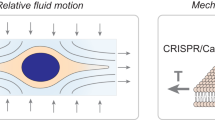
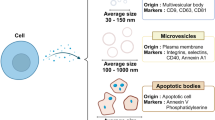
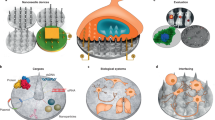
Abstract
Intracellular delivery of materials has become a critical component of genome-editing approaches, ex vivo cell-based therapies, and a diversity of fundamental research applications. Limitations of current technologies motivate development of next-generation systems that can deliver a broad variety of cargo to diverse cell types. Here we review in vitro and ex vivo intracellular delivery approaches with a focus on mechanisms, challenges and opportunities. In particular, we emphasize membrane-disruption-based delivery methods and the transformative role of nanotechnology, microfluidics and laboratory-on-chip technology in advancing the field.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Yin, H. et al. Non-viral vectors for gene-based therapy. Nat. Rev. Genet. 15, 541–555 (2014)
Wittrup, A. & Lieberman, J. Knocking down disease: a progress report on siRNA therapeutics. Nat. Rev. Genet. 16, 543–552 (2015)
Naldini, L. Ex vivo gene transfer and correction for cell-based therapies. Nat. Rev. Genet. 12, 301–315 (2011)
Naldini, L. Gene therapy returns to centre stage. Nature 526, 351–360 (2015)
June, C. H., Riddell, S. R. & Schumacher, T. N. Adoptive cellular therapy: a race to the finish line. Sci. Transl. Med. 7, 280ps7 (2015)
Rosenberg, S. A. & Restifo, N. P. Adoptive cell transfer as personalized immunotherapy for human cancer. Science 348, 62–68 (2015)
Tebas, P. et al. Gene editing of CCR5 in autologous CD4 T cells of persons infected with HIV. N. Engl. J. Med. 370, 901–910 (2014)
Sather, B. D. et al. Efficient modification of CCR5 in primary human hematopoietic cells using a megaTAL nuclease and AAV donor template. Sci. Transl. Med. 7, 307ra156 (2015)
Doudna, J. A. & Charpentier, E. Genome editing. The new frontier of genome engineering with CRISPR-Cas9. Science 346, 1258096 (2014)
Cox, D. B., Platt, R. J. & Zhang, F. Therapeutic genome editing: prospects and challenges. Nat. Med. 21, 121–131 (2015)
Kim, D. et al. Generation of human induced pluripotent stem cells by direct delivery of reprogramming proteins. Cell Stem Cell 4, 472–476 (2009)
Warren, L. et al. Highly efficient reprogramming to pluripotency and directed differentiation of human cells with synthetic modified mRNA. Cell Stem Cell 7, 618–630 (2010)
Anokye-Danso, F. et al. Highly efficient miRNA-mediated reprogramming of mouse and human somatic cells to pluripotency. Cell Stem Cell 8, 376–388 (2011)
Marschall, A. L. J. et al. Delivery of antibodies to the cytosol: debunking the myths. Mabs 6, 943–956 (2014)
Liu, J., Wen, J., Zhang, Z., Liu, H. & Sun, Y. Voyage inside the cell: microsystems and nanoengineering for intracellular measurement and manipulation. Microsyst. Nanoeng. 1, 15020 (2015)
Lee, S. E., Liu, G. L., Kim, F. & Lee, L. P. Remote optical switch for localized and selective control of gene interference. Nano Lett. 9, 562–570 (2009)
Heller, D. A., Baik, S., Eurell, T. E. & Strano, M. S. Single-walled carbon nanotube spectroscopy in live cells: Towards long-term labels and optical sensors. Adv. Mater. 17, 2793–2799 (2005)
Michalet, X. et al. Quantum dots for live cells, in vivo imaging, and diagnostics. Science 307, 538–544 (2005)
Karra, D. & Dahm, R. Transfection techniques for neuronal cells. J. Neurosci. 30, 6171–6177 (2010)
Peer, D. A daunting task: manipulating leukocyte function with RNAi. Immunol. Rev. 253, 185–197 (2013)
Hendel, A. et al. Chemically modified guide RNAs enhance CRISPR-Cas genome editing in human primary cells. Nat. Biotechnol. 33, 985–989 (2015)
