Abstract
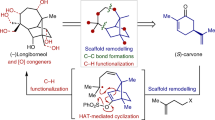
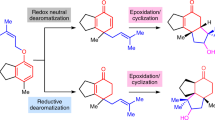
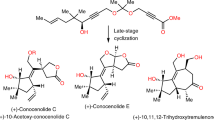
Phorbol, the flagship member of the tigliane diterpene family, has been known for over 80 years and has attracted attention from many chemists and biologists owing to its intriguing chemical structure and the medicinal potential of phorbol esters1. Access to useful quantities of phorbol and related analogues has relied on isolation from natural sources and semisynthesis. Despite efforts spanning 40 years, chemical synthesis has been unable to compete with these strategies, owing to its complexity and unusual placement of oxygen atoms. Purely synthetic enantiopure phorbol has remained elusive, and biological synthesis has not led to even the simplest members of this terpene family. Recently, the chemical syntheses of eudesmanes2, germacrenes3, taxanes4,5 and ingenanes6,7,8 have all benefited from a strategy inspired by the logic of two-phase terpene biosynthesis in which powerful C–C bond constructions and C–H bond oxidations go hand in hand. Here we implement a two-phase terpene synthesis strategy to achieve enantiospecific total synthesis of (+)-phorbol in only 19 steps from the abundant monoterpene (+)-3-carene. The purpose of this synthesis route is not to displace isolation or semisynthesis as a means of generating the natural product per se, but rather to enable access to analogues containing unique placements of oxygen atoms that are otherwise inaccessible.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Wang, H.-B., Wang, X.-Y., Liu, L.-P., Qin, G.-W. & Kang, T.-G. Tigliane diterpenoids from the euphorbiaceae and thymelaeaceae families. Chem. Rev. 115, 2975–3011 (2015)
Chen, K. & Baran, P. S. Total synthesis of eudesmane terpenes by site-selective C–H oxidations. Nature 459, 824–828 (2009)
Foo, K. et al. Scalable, enantioselective synthesis of germacrenes and related sesquiterpenes inspired by terpene cyclase phase logic. Angew. Chem. Int. Ed. 51, 11491–11495 (2012)
Mendoza, A., Ishihara, Y. & Baran, P. S. Scalable enantioselective total synthesis of taxanes. Nature Chem. 4, 21–25 (2012)
Wilde, N. C., Isomura, M., Mendoza, A. & Baran, P. S. Two-phase synthesis of (−)-taxuyunnanine D. J. Am. Chem. Soc. 136, 4909–4912 (2014)
Jørgensen, L. et al. 14-step synthesis of (–)-ingenol from (+)-3-carene. Science 341, 878–882 (2013)
McKerrall, S. J., Jørgensen, L., Kuttruff, C. A., Ungeheuer, F. & Baran, P. S. Development of a concise synthesis of (–)-ingenol. J. Am. Chem. Soc. 136, 5799–5810 (2014)
Jin, Y. et al. C–H oxidation of ingenanes enables potent and selective protein kinase C isoform activation. Angew. Chem. Int. Ed. 54, 14044–14048 (2015)
Isakov, N. & Altman, A. Regulation of immune system cell functions by protein kinase C. Front. Immunol. 4, 384 (2013)
McKernan, L. N., Momjian, D. & Kulkosky, J. Protein kinase C: one pathway towards the eradication of latent HIV-1 reservoirs. Adv. Virol. 2012, 805347 (2012)
Mackay, H. J. & Twelves, C. J. Targeting the protein kinase C family: are we there yet? Nature Rev. Cancer 7, 554–562 (2007); corrigendum 8, doi:10.1038/nrc2350 (2008)
Wender, P. A. et al. Studies on tumor promoters. 8. The synthesis of phorbol. J. Am. Chem. Soc. 111, 8957–8958 (1989)
Wender, P. A., Lee, H. Y., Wilhelm, R. S. & Williams, P. D. Studies on tumor promoters. 7. The synthesis of a potentially general precursor of the tiglianes, daphnanes, and ingenanes. J. Am. Chem. Soc. 111, 8954–8957 (1989)
Wender, P. A., Rice, K. D. & Schnute, M. E. The first formal asymmetric synthesis of phorbol. J. Am. Chem. Soc. 119, 7897–7898 (1997)
Wender, P. A. & McDonald, F. E. Studies on tumor promoters. 9. A second-generation synthesis of phorbol. J. Am. Chem. Soc. 112, 4956–4958 (1990)
Lee, K. & Cha, J. K. Formal synthesis of (+)-phorbol. J. Am. Chem. Soc. 123, 5590–5591 (2001)
Sugita, K., Shigeno, K., Neville, C. F., Sasai, H. & Shibasaki, M. Synthetic studies towards phorbols: synthesis of B or C ring substituted phorbol skeletons in the naturally occurring form. Synlett 1994, 325–329 (1994)
