Abstract
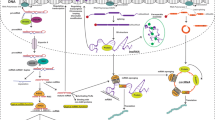
MicroRNAs are short non-coding RNAs expressed in different tissue and cell types that suppress the expression of target genes. As such, microRNAs are critical cogs in numerous biological processes1,2, and dysregulated microRNA expression is correlated with many human diseases. Certain microRNAs, called oncomiRs, play a causal role in the onset and maintenance of cancer when overexpressed. Tumours that depend on these microRNAs are said to display oncomiR addiction3,4,5. Some of the most effective anticancer therapies target oncogenes such as EGFR and HER2; similarly, inhibition of oncomiRs using antisense oligomers (that is, antimiRs) is an evolving therapeutic strategy6,7. However, the in vivo efficacy of current antimiR technologies is hindered by physiological and cellular barriers to delivery into targeted cells8. Here we introduce a novel antimiR delivery platform that targets the acidic tumour microenvironment, evades systemic clearance by the liver, and facilitates cell entry via a non-endocytic pathway. We find that the attachment of peptide nucleic acid antimiRs to a peptide with a low pH-induced transmembrane structure (pHLIP) produces a novel construct that could target the tumour microenvironment, transport antimiRs across plasma membranes under acidic conditions such as those found in solid tumours (pH approximately 6), and effectively inhibit the miR-155 oncomiR in a mouse model of lymphoma. This study introduces a new model for using antimiRs as anti-cancer drugs, which can have broad impacts on the field of targeted drug delivery.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
He, L. & Hannon, G. J. MicroRNAs: small RNAs with a big role in gene regulation. Nature Rev. Genet. 5, 522–531 (2004)
Calin, G. A. & Croce, C. M. MicroRNA signatures in human cancers. Nature Rev. Cancer 6, 857–866 (2006)
Esquela-Kerscher, A. & Slack, F. J. Oncomirs — microRNAs with a role in cancer. Nature Rev. Cancer 6, 259–269 (2006)
Medina, P. P., Nolde, M. & Slack, F. J. OncomiR addiction in an in vivo model of microRNA-21-induced pre-B-cell lymphoma. Nature 467, 86–90 (2010)
Babar, I. A. et al. Nanoparticle-based therapy in an in vivo microRNA-155 (miR-155)-dependent mouse model of lymphoma. Proc. Natl Acad. Sci. USA 109, E1695–E1704 (2012)
Stenvang, J., Petri, A., Lindow, M., Obad, S. & Kauppinen, S. Inhibition of microRNA function by antimiR oligonucleotides. Silence 3, 1 (2012)
Kasinski, A. L. & Slack, F. J. Epigenetics and genetics. MicroRNAs en route to the clinic: progress in validating and targeting microRNAs for cancer therapy. Nature Rev. Cancer 11, 849–864 (2011)
Cheng, C. J., Saltzman, W. M. & Slack, F. J. Canonical and non-canonical barriers facing antimiR cancer therapeutics. Curr. Med. Chem. 20, 3582–3593 (2013)
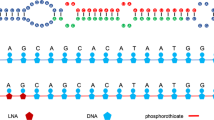
Elmen, J. et al. LNA-mediated microRNA silencing in non-human primates. Nature 452, 896–899 (2008)
Krutzfeldt, J. et al. Silencing of microRNAs in vivo with ‘antagomirs’. Nature 438, 685–689 (2005)
White, P. J., Anastasopoulos, F., Pouton, C. W. & Boyd, B. J. Overcoming biological barriers to in vivo efficacy of antisense oligonucleotides. Expert Rev. Mol. Med. 11, e10 (2009)
Vaupel, P., Kallinowski, F. & Okunieff, P. Blood flow, oxygen and nutrient supply, and metabolic microenvironment of human tumors: a review. Cancer Res. 49, 6449–6465 (1989)
