Abstract
Diploidy is a fundamental genetic feature in mammals, in which haploid cells normally arise only as post-meiotic germ cells that serve to ensure a diploid genome upon fertilization. Gamete manipulation has yielded haploid embryonic stem (ES) cells from several mammalian species1,2,3,4,5,6, but haploid human ES cells have yet to be reported. Here we generated and analysed a collection of human parthenogenetic ES cell lines originating from haploid oocytes, leading to the successful isolation and maintenance of human ES cell lines with a normal haploid karyotype. Haploid human ES cells exhibited typical pluripotent stem cell characteristics, such as self-renewal capacity and a pluripotency-specific molecular signature. Moreover, we demonstrated the utility of these cells as a platform for loss-of-function genetic screening. Although haploid human ES cells resembled their diploid counterparts, they also displayed distinct properties including differential regulation of X chromosome inactivation and of genes involved in oxidative phosphorylation, alongside reduction in absolute gene expression levels and cell size. Surprisingly, we found that a haploid human genome is compatible not only with the undifferentiated pluripotent state, but also with differentiated somatic fates representing all three embryonic germ layers both in vitro and in vivo, despite a persistent dosage imbalance between the autosomes and X chromosome. We expect that haploid human ES cells will provide novel means for studying human functional genomics and development.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Leeb, M. & Wutz, A. Derivation of haploid embryonic stem cells from mouse embryos. Nature 479, 131–134 (2011)
Elling, U. et al. Forward and reverse genetics through derivation of haploid mouse embryonic stem cells. Cell Stem Cell 9, 563–574 (2011)
Yang, H. et al. Generation of genetically modified mice by oocyte injection of androgenetic haploid embryonic stem cells. Cell 149, 605–617 (2012)
Li, W. et al. Androgenetic haploid embryonic stem cells produce live transgenic mice. Nature 490, 407–411 (2012)
Li, W. et al. Genetic modification and screening in rat using haploid embryonic stem cells. Cell Stem Cell 14, 404–414 (2014)
Yang, H. et al. Generation of haploid embryonic stem cells from Macaca fascicularis monkey parthenotes. Cell Res. 23, 1187–1200 (2013)
Wutz, A. Haploid mouse embryonic stem cells: rapid genetic screening and germline transmission. Annu. Rev. Cell Dev. Biol. 30, 705–722 (2014)
Egli, D. et al. Impracticality of egg donor recruitment in the absence of compensation. Cell Stem Cell 9, 293–294 (2011)
Revazova, E. S. et al. Patient-specific stem cell lines derived from human parthenogenetic blastocysts. Cloning Stem Cells 9, 432–449 (2007)
Kim, K. et al. Recombination signatures distinguish embryonic stem cells derived by parthenogenesis and somatic cell nuclear transfer. Cell Stem Cell 1, 346–352 (2007)
Paull, D. et al. Nuclear genome transfer in human oocytes eliminates mitochondrial DNA variants. Nature 493, 632–637 (2013)
Tarkowski, A. K., Witkowska, A. & Nowicka, J. Experimental partheonogenesis in the mouse. Nature 226, 162–165 (1970)
Kaufman, M. H., Robertson, E. J., Handyside, A. H. & Evans, M. J. Establishment of pluripotential cell lines from haploid mouse embryos. J. Embryol. Exp. Morphol. 73, 249–261 (1983)
Leeb, M. et al. Germline potential of parthenogenetic haploid mouse embryonic stem cells. Development 139, 3301–3305 (2012)
Takahashi, S. et al. Induction of the G2/M transition stabilizes haploid embryonic stem cells. Development 141, 3842–3847 (2014)
Ben-David, U., Nudel, N. & Benvenisty, N. Immunologic and chemical targeting of the tight-junction protein Claudin-6 eliminates tumorigenic human pluripotent stem cells. Nat. Commun. 4, 1992 (2013)
Doench, J. G. et al. Optimized sgRNA design to maximize activity and minimize off-target effects of CRISPR–Cas9. Nature Biotechnol. 34, 184–191 (2016)
Silva, S. S., Rowntree, R. K., Mekhoubad, S. & Lee, J. T. X-chromosome inactivation and epigenetic fluidity in human embryonic stem cells. Proc. Natl Acad. Sci. USA 105, 4820–4825 (2008)
