Pongo (ANOVA with Bonferroni correction, P < 0.01), both in juveniles and in adults. KNM-NP 59050 falls exclusively in the range of extant hylobatid juveniles.c, d, Box plots of the nasal aperture width in juveniles (c) and adults (d), size-adjusted as in Extended Data Fig. 4a–d. Pongo has a significantly narrower nasal aperture than African great apes both in juveniles and in adults (ANOVA with Bonferroni correction, P < 0.01). KNM-NP 59050 falls closest to the median of extant Pongo juveniles.e, f, Ratio of nasion–bregma/bregma–lambda in juveniles (e) and adults (f). Hylobatids have a significantly greater ratio compared with the great apes (ANOVA with Bonferroni correction, P < 0.01) both in juveniles and in adults. KNM-NP 59050 falls exclusively in the range of extant great ape juveniles, a ratio probably maintained into adulthood. Box and whiskers as in Fig. 3, and samples are given in Supplementary Data 2.
Extended Data Figure 6 Virtual histological slices of KNM-NP 59050.
a, Three-dimensional rendering of the right unerupted permanent teeth germs. b, Virtual histological slices of these teeth with coloured arrows showing the stress lines in dentine used for developmental cross-correlations of all the teeth and cusps. Scale bar, 5 mm. c, Detail of the Andresen lines in the dentine, and reference stress lines in the right I2 germ. Colours correspond to the lines represented in Fig. 4. Scale bar, 1 mm. d, Details of the enamel microstructure of this tooth, showing four daily lines (dark orange arrows) between consecutives Retzius lines (light orange arrows), indicating a long-period line periodicity of 5 days. Scale bar, 100 μm.
Extended Data Figure 7 Endocranial volume and the bony labyrinth.
a, b, Bivariate double logarithmic plots of the endocranial volume (a, ECV, in millilitres) and the mean radius of curvature of the three semicircular canals (b, SC-R, in millimetres) against body mass (BM, in grams) for extant hominids (blue dots), hylobatids (cyan dots), cercopithecids (small open triangles), and strepsirrhines (small black diamonds, a only), Aegyptopithecus (black square), Pliobates (purple dot, a), Saadinius (purple dot, b), Victoriapithecus (grey triangle), Ekembo (magenta dot), Oreopithecus (orange dot), Hispanopithecus (green dot), Rudapithecus (teal dot), and KNM-NP 59050 (red dot). For KNM-NP 59050 and Oreopithecus (b only), the line represents a range of body mass values (Supplementary Note 1). Reduced major axis regression lines are given for the extant hominids, hylobatids (a only), cercopithecids, and strepsirrhines (a only). Data from refs 43, 44, 45, 54 and Supplementary Note 1 and Supplementary Table 1. c–n, Lateral (c–h) and superior (i–n) views of the left bony labyrinth of P. troglodytes (c, i), G. gorilla (d, j), Pongo abelli (e, k), KNM-NP 59050 (f, l), Symphalangus syndactylus (g, m), and H. muelleri (h, n). Scale bar, 5 mm. The extant species are represented by mean shapes of between 6 and 39 specimens per species (Supplementary Table 1).
Extended Data Figure 8 Phylogenetic analyses of N. alesi.
Strict consensus trees of the unscaled (a) and scaled (b) analyses showing the placement of KNM-NP 59050 as part of the Nyanzapithecus clade. Trees show the summary of ten most parsimonious trees (unscaled: 1382; consistency index = 0.289; homoplasy index = 0.711; retention index = 0.597) and three most parsimonious trees (scaled: TL = 1274.25; consistency index = 0.288; homoplasy index = 0.712; retention index = 0.599). Numbered nodes correspond with clades listed in Extended Data Table 2. Bootstrap support values >50% (1,000 replicates) are shown below nodes. Note that in the unscaled analysis, aSivapithecus + Pongo clade is supported in 72% of bootstrap replicates, but not found in the strict consensus tree.
Supplementary information
Supplementary Information
This file contains Supplementary Note 1, Supplementary Tables 1-3 and additional references. (PDF 263 kb)
Supplementary Data 1
This zipped file contains unerupted tooth crowns of KNM-NP 59050. Three-dimensional surface models of the left and right I1, M1 and M2 are provided as 3D PDFs. Surfaces rendered from segmented synchrotron microtomographic images. (ZIP 14911 kb)
Supplementary Data 2
Comparative cranial and dental measurements. This file contains measurements of KNM-NP 59050 and all individual juvenile and adult specimens in the comparative sample. (XLSX 49 kb)
Supplementary Data 3
40Ar/39Ar incremental-heating data and analytical conditions for sample 15-NPD-03. (XLSX 23 kb)
Supplementary Data 4
Calculation sheet for dental development of KNM-NP 59050. The calculations to determine the age at death and the general developmental timing of Nyanzapithecus alesi based on dental increments. (XLSX 43 kb)
Supplementary Data 5
Morphological Characters used in Phylogenetic Analyses. This spreadsheet lists the characters scored in this study along with character state definitions, character type designations, and the source of each character. (XLSX 38 kb)
Supplementary Data 6
The matrix used in phylogenetic analysis. This file contains the Nexus file with the character matrix used in this study, written with Mesquite version 2.75, build 566 (www.mesquiteproject.org ). The file can be read with any text editor. (TXT 32 kb)
Rights and permissions
About this article
Cite this article
Nengo, I., Tafforeau, P., Gilbert, C. et al. New infant cranium from the African Miocene sheds light on ape evolution. Nature 548, 169–174 (2017). https://doi.org/10.1038/nature23456
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature23456
This article is cited by
-
The Miocene primate Pliobates is a pliopithecoid
Nature Communications (2024)
-
A new ape from Türkiye and the radiation of late Miocene hominines
Communications Biology (2023)
-
Palaeoecological differences underlie rare co-occurrence of Miocene European primates
BMC Biology (2021)
-
Early anthropoid femora reveal divergent adaptive trajectories in catarrhine hind-limb evolution
Nature Communications (2019)
-
Wrist morphology reveals substantial locomotor diversity among early catarrhines: an analysis of capitates from the early Miocene of Tinderet (Kenya)
Scientific Reports (2019)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.