Reshetnyak, Y. K., Andreev, O. A., Lehnert, U. & Engelman, D. M. Translocation of molecules into cells by pH-dependent insertion of a transmembrane helix. Proc. Natl Acad. Sci. USA 103, 6460–6465 (2006)
Thevenin, D., An, M. & Engelman, D. M. pHLIP-mediated translocation of membrane impermeable molecules into cells. Chem. Biol. 16, 754–762 (2009)
Reshetnyak, Y. K. et al. Measuring tumor aggressiveness and targeting metastatic lesions with fluorescent pHLIP. Mol. Imaging Biol. 13, 1146–1156 (2011)
Nielsen, P. E., Egholm, M. & Buchardt, O. Peptide nucleic acid (PNA). A DNA mimic with a peptide backbone. Bioconjug. Chem. 5, 3–7 (1994)
Østergaard, H., Tachibana, C. & Winther, J. R. Monitoring disulfide bond formation in the eukaryotic cytosol. J. Cell Biol. 166, 337–345 (2004)
Rai, D., Karanti, S., Jung, I., Dahia, P. L. M. & Aguiar, R. C. T. Coordinated expression of microRNA-155 and predicted target genes in diffuse large B-cell lymphoma. Cancer Genet. Cytogenet. 181, 8–15 (2008)
Rather, M. I., Nagashri, M. N., Swamy, S. S., Gopinath, K. S. & Kumar, A. Oncogenic microRNA-155 down-regulates tumor suppressor CDC73 and promotes oral squamous cell carcinoma cell proliferation: implications for cancer therapeutics. J. Biol. Chem. 288, 608–618 (2013)
Costinean, S. et al. Pre-B cell proliferation and lymphoblastic leukemia/high-grade lymphoma in Eμ-miR155 transgenic mice. Proc. Natl Acad. Sci. USA 103, 7024–7029 (2006)
Sandhu, S. K., Croce, C. M. & Garzon, R. Micro-RNA expression and function in lymphomas. Adv. Hematol. 2011, 1–12 (2011)
Cheng, C. J. & Slack, F. J. The duality of oncomiR addiction in the maintenance and treatment of cancer. Cancer J. 18, 232–237 (2012)
DeVita, V. T., Lawrence, T. S. & Rosenberg, S. A. DeVita, Hellman, and Rosenberg’s Cancer: Principles & Practice of Oncology 9th edn (Lippincott Williams & Wilkins, 2011)
Zhang, Y. et al. LNA-mediated anti-miR-155 silencing in low-grade B-cell lymphomas. Blood 120, 1678–1686 (2012)
Gottwein, E. et al. A viral microRNA functions as an orthologue of cellular miR-155. Nature 450, 1096–1099 (2007)
Dweep, H., Sticht, C., Pandey, P. & Gretz, N. miRWalk–database: prediction of possible miRNA binding sites by ‘walking’ the genes of three genomes. J. Biomed. Inform. 44, 839–847 (2011)
Li, S. et al. microRNA-155 silencing inhibits proliferation and migration and induces apoptosis by upregulating BACH1 in renal cancer cells. Mol. Med. Report. 5, 949–954 (2012)
Brognara, E. et al. Uptake by human glioma cell lines and biological effects of a peptide-nucleic acids targeting miR-221. J. Neurooncol. 118, 19–28 (2014)
Wang, Y.-Z. et al. Delivery of an miR155 inhibitor by anti-CD20 single-chain antibody into B cells reduces the acetylcholine receptor-specific autoantibodies and ameliorates experimental autoimmune myasthenia gravis. Clin. Exp. Immunol. 176, 207–221 (2014)
Christensen, L. et al. Solid-phase synthesis of peptide nucleic acids. J. Pept. Sci. 1, 175–183 (1995)
Obad, S. et al. Silencing of microRNA families by seed-targeting tiny LNAs. Nature Genet. 43, 371–378 (2011)
Sahu, B. et al. Synthesis and characterization of conformationally preorganized, (R)-diethylene glycol-containing γ-peptide nucleic acids with superior hybridization properties and water solubility. J. Org. Chem. 76, 5614–5627 (2011)
Fabani, M. M. et al. Efficient inhibition of miR-155 function in vivo by peptide nucleic acids. Nucleic Acids Res. 38, 4466–4475 (2010)
Shiraishi, T. & Nielsen, P. E. Peptide nucleic acid (PNA) cell penetrating peptide (CPP) conjugates as carriers for cellular delivery of antisense oligomers. Artif. DNA PNA XNA 2, 90–99 (2011)
