Abstract
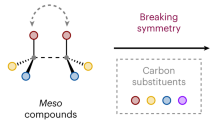
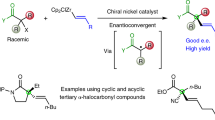
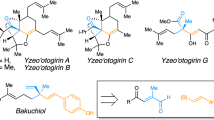
Quaternary carbon stereocentres—carbon atoms to which four distinct carbon substituents are attached—are common features of molecules found in nature. However, before recent advances in chemical catalysis, there were few methods of constructing single stereoisomers of this important structural motif. Here we discuss the many catalytic enantioselective reactions developed during the past decade for the synthesis of single stereoisomers of such organic molecules. This progress now makes it possible to incorporate quaternary stereocentres selectively in many organic molecules that are useful in medicine, agriculture and potentially other areas such as flavouring, fragrances and materials.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout










Similar content being viewed by others
References
Bartholow, M. Top 200 drugs of 2011. Pharm. Times 48–51 (10 July 2012)
Ding, H. X., Liu, K. K.-C., Sakya, S. M., Flick, A. C. & O’Donnell, C. J. Synthetic approaches to the 2011 new drugs. Bioorg. Med. Chem. 21, 2795–2825 (2013)
Christoffers J., Baro A., eds. Quaternary Stereocenters — Challenges and Solutions for Organic Synthesis (Wiley-VCH, 2005)
Hong, A. Y. & Stoltz, B. M. The construction of all-carbon quaternary stereocenters by use of Pd-catalyzed asymmetric allylic alkylation reactions in total synthesis. Eur. J. Org. Chem. 2745–2759 (2013)
Douglas, C. J. & Overman, L. E. Catalytic asymmetric synthesis of all-carbon quaternary stereocenters. Proc. Natl Acad. Sci. USA 101, 5363–5367 (2004)
Wilson, R. M., Jen, W. S. & MacMillan, D. W. C. Enantioselective organocatalytic intramolecular Diels–Alder reactions. The asymmetric synthesis of solanapyrone D. J. Am. Chem. Soc. 127, 11616–11617 (2005)
Jones, S. B., Simmons, B., Mastracchio, A. & MacMillan, D. W. C. Collective synthesis of natural products by means of organocascade catalysis. Nature 475, 183–188 (2011)A highlight of the use of iminium activation and cascade catalysis in the synthesis of alkaloid natural products.
Laforteza, B. N., Pickworth, M. & MacMillan, D. W. C. Enantioselective total synthesis of (–)-minovincine in nine chemical steps: an approach to ketone activation in cascade catalysis. Angew. Chem. Int. Edn 52, 11269–11272 (2013)
Liu, Y., Nappi, M., Arceo, E., Vera, S. & Melchiorre, P. Asymmetric catalysis of Diels–Alder reactions with in situ generated heterocyclic ortho-quinodimethanes. J. Am. Chem. Soc. 133, 15212–15218 (2011)
Tan, B., Hernández-Torres, G. & Barbas, C. F., III Highly efficient hydrogen-bonding catalysis of the Diels–Alder reaction of 3-vinylindoles and methyleneindolinones provides carbazolespirooxindole skeletons. J. Am. Chem. Soc. 133, 12354–12357 (2011)
Shimizu, Y., Shi, S.-L., Usuda, H., Kanai, M. & Shibasaki, M. Catalytic asymmetric total synthesis of ent-hyperforin. Angew. Chem. Int. Edn 49, 1103–1106 (2010)
Snyder, S. A. & Corey, E. J. Concise total syntheses of palominol, dolabellatrienone, β-araneosene, and isoedunol via an enantioselective Diels–Alder macrobicyclization. J. Am. Chem. Soc. 128, 740–742 (2006)
Balskus, E. P. & Jacobsen, E. N. Asymmetric catalysis of the transannular Diels–Alder reaction. Science 317, 1736–1740 (2007)
Lian, Y. & Davies, H. M. L. Rhodium-catalyzed [3 + 2] annulation of indoles. J. Am. Chem. Soc. 132, 440–441 (2010)
Trost, B. M., Silverman, S. M. & Stambuli, J. P. Development of an asymmetric trimethylenemethane cycloaddition reaction: application in the enantioselective synthesis of highly substituted carbocycles. J. Am. Chem. Soc. 133, 19483–19497 (2011)
Trost, B. M., Bringley, D. A., Zhang, T. & Cramer, N. Rapid access to spirocyclic oxindole alkaloids: application of the asymmetric palladium-catalyzed [3 + 2] trimethylenemethane cycloaddition. J. Am. Chem. Soc. 135, 16720–16735 (2013)An informative discussion of development of the TMM reaction for the synthesis of spirocyclic oxindoles.