Thomas, C. E., Ehrhardt, A. & Kay, M. A. Progress and problems with the use of viral vectors for gene therapy. Nat. Rev. Genet. 4, 346–358 (2003)
Kay, M. A. State-of-the-art gene-based therapies: the road ahead. Nat. Rev. Genet. 12, 316–328 (2011)
Mintzer, M. A. & Simanek, E. E. Nonviral vectors for gene delivery. Chem. Rev. 109, 259–302 (2009)
Yoo, J. W., Irvine, D. J., Discher, D. E. & Mitragotri, S. Bio-inspired, bioengineered and biomimetic drug delivery carriers. Nat. Rev. Drug Discov. 10, 521–535 (2011)
Torchilin, V. P. Multifunctional, stimuli-sensitive nanoparticulate systems for drug delivery. Nat. Rev. Drug Discov. 13, 813–827 (2014)
Mitragotri, S., Burke, P. A. & Langer, R. Overcoming the challenges in administering biopharmaceuticals: formulation and delivery strategies. Nat. Rev. Drug Discov. 13, 655–672 (2014)
Khalil, I. A., Kogure, K., Akita, H. & Harashima, H. Uptake pathways and subsequent intracellular trafficking in nonviral gene delivery. Pharmacol. Rev. 58, 32–45 (2006)
Sahay, G., Alakhova, D. Y. & Kabanov, A. V. Endocytosis of nanomedicines. J. Control. Release 145, 182–195 (2010)
Stewart, M. P., Lorenz, A., Dahlman, J. & Sahay, G. Challenges in carrier-mediated intracellular delivery: moving beyond endosomal barriers. Wiley Interdiscip. Rev. Nanomed. Nanobiotechnol. 8, 465–478 (2016)
Sahay, G. et al. Efficiency of siRNA delivery by lipid nanoparticles is limited by endocytic recycling. Nat. Biotechnol. 31, 653–658 (2013)
Gilleron, J. et al. Image-based analysis of lipid nanoparticle-mediated siRNA delivery, intracellular trafficking and endosomal escape. Nat. Biotechnol. 31, 638–646 (2013)
Schaffer, D. V., Fidelman, N. A., Dan, N. & Lauffenburger, D. A. Vector unpacking as a potential barrier for receptor-mediated polyplex gene delivery. Biotechnol. Bioeng. 67, 598–606 (2000)
Lv, H., Zhang, S., Wang, B., Cui, S. & Yan, J. Toxicity of cationic lipids and cationic polymers in gene delivery. J. Control. Release 114, 100–109 (2006)
Yang, B. et al. High-throughput screening identifies small molecules that enhance the pharmacological effects of oligonucleotides. Nucleic Acids Res. 43, 1987–1996 (2015)
Furusawa, M., Nishimura, T., Yamaizumi, M. & Okada, Y. Injection of foreign substances into single cells by cell fusion. Nature 249, 449–450 (1974)
Helenius, A., Doxsey, S. & Mellman, I. Viruses as tools in drug delivery. Ann. NY Acad. Sci. 507, 1–6 (1987)
Daemen, T. et al. Virosomes for antigen and DNA delivery. Adv. Drug Deliv. Rev. 57, 451–463 (2005)
Montecalvo, A. et al. Mechanism of transfer of functional microRNAs between mouse dendritic cells via exosomes. Blood 119, 756–766 (2012)
El Andaloussi, S., Mäger, I., Breakefield, X. O. & Wood, M. J. A. Extracellular vesicles: biology and emerging therapeutic opportunities. Nat. Rev. Drug Discov. 12, 347–357 (2013)
He, W. et al. Discovery of siRNA lipid nanoparticles to transfect suspension leukemia cells and provide in vivo delivery capability. Mol. Ther. 22, 359–370 (2014)
Blanco, E., Shen, H. & Ferrari, M. Principles of nanoparticle design for overcoming biological barriers to drug delivery. Nat. Biotechnol. 33, 941–951 (2015)
Van Meirvenne, S. et al. Efficient genetic modification of murine dendritic cells by electroporation with mRNA. Cancer Gene Ther. 9, 787–797 (2002)
Wang, Y. et al. Poking cells for efficient vector-free intracellular delivery. Nat. Commun. 5, 4466 (2014)
Sharei, A. et al. A vector-free microfluidic platform for intracellular delivery. Proc. Natl Acad. Sci. USA 110, 2082–2087 (2013).In this paper, rapid mechanical deformation of cells through microfluidic constrictions was shown to achieve efficient intracellular delivery of a wide range of molecular cargo.