Fabbri, E. et al. miRNA therapeutics: delivery and biological activity of peptide nucleic acids targeting miRNAs. Epigenomics 3, 733–745 (2011)
Xu, G. et al. Transcriptome and targetome analysis in MIR155 expressing cells using RNA-seq. RNA 16, 1610–1622 (2010)
Loeb, G. B. et al. Transcriptome-wide miR-155 binding map reveals widespread noncanonical microRNA targeting. Mol. Cell 48, 760–770 (2012)
Acknowledgements
We thank M. Bosenberg, Y. Dang, A. Karabadzhak, and J. Zhou for discussions and suggestions; R. Ardito, M. Bonk, K. Card, D. Caruso, D. Jenci, D. Laliberte, W. Nazzaro, N. Santiago, and S. Wilson for rodent services; A. Brooks for tissue pathology services; Antech Diagnostics for complete blood count analysis; E. Aronesty, B. Cooper, and E. Norris at Expression Analysis for RNA-seq services; and J. Deacon, A. Kasinski, J. Sawyer, and C. Stahlhut for reading the manuscript. C.J.C. is the recipient of a Ruth L. Kirschstein Postdoctoral Fellowship from the National Cancer Institute/National Institutes of Health (NCI/NIH) (F32CA174247). Our work has been supported by grants from the NCI/NIH (R01CA131301), the National Heart, Lung, and Blood Institute (NHLBI)/NIH (R01HL085416), the National Institute of General Medical Sciences (NIGMS)/NIH (R01GM073857), the National Institute of Environmental Health Sciences (NIEHS)/NIH (R01ES005775), the NCI/NIH (R01CA148996), the National Institute of Biomedical Imaging and Bioengineering (NIBIB)/NIH (R01EB000487), the NHLBI/NIH (2T32HL007974), and pilot grants from the Yale Comprehensive Cancer Center.
Author information
Authors and Affiliations
Contributions
C.J.C., R.B., F.B., A.S., P.M.G., D.M.E., W.M.S., and F.J.S. designed the research; C.J.C. performed the research; R.B. synthesized the PNA; I.A.B. and C.J.C. developed and maintained the rodent colonies; C.J.C., Z.P., and C.L. performed the bioinformatics analysis; D.T.B. performed the pathological analysis; C.J.C., R.B., D.B., P.M.G., D.M.E., W.M.S., and F.J.S. analysed the data and wrote the paper.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Extended data figures and tables
Extended Data Figure 1 Distribution of pHLIP to the renal system and lymph node metastases.
a, Intravenous injection of A750-pHLIP distributes to the (white arrow) kidneys and (blue arrow) tumour in a representative mir-155LSLtTA subcutaneous flank model (n = 3); time points indicate hours after a single injection of A750-pHLIP. Previous reports have observed systemic distribution of pHLIP to kidneys in other mouse models15. Similarly, we speculate that the increased uptake of pHLIP peptide in the kidneys is due to excretion and increased acidity of renal tubule cells. Initially kidneys are highly enriched for pHLIP, which is gradually excreted while pHLIP shows a more steady accumulation in the tumour. b, Representative example showing A750-pHLIP distribution to the (white arrow) bladder and (yellow arrow) enlarged axillary lymph node 36 h after intravenous administration into mir-155LSLtTA mice with lymphadenopathy (n = 3). c, In addition to distributing to the (white arrow) primary mir-155LSLtTA flank tumour and (red arrow) kidneys, A750-pHLIP distributes to (black arrows) enlarged lymph nodes that resulted from metastatic spread; intravital fluorescence of A750-pHLIP was detected 48 h after intravenous injection into nude transplant mice with conspicuous lymphadenopathy (a representative animal from n = 3 is shown).