Ohmatsu, K., Imagawa, N. & Ooi, T. Ligand-enabled multiple absolute stereocontrol in metal-catalysed cycloaddition for construction of contiguous all-carbon quaternary stereocentres. Nature Chem. 6, 47–51 (2014)
Chen, X.-H., Wei, Q., Luo, S.-W., Xiao, H. & Gong, L.-Z. Organocatalytic synthesis of spiro[pyrrolidin-3,3′-oxindoles] with high enantiopurity and structural diversity. J. Am. Chem. Soc. 131, 13819–13825 (2009)
Martínez, A., Webber, M. J., Müller, S. & List, B. Versatile access to chiral indolines by catalytic asymmetric Fischer indolization. Angew. Chem. Int. Edn 52, 9486–9490 (2013)
Pellissier, H. Recent developments in asymmetric cyclopropanation. Tetrahedron 64, 7041–7095 (2008)
Schwartz, B. D., Denton, J. R., Lian, Y., Davies, H. M. L. & Williams, C. M. Asymmetric [4 + 3] cycloadditions between vinylcarbenoids and dienes: application to the total synthesis of the natural product (–)-5-epi-vibsanin E. J. Am. Chem. Soc. 131, 8329–8332 (2009)
Ishibashi, H., Ishihara, K. & Yamamoto, H. A new artificial cyclase for polyprenoids: enantioselective total synthesis of (–)-chromazonarol, (+)-8-epi-puupehedione, and (–)-11′-deoxytaondiol methyl ether. J. Am. Chem. Soc. 126, 11122–11123 (2004)
Surendra, K. & Corey, E. J. Highly enantioselective proton-initiated polycyclization of polyenes. J. Am. Chem. Soc. 134, 11992–11994 (2012)
Surendra, K., Rajendar, G. & Corey, E. J. Useful catalytic enantioselective cationic double annulation reactions initiated at an internal π-bond: method and applications. J. Am. Chem. Soc. 136, 642–645 (2014)
Schafroth, M. A., Sarlah, D., Krautwald, S. & Carreira, E. M. Iridium-catalyzed enantioselective polyene cyclization. J. Am. Chem. Soc. 134, 20276–20278 (2012)A potentially broadly applicable approach for orchestrating enantioselective polyene cyclizations.
Jeker, O. F., Kravina, A. G. & Carreira, E. M. Total synthesis of (+)-asperolide C by iridium-catalyzed enantioselective polyene cyclization. Angew. Chem. Int. Edn 52, 12166–12169 (2013)
Sethofer, S. G., Mayer, T. & Toste, F. D. Gold(I)-catalyzed enantioselective polycyclization reactions. J. Am. Chem. Soc. 132, 8276–8277 (2010)
Brazeau, J.-F., Zhang, S., Colomer, I., Corkey, B. K. & Toste, F. D. Enantioselective cyclizations of silyloxyenynes catalyzed by cationic metal phosphine complexes. J. Am. Chem. Soc. 134, 2742–2749 (2012)
Corkey, B. K. & Toste, F. D. Catalytic enantioselective Conia-ene reaction. J. Am. Chem. Soc. 127, 17168–17169 (2005)
Shibata, T., Tahara, Y.-K., Tamura, K. & Endo, K. Enantioselective syntheses of various chiral multicyclic compounds with quaternary carbon stereocenters by catalytic intramolecular cycloaddition. J. Am. Chem. Soc. 130, 3451–3457 (2008)
Knowles, R. R., Lin, S. & Jacobsen, E. N. Enantioselective thiourea-catalyzed cationic polycyclizations. J. Am. Chem. Soc. 132, 5030–5032 (2010)
Rendler, S. & MacMillan, D. W. C. Enantioselective polyene cyclization via organo-SOMO catalysis. J. Am. Chem. Soc. 132, 5027–5029 (2010)
Dounay, A. B., Humphreys, P. G., Overman, L. E. & Wrobleski, A. D. Total synthesis of the strychnos alkaloid (+)-minfiensine: tandem enantioselective intramolecular Heck–iminium ion cyclization. J. Am. Chem. Soc. 130, 5368–5377 (2008)
Watson, M. P. & Jacobsen, E. N. Asymmetric intramolecular arylcyanation of unactivated olefins via C–CN bond activation. J. Am. Chem. Soc. 130, 12594–12595 (2008)
Nakao, Y. et al. Intramolecular arylcyanation of alkenes catalyzed by nickel/AlMe2Cl. J. Am. Chem. Soc. 130, 12874–12875 (2008)
García-Fortanet, J., Kessler, F. & Buchwald, S. L. Palladium-catalyzed asymmetric dearomatization of naphthalene derivatives. J. Am. Chem. Soc. 131, 6676–6677 (2009)
Xu, T., Ko, H. M., Savage, N. A. & Dong, G. Highly enantioselective Rh-catalyzed carboacylation of olefins: efficient syntheses of chiral poly-fused rings. J. Am. Chem. Soc. 134, 20005–20008 (2012)
Mei, T.-S., Patel, H. H. & Sigman, M. S. Enantioselective construction of remote quaternary stereocentres. Nature 508, 340–344 (2014)A pioneering method for constructing remote stereocentres in acyclic substrates by enantioselective Heck reactions.