Schumann, K. et al. Generation of knock-in primary human T cells using Cas9 ribonucleoproteins. Proc. Natl Acad. Sci. USA 112, 10437–10442 (2015)
Schulz, I. Permeabilizing cells: some methods and applications for the study of intracellular processes. Methods Enzymol. 192, 280–300 (1990)
Hapala, I. Breaking the barrier: methods for reversible permeabilization of cellular membranes. Crit. Rev. Biotechnol. 17, 105–122 (1997)
Agarwal, J., Walsh, A. & Lee, R. C. Multimodal strategies for resuscitating injured cells. Ann. NY Acad. Sci. 1066, 295–309 (2005)
Gurtovenko, A. A., Anwar, J. & Vattulainen, I. Defect-mediated trafficking across cell membranes: insights from in silico modeling. Chem. Rev. 110, 6077–6103 (2010)
Bloom, M., Evans, E. & Mouritsen, O. G. Physical properties of the fluid lipid-bilayer component of cell membranes: a perspective. Q. Rev. Biophys. 24, 293–397 (1991)
Barber, M. A. A technic for the inoculation of bacteria and other substances into living cells. J. Infect. Dis. 8, 348–360 (1911).This is arguably amongst the first reports on intracellular delivery, demonstrating basic microinjection of isolated cells
Klein, T. M., Wolf, E. D., Wu, R. & Sanford, J. C. High-velocity microprojectiles for delivering nucleic-acids into living cells. Nature 327, 70–73 (1987)This report introduced the gene gun for intracellular delivery by exploiting biolistic particles.
Mcneil, P. L. Incorporation of macromolecules into living cells. Methods Cell Biol. 29, 153–173 (1988)
Clarke, M. S. F. & McNeil, P. L. Syringe loading introduces macromolecules into living mammalian cell cytosol. J. Cell Sci. 102, 533–541 (1992)
LaPlaca, M. C., Lee, V. M. Y. & Thibault, L. E. An in vitro model of traumatic neuronal injury: loading rate-dependent changes in acute cytosolic calcium and lactate dehydrogenase release. J. Neurotrauma 14, 355–368 (1997)
Hallow, D. M. et al. Shear-induced intracellular loading of cells with molecules by controlled microfluidics. Biotechnol. Bioeng. 99, 846–854 (2008)
Borle, A. B. & Snowdowne, K. W. Measurement of intracellular free calcium in monkey kidney cells with aequorin. Science 217, 252–254 (1982)
Groulx, N., Boudreault, F., Orlov, S. N. & Grygorczyk, R. Membrane reserves and hypotonic cell swelling. J. Membr. Biol. 214, 43–56 (2006)
Bischof, J. C. et al. Dynamics of cell membrane permeability changes at supraphysiological temperatures. Biophys. J. 68, 2608–2614 (1995)
He, X. M., Amin, A. A., Fowler, A. & Toner, M. Thermally induced introduction of trehalose into primary rat hepatocytes. Cell Preserv. Technol. 4, 178–187 (2006)
Weaver, J. C. & Chizmadzhev, Y. A. Theory of electroporation: a review. Bioelectrochem. Bioenerg. 41, 135–160 (1996)
Kandušer, M. & Miklavcˇicˇ, D. in Electrotechnologies for Extraction from Food Plants and Biomaterials (eds Vorobiev, E. & Lebovka, N.) Ch. 1, 1–37 (Springer, 2008)
Tsukakoshi, M., Kurata, S., Nomiya, Y., Ikawa, Y. & Kasuya, T. A novel method of DNA transfection by laser microbeam cell surgery. Appl. Phys. B 35, 135–140 (1984). This pioneering paper demonstrated DNA transfection of mammalian cells by laser optoporation.
Stevenson, D. J., Gunn-Moore, F. J., Campbell, P. & Dholakia, K. Single cell optical transfection. J. R. Soc. Interf. 7, 863–871 (2010)
Vogel, A., Noack, J., Huttman, G. & Paltauf, G. Mechanisms of femtosecond laser nanosurgery of cells and tissues. Appl. Phys. B 81, 1015–1047 (2005)This publication outlines a comprehensive framework covering the mechanisms of laser–membrane interactions.