Extended Data Figure 2 Assessment of pHLIP–PNA conjugation and activity.
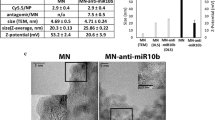
a, HPLC elution profiles of (top) free PNA, (middle) reaction mixture of PNA and pHLIP-C(Npys), and (bottom) purified pHLIP–PNA incubated in DTT. HPLC was used to purify pHLIP–PNA (black arrow). The fluorescence detection of TAMRA (ex/em: 540/575) that was conjugated to the PNA is shown; samples were also detected by absorbance at 260 and 280 nm (data not shown). b, Tricine SDS–PAGE evaluation of pHLIP–PNA conjugation. Gel was visualized by (top) TAMRA fluorescence to detect labelled PNA and (bottom) Coomassie stain to detect both PNA and peptide. c, Gelshift analysis of pHLIP-antimiR-155 binding to miR-155 and disulphide reduction in the presence of DTT. d, High-magnification confocal projections of A549 cells incubated with labelled pHLIP-antimiR (against control miR-182); scale bars, 7.5 µm. The diffuse intracellular fluorescence is indicative of freely distributed antimiR throughout the cytosol—note that the presence of marginal punctate fluorescence at both pH levels suggests that endocytosis is probably an additional mode of cell entry. e, Toledo DLBCL lymphocytes were incubated with labelled pHLIP-anti155 at pH 6.2; fluorescence of a representative live cell is overlaid on a bright field micrograph; scale bars, 2 µm. f, Flow cytometry analysis of Toledo cells incubated with labelled pHLIP-anti155; cell association was dependent on dose (top, pH 6.2) and pH (bottom, 500 nM dose). g, Inhibition of miR-155 demonstrated by de-repression of a miR-155 dual luciferase sensor in KB cells. h, Inhibition of miR-21 demonstrated by de-suppression of luciferase expression in A549 cells transfected with a Renilla luciferase sensor. Data are shown as mean ± s.d., with n = 3; statistical analysis performed with two-tailed Student’s t-test; **P < 0.01; ***P < 0.001.
Extended Data Figure 3 Pathology of the mir-155LSLtTA model of oncomiR addiction.
a, Organomegaly in representative diseased mir-155LSLtTA mice: top, conspicuous lymphadenopathy seen in the (black arrow) cervical and (white arrow) brachial lymph nodes; middle, enlarged exposed (white arrows) cervical and (black arrows) axillary lymph nodes; bottom, enlarged (black arrows) spleen. b, Histopathology of mir-155LSLtTA mice: H&E stain of an enlarged spleen shows expansion of the white pulp by a nodular, neoplastic infiltrate; staining of the spleen shows CD20+ and CD10+ B cells of follicular centre origin. Analysis of enlarged lymph nodes indicates DLBCL with lymph node architecture effaced by a confluent population of B220+ neoplastic lymphocytes and a Ki-67 proliferative index at nearly 100%; n = 5. c, Tumour regression due to DOX-induced miR-155 withdrawal in a subcutaneous tumour model established from transplanted splenic mir-155LSLtTA lymphocytes; time points indicate hours after initial administration of DOX. With a cancer phenotype that is relevant to human disease yet can be modulated by miRNA silencing, this is an excellent model for evaluating miR-155-targeted therapies.