Bruck, T., Yanuka, O. & Benvenisty, N. Human pluripotent stem cells with distinct X inactivation status show molecular and cellular differences controlled by the X–linked ELK-1 gene. Cell Rep . 4, 262–270 (2013)
Lovén, J. et al. Revisiting global gene expression analysis. Cell 151, 476–482 (2012)
Biancotti, J. C. et al. The in vitro survival of human monosomies and trisomies as embryonic stem cells. Stem Cell Res . 9, 218–224 (2012)
Otto, S. P. & Jarne, P. Evolution. Haploids–hapless or happening? Science 292, 2441–2443 (2001)
Noggle, S. et al. Human oocytes reprogram somatic cells to a pluripotent state. Nature 478, 70–75 (2011)
Chen, A. E. et al. Optimal timing of inner cell mass isolation increases the efficiency of human embryonic stem cell derivation and allows generation of sibling cell lines. Cell Stem Cell 4, 103–106 (2009)
Rao, P. H., Nandula, S. V. & Murty, V. V. Molecular cytogenetic applications in analysis of the cancer genome. Methods Mol. Biol. 383, 165–185 (2007)
Cadiñanos, J. & Bradley, A. Generation of an inducible and optimized piggyBac transposon system. Nucleic Acids Res. 35, e87 (2007)
Wang, W. et al. Chromosomal transposition of PiggyBac in mouse embryonic stem cells. Proc. Natl Acad. Sci. USA 105, 9290–9295 (2008)
Chen, L. et al. Transposon activation mutagenesis as a screening tool for identifying resistance to cancer therapeutics. BMC Cancer 13, 93 (2013)
Johannesson, B. et al. Comparable frequencies of coding mutations and loss of imprinting in human pluripotent cells derived by nuclear transfer and defined factors. Cell Stem Cell 15, 634–642 (2014)
Lonsdale, J. et al.The Genotype-Tissue Expression (GTEx) project. Nature Genet. 45, 580–585 (2013)
Yamada, M. et al. Human oocytes reprogram adult somatic nuclei of a type 1 diabetic to diploid pluripotent stem cells. Nature 510, 533–536 (2014)
Wanet, A. et al. Mitochondrial remodeling in hepatic differentiation and dedifferentiation. Int. J. Biochem. Cell Biol. 54, 174–185 (2014)
Kim, D.-S. et al. Robust enhancement of neural differentiation from human ES and iPS cells regardless of their innate difference in differentiation propensity. Stem Cell Rev. 6, 270–281 (2010)
Stelzer, Y., Sagi, I. & Benvenisty, N. Involvement of parental imprinting in the antisense regulation of onco-miR-372-373. Nat. Commun. 4, 2724 (2013)
Wang, L. et al. Differentiation of hypothalamic-like neurons from human pluripotent stem cells. J. Clin. Invest. 125, 796–808 (2015)
Chambers, S. M. et al. Highly efficient neural conversion of human ES and iPS cells by dual inhibition of SMAD signaling. Nature Biotechnol. 27, 275–280 (2009)
Lian, X. et al. Directed cardiomyocyte differentiation from human pluripotent stem cells by modulating Wnt/β-catenin signaling under fully defined conditions. Nature Protocols 8, 162–175 (2013)
Hua, H. et al. iPSC-derived β cells model diabetes due to glucokinase deficiency. J. Clin. Invest. 123, 3146–3153 (2013)
Pagliuca, F. W. et al. Generation of functional human pancreatic β cells in vitro. Cell 159, 428–439 (2014)
Rezania, A. et al. Reversal of diabetes with insulin-producing cells derived in vitro from human pluripotent stem cells. Nature Biotechnol. 32, 1121–1133 (2014)
Cowan, C. A., Atienza, J., Melton, D. A. & Eggan, K. Nuclear reprogramming of somatic cells after fusion with human embryonic stem cells. Science 309, 1369–1373 (2005)
Acknowledgements
We thank all members of the Benvenisty and Egli laboratories for input and support. We thank Y. Avior and W. Breuer for their assistance with experimental procedures. I.S. is supported by the Adams Fellowships Program for Doctoral Students, G.C. is supported by the A*STAR International Fellowship, U.W. is a Clore Fellow, D.E. is a NYSCF-Robertson Investigator, and N.B. is the Herbert Cohn Chair in Cancer Research. This work was partially supported by The Rosetrees Trust and by The Azrieli Foundation (to N.B.), by the Russell Berrie Foundation Program in Cellular Therapies of Diabetes, by the New York State Stem Cell Science (NYSTEM) IIRP Award number C026184, and by the New York Stem Cell Foundation (to D.E.).