Bischofberger, M., Iacovache, I. & van der Goot, F. G. Pathogenic pore-forming proteins: function and host response. Cell Host Microbe 12, 266–275 (2012)
Walev, I. et al. Delivery of proteins into living cells by reversible membrane permeabilization with streptolysin-O. Proc. Natl Acad. Sci. USA 98, 3185–3190 (2001)
Frenkel, N., Makky, A., Sudji, I. R., Wink, M. & Tanaka, M. Mechanistic investigation of interactions between steroidal saponin digitonin and cell membrane models. J. Phys. Chem. B 118, 14632–14639 (2014)
Cooper, S. T. & McNeil, P. L. Membrane repair: mechanisms and pathophysiology. Physiol. Rev. 95, 1205–1240 (2015)
Moe, A. M., Golding, A. E. & Bement, W. M. Cell healing: calcium, repair and regeneration. Semin. Cell Dev. Biol. 45, 18–23 (2015)
Jimenez, A. J. & Perez, F. Physico-chemical and biological considerations for membrane wound evolution and repair in animal cells. Semin. Cell Dev. Biol. 45, 2–9 (2015)
Boucher, E. & Mandato, C. A. Plasma membrane and cytoskeleton dynamics during single-cell wound healing. Biochim. Biophys. Acta 1853, 2649–2661 (2015)
Andrews, N. W., Corrotte, M. & Castro-Gomes, T. Above the fray: surface remodeling by secreted lysosomal enzymes leads to endocytosis-mediated plasma membrane repair. Semin. Cell Dev. Biol. 45, 10–17 (2015)
Steinhardt, R. A., Bi, G. & Alderton, J. M. Cell membrane resealing by a vesicular mechanism similar to neurotransmitter release. Science 263, 390–393 (1994). This groundbreaking paper alerted the research community to the active and responsive nature of cell plasma membrane repair, which was previously attributed to passive resealing.
Venslauskas, M. S. & Šatkauskas, S. Mechanisms of transfer of bioactive molecules through the cell membrane by electroporation. Eur. Biophys. J. 44, 277–289 (2015)
Neumann, E., Schaefer-Ridder, M., Wang, Y. & Hofschneider, P. H. Gene transfer into mouse lyoma cells by electroporation in high electric fields. EMBO J. 1, 841–845 (1982). This pioneering paper demonstrated DNA transfection of mammalian cells by electroporation.
Movahed, S. & Li, D. Q. Microfluidics cell electroporation. Microfluidics Nanofluidics 10, 703–734 (2011)
Geng, T. et al. Flow-through electroporation based on constant voltage for large-volume transfection of cells. J. Control. Release 144, 91–100 (2010)
Zhan, Y., Wang, J., Bao, N. & Lu, C. Electroporation of cells in microfluidic droplets. Anal. Chem. 81, 2027–2031 (2009)
Boukany, P. E. et al. Nanochannel electroporation delivers precise amounts of biomolecules into living cells. Nat. Nanotechnol. 6, 747–754 (2011)
McKnight, T. E. et al. Intracellular integration of synthetic nanostructures with viable cells for controlled biochemical manipulation. Nanotechnology 14, 551–556 (2003)This pioneering paper constitutes the first major implementation of nanoneedle arrays for DNA transfection.
Shalek, A. K. et al. Vertical silicon nanowires as a universal platform for delivering biomolecules into living cells. Proc. Natl Acad. Sci. USA 107, 1870–1875 (2010). This study expanded the use of nanoneedles for co-delivery of diverse biomolecules.
VanDersarl, J. J., Xu, A. M. & Melosh, N. A. Nanostraws for direct fluidic intracellular access. Nano Lett. 12, 3881–3886 (2012)
Xie, X. et al. Nanostraw-electroporation system for highly efficient intracellular delivery and transfection. ACS Nano 7, 4351–4358 (2013)
Shalek, A. K. et al. Nanowire-mediated delivery enables functional interrogation of primary immune cells: application to the analysis of chronic lymphocytic leukemia. Nano Lett. 12, 6498–6504 (2012)
Xu, A. M. et al. Quantification of nanowire penetration into living cells. Nat. Commun. 5, 3613 (2014)
Marmottant, P. & Hilgenfeldt, S. Controlled vesicle deformation and lysis by single oscillating bubbles. Nature 423, 153–156 (2003)
Liu, Y., Yan, J. & Prausnitz, M. R. Can ultrasound enable efficient intracellular uptake of molecules? A retrospective literature review and analysis. Ultrasound Med. Biol. 38, 876–888 (2012)
Fechheimer, M. et al. Transfection of mammalian cells with plasmid DNA by scrape loading and sonication loading. Proc. Natl Acad. Sci. USA 84, 8463–8467 (1987)This pioneering paper demonstrated DNA transfection of mammalian cells by ultrasound.