Extended Data Figure 4 Experimental schematics for mouse tumour studies.
a, Workflow for treatment of the mir-155LSLtTA subcutaneous flank model for the early endpoint and survival studies; day 1 indicates time of first injection. For the ‘early treatment’ experiments in Fig. 3a, b, d–f, h and Extended Data Fig. 5b, c, mice were treated on days 1 and 2 with pHLIP-anti155, mock buffer, pHLIP-antiscr and anti155 only; fed DOX starting on day 3; or treated with CHOP regimen on days 2–4. For survival experiments in Fig. 3c, g and Extended Data Fig. 5a, mice were treated on days 1–3 with pHLIP-anti155, LNA against miR-155, and mock buffer. b, Workflow for investigation of the mir-155LSLtTA model of lymphoma for the biodistribution and miR-155 silencing studies. For experiments in Fig. 4a and Extended Data Fig. 8a, b, mice were treated on day 1 with pHLIP-anti155, anti155 only, and mock buffer. For experiments in Fig. 4b–d, h and Extended Data Fig. 8c–g, mice were treated on day 1 and day 3 with pHLIP-anti155, pHLIP-antiscr, and mock buffer, or fed DOX 16 h before harvest.
Extended Data Figure 5 Administration of pHLIP-anti155 to mice with subcutaneous lymphoma flank tumours.
a, Fold change in tumour size in response to miR-155 withdrawal and CHOP treatment (n = 3); arrow represents initiation of DOX treatment (n = 3, food pellets enriched with DOX at 2.3 g/kg, Bio-Serv), white triangle represents CHOP treatment (systemic injection of cyclophosphamide at 40 mg/kg, doxorubicin at 3.3 mg/kg, and vincristine at 0.5 mg/kg; oral gavage of prednisone at 0.2 mg/kg), grey triangles represent maintenance administration of prednisone. b, Tumour growth response to systemically administered antimiR treatment; symbols represent intravenous injections of 2 (arrowhead) or 1 (arrow) mg kg−1 of pHLIP-conjugated antimiR-155, molar equivalent of phosphorothioated antimiR-155 LNA, or mock delivery solution; n = 5, data are shown as mean ± s.e.m.; statistical comparison of pHLIP-anti155 to LNA performed with two-way ANOVA; ***P < 0.001, ****P < 0.0001. c, Representative histological analysis of kidneys (H&E, ×100 magnification) harvested from early endpoint study, in which all of the mice from Fig. 3a and Extended Data Fig. 5a were killed at the same time for analysis. Kidney sections reveal an absence of microscopic changes in treated animals (pHLIP-anti155) that would be indicative of renal toxicity (compare with normal renal sections in mock control). d, Representative pHLIP-antiscr-treated mouse (top) with primary flank tumour (white arrow) and enlarged inguinal lymph node (black arrow) compared with an untreated mouse with no tumour (bottom). e, Measurement of circulating lymphocytes in blood collected at time of death in early endpoint study; dotted line denotes average level in nude mice with no tumour. f, Although pHLIP interacts with the outer leaflet of lipid membranes, no significant change in red blood cell (RBC) levels was detected after intravenous treatment of mice with subcutaneous mir-155LSLtTA transplant tumours. This supports the specificity of pHLIP treatments on cells of tumour origin since pHLIP-antimiR treatments affect the levels of circulating lymphocytes (Extended Data Fig. 5e); data are shown as mean ± s.d.
Extended Data Figure 6 Toxicology assessment of intravenously administered pHLIP-anti155 to C57BL/6J mice.
a, Serum-based clinical chemistry evaluation of systemic toxicity with focus on liver and kidney function; dosing schedule consisted of injections of 2 mg kg−1 of pHLIP-anti155 (and equimolar dose of LNA) on days 10 and 12, as well as 1 mg kg−1 on day 11. Blood samples were serially harvested retro-orbitally on day 0 (10 days before start of treatment), as well as 1 day and 14 days after treatment. b, Circulating white blood cell count collected 14 days after treatment. c, Mouse mass throughout duration of the study. d, Organ mass normalized to total body mass at time of harvest. a–d, For all analyses mock n = 4, pHLIP-anti155 n = 5, and LNA n = 5; dotted lines indicate typical wild-type values for C57BL/6J mice.