Author information
Authors and Affiliations
Contributions
I.S., D.E. and N.B. designed the study and wrote the manuscript with input from all authors. I.S. isolated and characterized haploid human ES cell lines, performed differentiation experiments and analysed the data. G.C. developed and performed the centromere quantification analysis and carried out neuronal differentiation. T.G.-L. assisted in tissue culture and performed karyotype analyses and tissue sectioning. I.S., M.P., U.W. and O.Y. were involved in the genetic screening. M.P. and U.W. assisted with teratoma assays. L.S. assisted with pancreatic differentiation. M.V.S. was involved in all aspects of oocyte donation and research. D.E. derived human pES cell lines from haploid oocytes. D.E. and N.B. supervised the study.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Extended data figures and tables
Extended Data Figure 1 Derivation of haploid human ES cell lines h-pES10 and h-pES12.
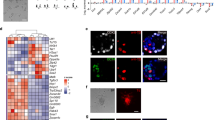
a, Left panel, diploidization rate model for a haploid egg with a theoretical diploidization probability of 10%, overlaid with an exponential decay fit (red curve, see Supplementary Notes). Approximated cell cycle numbers for different ES cell line derivation stages are indicated. Right panel, diploidization dynamics of h-pES10 over seven passages by flow cytometry, overlaid with an exponential fit to the data (red curve). Bars indicate the mean across biological replicates. b, Establishment of a haploid-enriched human ES cell line from pES12 cells after repeated sorting and enrichment of 1c cells using Hoechst 33342 staining. Top to bottom, DNA content profiles of unsorted diploid cells, partially purified haploid cells at the third sort, and mostly-purified haploid cells at the fifth sort. c, chromosomal copies. c, Karyotypes and haploid metaphase percentage over the course of enrichment and passaging. d, e, DNA FISH (d) and centromere protein immunofluorescence staining (e) in haploid-enriched pES12 cells (upper panels) and unsorted diploid pES10 cells (lower panels). Magnified insets show representative haploid and diploid nuclei with single or double hybridization signals (d) and 23 or 46 centromeres (e), respectively. Scale bars, 10 μm. f, SNP array-based CNV analysis comparing haploid pES10 and pES12 cells with their unsorted diploid counterparts (logarithmic scale).
Extended Data Figure 2 Determination of ploidy at single-cell level by quantification of centromere foci.
a, The counted number of centromeres correlates with ploidy. 1n, haploid-enriched pES10 cells grown for 4 passages after the fourth sort (n = 33; 76% haploids by this assay); 2n, unsorted diploid pES10 cells (n = 34); 3n, soPS2 cells23 (n = 27); 4n, Hybrid1 cells41 (n = 27). Black horizontal lines indicate mean ± s.e.m. and dashed lines mark expected chromosome numbers. b, Quantification of haploid and diploid cells by DNA FISH in the haploid-enriched (n = 152; 73% haploids by this assay) and diploid (n = 135) cells in a. c, DNA content profile of the haploid-enriched cells in a (73% haploids by this assay). c, chromosomal copies. d, e, Co-staining of centromeres and either phospho-histone 3 (pH3, Ser10) (d) or 5-ethynyl-2′-deoxyuridine (EdU) (e) for distinguishing between different stages of interphase in haploid pES12 cells. In blue, DNA staining. Scale bar, 5 μm. f, Quantification of centromere counts in the different cell cycle stages shown in d and e. n indicated in parenthesis. Black horizontal lines indicate mean ± s.e.m. See Supplementary Notes for details.