Vuagnoux-d’Augustin, M. & Alexakis, A. Copper-catalyzed asymmetric conjugate addition of trialkylaluminium reagents to trisubstituted enones: construction of chiral quaternary centers. Chem. Eur. J. 13, 9647–9662 (2007)
Müller, D. & Alexakis, A. Formation of quaternary stereogenic centers by copper-catalyzed asymmetric conjugate addition reactions of alkenylaluminums to trisubstituted enones. Chem. Eur. J. 19, 15226–15239 (2013)
Hawner, C., Li, K., Cirriez, V. & Alexakis, A. Copper-catalyzed asymmetric conjugate addition of aryl aluminum reagents to trisubstituted enones: construction of aryl-substituted quaternary centers. Angew. Chem. Int. Edn 47, 8211–8214 (2008)
May, T. L., Brown, M. K. & Hoveyda, A. H. Enantioselective synthesis of all-carbon quaternary stereogenic centers by catalytic asymmetric conjugate additions of alkyl and aryl aluminum reagents to five-, six-, and seven-membered-ring β-substituted cyclic enones. Angew. Chem. Int. Edn 47, 7358–7362 (2008)
May, T. L., Dabrowski, J. A. & Hoveyda, A. H. Formation of vinyl-, vinylhalide- or acyl-substituted quaternary carbon stereogenic centers through NHC–Cu-catalyzed enantioselective conjugate additions of Si-containing vinylaluminums to β-substituted cyclic enones. J. Am. Chem. Soc. 133, 736–739 (2011)
Lee, K.-S., Brown, M. K., Hird, A. W. & Hoveyda, A. H. A practical method for enantioselective synthesis of all-carbon quaternary stereogenic centers through NHC-Cu-catalyzed conjugate additions of alkyl and arylzinc reagents to β-substituted cyclic enones. J. Am. Chem. Soc. 128, 7182–7184 (2006)
Martin, D. et al. Copper-catalyzed asymmetric conjugate addition of Grignard reagents to trisubstituted enones. Construction of all-carbon quaternary chiral centers. J. Am. Chem. Soc. 128, 8416–8417 (2006)
Sidera, M., Roth, P. M. C., Maksymowicz, R. M. & Fletcher, S. P. Formation of quaternary carbon centers by copper-catalyzed asymmetric conjugate addition of alkylzirconium reagents. Angew. Chem. Int. Edn 52, 7995–7999 (2013)
Shintani, R., Duan, W.-L. & Hayashi, T. Rhodium-catalyzed asymmetric construction of quaternary stereocenters: ligand-dependent regiocontrol in the 1,4-addition to substituted maleimides. J. Am. Chem. Soc. 128, 5628–5629 (2006)
Shintani, R., Tsutsumi, Y., Nagaosa, M., Nishimura, T. & Hayashi, T. Sodium tetraarylborates as effective nucleophiles in rhodium/diene-catalyzed 1,4-addition to β,β-disubstituted α,β-unsaturated ketones: catalytic asymmetric construction of quaternary carbon stereocenters. J. Am. Chem. Soc. 131, 13588–13589 (2009)
Hawner, C. et al. Rhodium-catalyzed asymmetric 1,4-addition of aryl alanes to trisubstituted enones: Binap as an effective ligand in the formation of quaternary stereocenters. Angew. Chem. Int. Edn 49, 7769–7772 (2010)
Holder, J. C. et al. Mechanism and enantioselectivity in palladium-catalyzed conjugate addition of arylboronic acids to β-substituted cyclic enones: insights from computation and experiment. J. Am. Chem. Soc. 135, 14996–15007 (2013)
Mendoza, A., Ishihara, Y. & Baran, P. S. Scalable enantioselective total synthesis of taxanes. Nature Chem. 4, 21–25 (2012)