Extended Data Figure 7 Administration of pHLIP-anti155 to mice with KB oral squamous cell carcinoma xenograft tumours.
a, Intravenous injection of pHLIP-anti155 (**) and phosphorothioated LNA against miR-155 (*) significantly enhanced survival compared with mock buffer treatment; n = 4 for all groups; arrowheads indicate injections of 2 mg kg−1 (or molar equivalent for LNA). Survival cutoff criteria included tumour volume greater than 1 cm3 or compassionate euthanasia, which was mandated for three mock-treated mice with ulcerated tumours. b, Fold change in tumour size in response to treatment; measurements were plotted until the mock negative control group was euthanized. c, Tumour bioluminescence in response to treatment; day 8 represents luciferase activity before first injection. d, Representative images of tumour bioluminescence. Data are shown as mean ± s.e.m.; statistical analysis performed with (a) Mantel-Cox analysis or (c) two-tailed Student’s t-test, *P < 0.05; **P < 0.01.
Extended Data Figure 8 Administration of pHLIP-anti155 to mir-155LSLtTA mice with lymphoma.
a, Quantification of liver distribution of TAMRA-labelled PNA delivered with and without conjugation to pHLIP; ImageJ was used to measure fluorescence from five confocal sections per mouse liver; n = 3 mice per group. b, Visualization of whole liver fluorescence after antimiR administration; pHLIP-anti155 liver fluorescence is similar to the autofluorescence seen in the mock group. c, Lymph-node tumour burden (A, axillary; B, brachial; C, cervical; I, inguinal lymph nodes); in these specific images taken from diseased littermates, pHLIP-antiscr-treated mice had a more than threefold larger aggregate lymph node mass (3.179 g) than pHLIP-anti155-treated mice (1.006 g). d, e, Size of harvested (d) spleens (n = 4) and (e) lymph nodes (axillary, brachial, cervical, and inguinal; n = 5) with respect to wild type; n < 6 (that is, total number of treated mice) owing to size data not collected. f, g, TUNEL analysis of treated cervical lymph nodes of mir-155LSLtTA mice (n = 6). h, Percentage of white pulp in treated spleens; n = 6. i, Measurement of lymphocyte infiltration into liver; n = 6. j, Low-magnification H&E images of livers from Fig. 4d. k, Flow cytometry analysis of B220-positive cells comprising the spleens of treated mice; B220 is typically a marker for B cells, although varied expression is seen on some T cells, natural killer cells, and macrophages; n = 4. l, Representative H&E image of healthy kidneys from pHLIP-anti155-treated mice; n = 6. Data are shown as mean ± s.d. (a, d, e, g, h) or mean ± s.e.m. (i); statistical analysis performed with two-tailed Student’s t-test; **P < 0.01; ****P < 0.0001.
Extended Data Figure 9 Differential gene expression analysis of miR-155 withdrawal.
a, Experimental design for RNA-seq analysis of miR-155-addicted tumours compared with tumours undergoing miR-155 withdrawal and tumour regression. b, RNA-seq differential gene expression analysis of three independent tumours overexpressing miR-155 compared with three independent tumours undergoing DOX-induced miR-155 withdrawal; all differentially expressed genes with a false discovery rate less than 0.05 are shown; rows are clustered by Euclidean distance measure. c, KEGG pathway analysis of significantly upregulated genes after miR-155 withdrawal. d, Selection of potential miR-155 targets involved in tumour regression. Intersection of genes (group I) that are both predicted miR-155 targets (Supplementary Table 2) and overexpressed after miR-155 withdrawal from mir-155LSLtTA tumours (Supplementary Table 1) with genes inferred from three separate miR-155 target analyses. Group II: the study in ref. 36 used RNA-seq to compare Mutu I B cells that overexpress miR-155 with cells transformed with a control vector36. Group III: ref. 25 identified shared targets between miR-155 and a viral orthologue, miR-K12-11. Group IV: the study in ref. 37 used HITS-CLIP to identify miR-155 targets without perfect seed matches in T cells. e, qPCR determination of gene expression levels in Toledo cells treated for 48 h with 500 nM pHLIP-anti155 at pH 6.2; data are shown as mean ± s.d.; n = 3; statistical analysis performed with two-tailed Student’s t-test, *P < 0.05.