Extended Data Figure 3 Pluripotent stem cell markers in haploid human ES cells.
Co-staining of pluripotency markers NANOG, OCT4, SOX2, SSEA4 and TRA-1-60 (red), centromeres (green) and DNA (blue) in h-pES10 and h-pES12 at colony resolution (upper panels; scale bars, 50 μm) and single-cell resolution (lower panels; scale bars, 10 μm). Magnified insets show representative haploid cells with 23 centromeres.
Extended Data Figure 4 Analysis of parental imprinting and gene trap mutagenesis in haploid human parthenogenetic ES cells.
a, b, Clustering analysis of diploid in vitro fertilization (IVF) ES cells and G1-sorted haploid and diploid parthenogenetic ES (pES) cells by expression levels of imprinted genes (n = 75, see Supplementary Table 4) (a) and DNA methylation levels at imprinted differentially methylated regions (iDMRs, n = 35)29 (b). The symbols (1) and (2) indicate biological replicates. c, Relative mean expression levels ± s.e.m. of representative paternally expressed imprinted genes across seven chromosomes in the samples shown in a (RPKM ratios). d, Mean DNA methylation levels ± s.e.m. at representative paternally methylated and maternally methylated iDMRs (typically intermediately methylated in bi-parental control cells, and respectively hypomethylated and hypermethylated in parthenogenetic cells) in the samples shown in b. β values range from complete hypomethylation (0) to complete hypermethylation (1). e, Schematic outline of the piggyBac gene trap system. The gene trap vector26 is flanked by piggyBac inverted terminal repeats (ITRs) and FRT flox sites, and carries a 5′ splice acceptor (SA), an internal ribosome entry site (IRES) element followed by a promoterless puromycin resistance gene (PuroΔtk) and a 3′ poly(A) signal (pA). In the presence of the PiggyBac transposase (encoded on a separate plasmid27, not shown), the gene trap vector undergoes random transposition into the genome. Insertion into a transcriptionally active gene results in truncation of the endogenous transcript and introduction of resistance to puromycin.
Extended Data Figure 5 Comparative analyses of isogenic haploid and diploid human ES cells.
a, Sorting purity of haploid and diploid ES cells in G1. b, Log-scaled volcano plots of relative differential gene expression between haploid and diploid human ES cells, divided into panels by all genes (top), autosomal genes (middle) and X chromosomal genes (bottom). Q, false discovery rate (FDR). Significantly downregulated and upregulated genes (greater than twofold change, Q < 0.05) in haploid cells are marked in red and blue, respectively, and their totals are indicated to the right. Note that XIST is the most downregulated transcript in haploid cells. c, Smoothed distributions of the 1n/2n gene expression ratios for all expressed genes, all expressed autosomal genes and all expressed X chromosomal genes (expression threshold, mean RPKM >0.1). d, Genome-wide moving median plot of the gene expression ratio between haploid and diploid pES10 cells in G1 by expression microarray analysis (window size = 100 genes). e, f, Model for genome-wide autosomal gene expression level reduction in haploid human ES cell as inferred by differential X chromosome inactivation status. e, DNA content, RNA expression levels relative to total RNA and presumed equality of absolute X chromosomal gene dosage in haploid (Xa) and diploid (XaXi) human ES cells, enable the estimation of total RNA levels per haploid cell. Xa and Xi denote active (blue) and inactive (red) X chromosomes, respectively. A, autosomes; X, X chromosome; R, total RNA. f, Schematic genome-wide representation of relative and absolute RNA levels in the cells shown in e. g, Average diameter and calculated surface area and volume of G1-sorted haploid and diploid ES cells. Error bars represent s.d. *P < 0.01 (two-tailed unpaired Student’s t-test). h, i, Functional annotation enrichment analysis for relatively downregulated genes and differentially methylated regions (DMRs) (h), as well as relatively upregulated genes (i) in haploid ES cells compared with diploid ES cells.