Brown, M. K. & Hoveyda, A. H. Enantioselective total synthesis of clavirolide C. Applications of Cu-catalyzed asymmetric conjugate additions and Ru-catalyzed ring-closing metathesis. J. Am. Chem. Soc. 130, 12904–12906 (2008)
Wu, F., Li, H., Hong, R. & Deng, L. Construction of quaternary stereocenters by efficient and practical conjugate additions to α,β-unsaturated ketones with a chiral organic catalyst. Angew. Chem. Int. Edn 45, 947–950 (2006)
Poulsen, T. B., Bernardi, L., Alemán, J., Overgaard, J. & Jørgensen, K. A. Organocatalytic asymmetric direct α-alkynylation of cyclic β-ketoesters. J. Am. Chem. Soc. 129, 441–449 (2007)
Penon, O. et al. Quaternary stereogenic carbon atoms in complex molecules by an asymmetric, organocatalytic, triple-cascade reaction. Chemistry 14, 4788–4791 (2008)
Mase, N., Thayumanavan, R., Tanaka, F. & Barbas, C. F., III Direct asymmetric organocatalytic Michael reactions of α,α-disubstituted aldehydes with β-nitrostyrenes for the synthesis of quaternary carbon-containing products. Org. Lett. 6, 2527–2530 (2004)
Kerr, M. S. & Rovis, T. Enantioselective synthesis of quaternary stereocenters via a catalytic asymmetric Stetter reaction. J. Am. Chem. Soc. 126, 8876–8877 (2004)
Gao, L., Kang, B. C. & Ryu, D. H. Catalytic asymmetric insertion of diazoesters into aryl-CHO bonds: highly enantioselective construction of chiral all-carbon quaternary centers. J. Am. Chem. Soc. 135, 14556–14559 (2013)
Doyle, A. G. & Jacobsen, E. N. Enantioselective alkylation of acyclic α,α-disubstituted tributyltin enolates catalyzed by a {Cr(salen)} complex. Angew. Chem. Int. Edn 46, 3701–3705 (2007)
Krautwald, S., Sarlah, D., Schafroth, M. A. & Carreira, E. M. Enantio- and diastereodivergent dual catalysis: α-allylation of branched aldehydes. Science 340, 1065–1068 (2013)
Gao, F., Carr, J. L. & Hoveyda, A. H. Copper-catalyzed enantioselective allylic substitution with readily accessible carbonyl- and acetal-containing vinylboron reagents. Angew. Chem. Int. Edn 51, 6613–6617 (2012)
Van Veldhuizen, J. J., Campbell, J. E., Giudici, R. E. & Hoveyda, A. H. A readily available chiral Ag-based N-heterocyclic carbene complex for use in efficient and highly enantioselective Ru-catalyzed olefin metathesis and Cu-catalyzed allylic alkylation reactions. J. Am. Chem. Soc. 127, 6877–6882 (2005)
Dabrowski, J. A., Gao, F. & Hoveyda, A. H. Enantioselective synthesis of alkyne-substituted quaternary carbon stereogenic centers through NHC-Cu-catalyzed allylic substitution reactions with (i-Bu)2(alkynylaluminum reagents. J. Am. Chem. Soc. 133, 4778–4781 (2011)
Feng, J., Garza, V. J. & Krische, M. J. Redox-triggered C−C coupling of alcohols and vinyl epoxides: diastereo- and enantioselective formation of all-carbon quaternary centers via tert-(hydroxy)-prenylation. J. Am. Chem. Soc. 136, 8911–8914 (2014)
Behenna, D. C. & Stoltz, B. M. The enantioselective Tsuji allylation. J. Am. Chem. Soc. 126, 15044–15045 (2004)
Trost, B. M. & Xu, J. Regio- and enantioselective Pd-catalyzed allylic alkylation of ketones through allyl enol carbonates. J. Am. Chem. Soc. 127, 2846–2847 (2005)An excellent synopsis of the development of the asymmetric allylic alkylation reaction.