Extended Data Figure 10 Expression levels of putative targets in response to miR-155 silencing in mir-155LSLtTA mice.
qPCR validation of potential miR-155 targets involved in tumour regression using mir-155LSLtTA mice with conspicuous lymphadenopathy treated with (black bars) DOX for 16 h compared with (white bars) untreated mice with lymphadenopathy; all samples are normalized to β-actin; n = 3. Genes were selected on the basis of criteria described in Supplementary Table 3. As shown in Fig. 4f, both Bach1 and Mafb have utility as biomarkers for miR-155 withdrawal-induced tumour regression.
Supplementary information
Supplementary Table 1
Significantly differentially expressed genes upon withdrawal of miR-155 hyperexpression in miR-155-addicted tumors; genes are sorted by log2(fold change), with an FDR cutoff of 0.05. (XLSX 146 kb)
Supplementary Table 2
Identification of potential miR-155 targets. The first gene list includes miR-155 targets predicted using the miRWalk algorithm. The second list intersects the miRWalk targets with all genes that were upregulated upon miR-155 withdrawal; based on the hypergeometric distribution, the probability of achieving this observed overlap by chance is 3.7 x 10-9. The third list includes validated and predicted miR-155 targets compiled by SABiosciences. The remaining lists show the intersection of these sets with published sets of validated and putative miR-155 targets. (XLSX 58 kb)
Supplementary Table 3
Selection criteria for potential targets of miR-155 that are derepressed during miR-155 withdrawal-induced tumor regression. (XLSX 13 kb)
Mouse with hind limb paresis before pHLIP-anti155 treatment
Mouse with hind limb paresis before pHLIP-anti155 treatment. (MOV 22256 kb)
Response of mouse in Supplementary Video 1 to pHLIP-anti155 treatment.
Four days after initiating IV administration of pHLIP-anti155 (two injections of 2 mg/kg spaced 2 days apart), paresis in the mouse is alleviated. (MOV 24511 kb)
Additional mouse with hind limb paresis before pHLIP-anti155 treatment.
Additional mouse with hind limb paresis before pHLIP-anti155 treatment. (MOV 18981 kb)
Response of mouse from Supplementary Video 3 to pHLIP-anti155 treatment
Four days after initiating IV administration of pHLIP-anti155 (one injection of 2.5 mg/kg), the mouse from Supplementary Video 3 has improved posture and gait. (MOV 21718 kb)
Rights and permissions
About this article
Cite this article
Cheng, C., Bahal, R., Babar, I. et al. MicroRNA silencing for cancer therapy targeted to the tumour microenvironment. Nature 518, 107–110 (2015). https://doi.org/10.1038/nature13905
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature13905
This article is cited by
-
Non-coding RNAs in disease: from mechanisms to therapeutics
Nature Reviews Genetics (2024)
-
CircRNA ITCH Inhibits Epithelial–Mesenchymal Transformation and Promotes Apoptosis in Papillary Thyroid Carcinoma via miR-106a-5p/JAZF1 Axis
Biochemical Genetics (2024)
-
Targeted therapy using nanocomposite delivery systems in cancer treatment: highlighting miR34a regulation for clinical applications
Cancer Cell International (2023)
-
MicroRNA 29a therapy for CEACAM6-expressing lung adenocarcinoma
BMC Cancer (2023)
-
Specific expression profile of follicular fluid-derived exosomal microRNAs in patients with diminished ovarian reserve
BMC Medical Genomics (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.