Extended Data Figure 6 EB differentiation of haploid human ES cells.
a, Representative image of plated cells dissociated from h-pES12-derived 21-day EBs. Karyotype is shown in Fig. 4b. Scale bar, 100 μm. b, DNA content profiles of dissociated EBs derived from haploid-enriched and diploid pES12 cells. c: chromosomal copies. c, Expression levels (RPKM) of tissue- and pluripotency-specific genes in undifferentiated (ES) and differentiated (EB) G1-sorted haploid (1n) pES10 cells.
Extended Data Figure 7 Directed differentiation of haploid human ES cells.
a, b, Flow cytometry analysis with co-staining of DNA and NCAM1 in h-pES10 cells following neural differentiation. a, Gating for NCAM1-positive cells (right) based on a negative secondary antibody stained control sample (left). b, DNA content profiles of the entire cell population (left) and NCAM1-positive cells (right). c, chromosomal copies. c, d, Expression levels of neural- and pluripotency-specific genes in haploid G1-sorted ES cells and NPCs. e, XIST expression levels in haploid and diploid pES10-derived EBs and NPCs. f, TUJ1 staining in h-pES12-derived neurons. Scale bar, 100 μm. g, DNA FISH in the neurons shown in f. Magnified insets show representative haploid and diploid nuclei with single and double hybridization signals, respectively. Scale bar, 10 μm. h, TNNT2 staining in G1-sorted haploid pES12-derived cardiomyocytes. Scale bar, 10 μm. i, j, Flow cytometry analysis with co-staining of DNA and PDX1 in h-pES10 cells following pancreatic differentiation. i, Gating for PDX1-positive cells (right) based on a negative secondary antibody stained control sample (left). j, DNA content profile of the entire cell population (related to Fig. 4k). c, chromosomal copies.
Extended Data Figure 8 In vivo differentiation of haploid human ES cells.
a, Haematoxylin and eosin histological sections of teratomas derived from h-pES10 and h-pES12. Scale bar, 50 μm. b, TUJ1 (ectoderm), α-SMA (mesoderm), AFP (endoderm) and OCT4 (pluripotency) staining in an h-pES10-derived teratoma. DNA staining is shown in blue. Note the absence of nuclear OCT4 staining. Scale bars, 100 μm.
Supplementary information
Supplementary Information
This file contains Supplementary Tables 1-4, a Supplementary Discussion, Supplementary Notes and additional references. (PDF 290 kb)
Spontaneous beating of h-pES12-derived cardiomyocytes
This file shows spontaneous beating of h-pES12-derived cardiomyocytes. (MOV 9789 kb)
Source data
Rights and permissions
About this article
Cite this article
Sagi, I., Chia, G., Golan-Lev, T. et al. Derivation and differentiation of haploid human embryonic stem cells. Nature 532, 107–111 (2016). https://doi.org/10.1038/nature17408
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature17408
This article is cited by
-
Functional annotation of variants of the BRCA2 gene via locally haploid human pluripotent stem cells
Nature Biomedical Engineering (2023)
-
Epigenetic integrity of paternal imprints enhances the developmental potential of androgenetic haploid embryonic stem cells
Protein & Cell (2022)
-
Identifying regulators of parental imprinting by CRISPR/Cas9 screening in haploid human embryonic stem cells
Nature Communications (2021)
-
Generation of developmentally competent oocytes and fertile mice from parthenogenetic embryonic stem cells
Protein & Cell (2021)
-
In vitro expansion of human sperm through nuclear transfer
Cell Research (2020)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.