Trost, B. M. & Frederiksen, M. U. Palladium-catalyzed asymmetric allylation of prochiral nucleophiles: synthesis of 3-allyl-3-aryl oxindoles. Angew. Chem. Int. Edn 44, 308–310 (2005)
Trost, B. M., Xie, J. & Sieber, J. D. The palladium catalyzed asymmetric addition of oxindoles and allenes: an atom-economical versatile method for the construction of chiral indole alkaloids. J. Am. Chem. Soc. 133, 20611–20622 (2011)
Trost, B. M., Malhotra, S. & Chan, W. H. Exercising regiocontrol in palladium-catalyzed asymmetric prenylations and geranylation: unifying strategy toward flustramines A and B. J. Am. Chem. Soc. 133, 7328–7331 (2011)
Enquist, J. A., Jr & Stoltz, B. M. The total synthesis of (–)-cyanthiwigin F by means of double catalytic enantioselective alkylation. Nature 453, 1228–1231 (2008)
Du, C., Li, L., Li, Y. & Xie, Z. Construction of two vicinal quaternary carbons by asymmetric allylic alkylation: total synthesis of hyperolactone C and (–)-biyouyanagin A. Angew. Chem. Int. Edn 48, 7853–7856 (2009)
Trost, B. M., Miller, J. R. & Hoffman, C. M., Jr A highly enantio- and diastereoselective molybdenum-catalyzed asymmetric allylic alkylation of cyanoesters. J. Am. Chem. Soc. 133, 8165–8167 (2011)
Liu, W.-B., Reeves, C. M., Virgil, S. C. & Stoltz, B. M. Construction of vicinal tertiary and all-carbon quaternary stereocenters via Ir-catalyzed regio-, diastereo-, and enantioselective allylic alkylation and applications in sequential Pd catalysis. J. Am. Chem. Soc. 135, 10626–10629 (2013)
Trost, B. M. & Quancard, J. Palladium-catalyzed enantioselective C-3 allylation of 3-substituted-1H-indoles using trialkylboranes. J. Am. Chem. Soc. 128, 6314–6315 (2006)
Hills, I. D. & Fu, G. C. Catalytic enantioselective synthesis of oxindoles and benzofuranones that bear a quaternary stereocenter. Angew. Chem. Int. Edn 42, 3921–3924 (2003)
Shaw, S. A. et al. Enantioselective TADMAP-catalyzed carboxyl migration reactions for the synthesis of stereogenic quaternary carbon. J. Am. Chem. Soc. 128, 925–934 (2006)
Mermerian, A. H. & Fu, G. C. Nucleophile-catalyzed asymmetric acylations of silyl ketene imines: application to the enantioselective synthesis of verapamil. Angew. Chem. Int. Edn 44, 949–952 (2005)
DeLorbe, J. E., Jabri, S. Y., Mennen, S. M., Overman, L. E. & Zhang, F.-L. Enantioselective total synthesis of (+)-gliocladine C: convergent construction of cyclotryptamine-fused polyoxopiperazines and a general approach for preparing epidithiodioxopiperazines from trioxopiperazine precursors. J. Am. Chem. Soc. 133, 6549–6552 (2011)
Austin, J. F., Kim, S.-G., Sinz, C. J., Xiao, W.-J. & MacMillan, D. W. C. Enantioselective organocatalytic construction of pyrroloindolines by a cascade addition–cyclization strategy: synthesis of (–) flustramine B. Proc. Natl Acad. Sci. USA 101, 5482–5487 (2004)
Zhu, S. & MacMillan, D. W. C. Enantioselective copper-catalyzed construction of aryl pyrroloindolines via an arylation−cyclization cascade. J. Am. Chem. Soc. 134, 10815–10818 (2012)
Xie, X., Chen, Y. & Ma, D. Enantioselective arylation of 2-methylacetoacetates catalyzed by CuI/trans-4-hydroxy-L-proline at low reaction temperatures. J. Am. Chem. Soc. 128, 16050–16051 (2006)
García-Fortanet, J. & Buchwald, S. L. Asymmetric palladium-catalyzed intramolecular α-arylation of aldehydes. Angew. Chem. Int. Edn 47, 8108–8111 (2008)
Taylor, A. M., Altman, R. A. & Buchwald, S. L. Palladium-catalyzed enantioselective α-arylation and α-vinylation of oxindoles facilitated by an axially chiral P-stereogenic ligand. J. Am. Chem. Soc. 131, 9900–9901 (2009)
Mitsunuma, H., Shibasaki, M., Kanai, M. & Matsunaga, S. Catalytic asymmetric total synthesis of chimonanthine, folicanthine, and calycanthine through double Michael reaction of bisoxindole. Angew. Chem. Int. Edn 51, 5217–5221 (2012)
Zhang, H., Hong, L., Kang, H. & Wang, R. Construction of vicinal all-carbon quaternary stereocenters by catalytic asymmetric alkylation reaction of 3-bromooxindoles with 3-substituted indoles: total synthesis of (+)-perophoramidine. J. Am. Chem. Soc. 135, 14098–14101 (2013)
Malcolmson, S. J., Meek, S. J., Sattely, E. S., Schrock, R. R. & Hoveyda, A. H. Highly efficient molybdenum-based catalysts for enantioselective alkene metathesis. Nature 456, 933–937 (2008)An excellent example of using desymmetrization to install a key quaternary stereocentre in a structurally complex natural product.