Sugita, K., Neville, C. F., Sodeoka, M., Sasai, H. & Shibasaki, M. Stereocontrolled syntheses of phorbol analogs and evaluation of their binding affinity to PKC. Tetrahedr. Lett. 36, 1067–1070 (1995)
Newhouse, T. & Baran, P. S. If C–H bonds could talk: selective C–H bond oxidation. Angew. Chem. Int. Ed. 50, 3362–3374 (2011)
Zou, L. et al. Enhanced reactivity in dioxirane C–H oxidations via strain release: a computational and experimental study. J. Org. Chem. 78, 4037–4048 (2013)
Michaudel, Q. et al. Improving physical properties via C–H oxidation: chemical and enzymatic approaches. Angew. Chem. Int. Ed. 53, 12091–12096 (2014)
McCormick, J. P. & Barton, D. L. Synthetic applications of metal halides. Conversion of cyclopropylmethanols into homoallylic halides. J. Org. Chem. 45, 2566–2570 (1980)
Miyoshi, N., Takeuchi, S. & Ohgo, Y. A facile synthesis of 2,3-dihydroxyketones from 1,2-diketones and aldehydes using samarium diiodide. Chem. Lett. 22, 959–962 (1993)
Krohn, K., Frese, P. & Flörke, U. Biomimetic synthesis of the racemic angucyclinones of the aquayamycin and WP 3688-2 Types. Chemistry 6, 3887–3896 (2000)
Bartsch, H. & Hecker, E. Zur chemie des phorbols, XIII. Über eine acyloin-umlagerung des 12-desoxy-12-oxo-phorbol-13.20-diacetats. Liebigs Ann. Chem. 725, 142–153 (1969)
Salmond, W. G., Barta, M. A. & Havens, J. L. Allylic oxidation with 3,5-dimethylpyrazole. Chromium trioxide complex. Steroidal Δ5-7-ketones. J. Org. Chem. 43, 2057–2059 (1978)
Sha, C.-K. & Huang, S.-J. Synthesis of β-substituted α-iodocycloalkenones. Tetrahedr. Lett. 36, 6927–6928 (1995)
Stille, J. K. The palladium-catalyzed cross-coupling reactions of organotin reagents with organic electrophiles. Angew. Chem. Int. Ed. Engl. 25, 508–524 (1986)
Nicolaou, K. C. & Sorensen, E. J. Classics in Total Synthesis: Targets, Strategies, Methods 821 (Wiley, 1996)
Gutekunst, W. R. & Baran, P. S. C–H functionalization logic in total synthesis. Chem. Soc. Rev. 40, 1976–1991 (2011)
Acknowledgements
This work was supported by LEO Pharma, the Uehara Memorial Foundation (postdoctoral fellowship to S.K.) and the National Institute of General Medical Sciences Grant GM-097444. We are especially grateful to S. Natarajan of KemXtree and his team for providing ample quantities of compound 5. We thank D.-H. Huang and L. Pasternack for assistance with NMR spectroscopy, and A. L. Rheingold and C. E. Moore for X-ray crystallographic analysis.
Author information
Authors and Affiliations
Contributions
S.K. and P.S.B. conceived this work; J.F. provided compound 5; S.K., H.C. and P.S.B. designed the experiments and analysed that data; S.K. and H.C. conducted the experiments; S.K. performed the molecular mechanics calculations; and S.K. and P.S.B. wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Additional information
Metrical parameters for the structures of 12 and 19 are available free of charge from the Cambridge Crystallographic Data Centre (CCDC) under reference numbers 1434376 and 1434377.
Supplementary information
Supplementary Information
This file contains Supplementary Text and Data – see contents page for details. (PDF 4624 kb)
Rights and permissions
About this article
Cite this article
Kawamura, S., Chu, H., Felding, J. et al. Nineteen-step total synthesis of (+)-phorbol. Nature 532, 90–93 (2016). https://doi.org/10.1038/nature17153
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature17153
This article is cited by
-
Catalytic 4-exo-dig carbocyclization for the construction of furan-fused cyclobutanones and synthetic applications
Nature Communications (2023)
-
Synthesis of nimbolide and its analogues and their application as poly(ADP-ribose) polymerase-1 trapping inducers
Nature Synthesis (2023)
-
Practical synthesis of the therapeutic leads tigilanol tiglate and its analogues
Nature Chemistry (2022)
-
A general strategy for diversifying complex natural products to polycyclic scaffolds with medium-sized rings
Nature Communications (2019)
-
Stabilizing short-lived Schiff base derivatives of 5-aminouracils that activate mucosal-associated invariant T cells
Nature Communications (2017)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.