Prentice, P., Cuschierp, A., Dholakia, K., Prausnitz, M. & Campbell, P. Membrane disruption by optically controlled microbubble cavitation. Nat. Phys. 1, 107–110 (2005)
Ohl, C. D. et al. Sonoporation from jetting cavitation bubbles. Biophys. J. 91, 4285–4295 (2006)
Wu, Y. C. et al. Massively parallel delivery of large cargo into mammalian cells with light pulses. Nat. Methods 12, 439–444 (2015)
Cui, X., Dean, D., Ruggeri, Z. M. & Boland, T. Cell damage evaluation of thermal inkjet printed Chinese hamster ovary cells. Biotechnol. Bioeng. 106, 963–969 (2010)
Xiong, R. et al. Comparison of gold nanoparticle mediated photoporation: vapor nanobubbles outperform direct heating for delivering macromolecules in live cells. ACS Nano 8, 6288–6296 (2014)
Schomaker, M. et al. Characterization of nanoparticle mediated laser transfection by femtosecond laser pulses for applications in molecular medicine. J. Nanobiotechnol. 13:10, http://dx.doi.org/10.1186/s12951-014-0057-1 (2015)
Yao, C., Qu, X., Zhang, Z., Hüttmann, G. & Rahmanzadeh, R. Influence of laser parameters on nanoparticle-induced membrane permeabilization. J. Biomed. Opt. 14, 054034 (2009)
Time to deliver. Nat. Biotechnol. 32, 961 (2014)
Park, K. Facing the truth about nanotechnology in drug delivery. ACS Nano 7, 7442–7447 (2013)
Rajendran, L., Knölker, H. J. & Simons, K. Subcellular targeting strategies for drug design and delivery. Nat. Rev. Drug Discov. 9, 29–42 (2010)
Acknowledgements
This work was supported by the US National Institute of Health (R01GM101420-01A1). M.P.S. was supported by the Swiss NSF through the advanced postdoc mobility fellowship P300P3_151179. M.P.S. acknowledges support from a Keith Murdoch Fellowship via the American Australian Association, a Life Sciences Research Foundation Fellowship sponsored by Good Ventures, and a Broadnext10 Catalytic Steps funding gift from the Broad Institute. A.S. was supported by a Ragon Institute fellowship. We thank the following people for comments and constructive criticism: E. van Leen, D. Irvine, J. Voldman, S. Manalis, J. Weaver, J. Lieberman, R. Karnik, R. Lee, D. Mueller, S. Bhakdi, Y. Toyoda, Z. Maliga, H.-J. Lee, N. Yang, E. Lim, R. Sayde and K. Blagovic.
Author information
Authors and Affiliations
Contributions
R.L. and K.F.J. shaped ideas and provided guidance. M.P.S. constructed the figures. M.P.S. and A.S. wrote the manuscript (with assistance from X.D. and G.S.).
Corresponding authors
Ethics declarations
Competing interests
A.S., K.F.J. and R.L. have a financial interest in SQZ Biotech, a startup company focused on engineering immune cell function for cell based therapies (http://sqzbiotech.com/).
Additional information
Reviewer Information
Nature thanks L. Lee, M. Prausnitz and the other anonymous reviewer(s) for their contribution to the peer review of this work.
Rights and permissions
About this article
Cite this article
Stewart, M., Sharei, A., Ding, X. et al. In vitro and ex vivo strategies for intracellular delivery. Nature 538, 183–192 (2016). https://doi.org/10.1038/nature19764
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature19764
This article is cited by
-
Upconverting-photon quenching-mediated perforation influx as an intracellular delivery method using posAuNP@UCNPs nanocomposites for osteoarthritis treatment
Nano Convergence (2024)
-
In situ label-free X-ray imaging for visualizing the localization of nanomedicines and subcellular architecture in intact single cells
Nature Protocols (2024)
-
Electroactive nanoinjection platform for intracellular delivery and gene silencing
Journal of Nanobiotechnology (2023)
-
Biodegradable functionalized magnetite nanoparticles as binary-targeting carrier for breast carcinoma
BMC Chemistry (2023)
-
Engineering siRNA therapeutics: challenges and strategies
Journal of Nanobiotechnology (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.