Giudici, R. E. & Hoveyda, A. H. Directed catalytic asymmetric olefin metathesis. Selectivity control by enoate and ynoate groups in Ru-catalyzed asymmetric ring-opening/cross-metathesis. J. Am. Chem. Soc. 129, 3824–3825 (2007)
Kleinbeck, F. & Toste, F. D. Gold(I)-catalyzed enantioselective ring expansion of allenylcyclopropanols. J. Am. Chem. Soc. 131, 9178–9179 (2009)
Phan, D. H. T., Kou, K. G. M. & Dong, V. M. Enantioselective desymmetrization of cyclopropenes by hydroacylation. J. Am. Chem. Soc. 132, 16354–16355 (2010)
Souillart, L., Parker, E. & Cramer, N. Highly enantioselective rhodium(I)-catalyzed activation of enantiotopic cyclobutanone C–C bonds. Angew. Chem. Int. Edn 53, 3001–3005 (2014)
Shi, B.-F., Zhang, Y.-H., Lam, J. K., Wang, D.-H. & Yu, J.-Q. Pd(II)-catalyzed enantioselective C-H olefination of diphenylacetic acids. J. Am. Chem. Soc. 132, 460–461 (2010)
Aikawa, K., Okamoto, T. & Mikami, K. Copper(I)-catalyzed asymmetric desymmetrization: synthesis of five-membered-ring compounds containing all-carbon quaternary stereocenters. J. Am. Chem. Soc. 134, 10329–10332 (2012)
Zhang, E., Fan, C.-A., Tu, Y.-Q., Zhang, F.-M. & Song, Y.-L. Organocatalytic asymmetric vinylogous α-ketol rearrangement: enantioselective construction of chiral all-carbon quaternary stereocenters in spirocyclic diketones via semipinacol-type 1,2-carbon migration. J. Am. Chem. Soc. 131, 14626–14627 (2009)
Wadamoto, M., Phillips, E. M., Reynolds, T. E. & Scheidt, K. A. Enantioselective synthesis of α,α-disubstituted cyclopentenes by an N-heterocyclic carbene-catalyzed desymmetrization of 1,3-diketones. J. Am. Chem. Soc. 129, 10098–10099 (2007)
Das, J. P. & Marek, I. Enantioselective synthesis of all-carbon quaternary stereogenic centers in acyclic systems. Chem. Commun. 47, 4593–4623 (2011)
Xiao, K.-J. et al. Palladium(II)-catalyzed enantioselective C(sp3)–H activation using a chiral hydroxamic acid ligand. J. Am. Chem. Soc. 136, 8138–8142 (2014)
Notte, G. T. New chemical entities entering phase III trials in 2012. Annu. Rep. Med. Chem. 48, 451–469 (2013)
Lovering, F. Escape from flatland 2: complexity and promiscuity. MedChemComm. 4, 515–519 (2013)
Acknowledgements
Our research in this area was supported by the US National Institutes of Health (R01 GM030859 and GM098601).
Author information
Authors and Affiliations
Contributions
Both K.W.Q. and L.E.O. prepared the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Quasdorf, K., Overman, L. Catalytic enantioselective synthesis of quaternary carbon stereocentres. Nature 516, 181–191 (2014). https://doi.org/10.1038/nature14007
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature14007
This article is cited by
-
Alkene dialkylation by triple radical sorting
Nature (2024)
-
Ketone α-alkylation at the more-hindered site
Nature Communications (2023)
-
Control of dynamic sp3-C stereochemistry
Nature Chemistry (2023)
-
An efficient approach to angular tricyclic molecular architecture via Nazarov-like cyclization and double ring-expansion cascade
Nature Communications (2022)
-
Non-enzymatic catalytic asymmetric cyanation of acylsilanes
Communications Chemistry (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.