Abstract
Immunoglobulins protect against disease to a considerable extent by activating complement and stimulatory immunoglobulin crystallizable fragment receptors (Ig FcRs), and aggregating microbial pathogens1,2. Yet IgG1, the predominant murine serum Ig isotype, cannot activate complement by the classical pathway, binds more avidly to an inhibitory than to stimulatory FcRs, and has limited ability to aggregate pathogens1,2,3. In these regards, it resembles human IgG4 (ref. 4). We hypothesized that limited ability to activate effector mechanisms might protect against immune complex immunopathology. Here we show that IgG1-deficient (γ1−) mice5, immunized with a potent antigen, develop lethal renal disease soon after they begin to produce antigen-specific antibody, whereas similarly immunized wild-type mice remain healthy. Surprisingly, renal disease in this model is complement and FcR independent and results from immune complex precipitation in glomerular capillaries, as in some cryoglobulinaemic humans6. IgG3, which self-associates to form large immune complexes7,8, accounts for more than 97% of the mouse Ig in this cryoglobulin; furthermore, glomerular disease develops when mice are injected with IgG3 anti-trinitrophenyl (TNP) monoclonal antibody followed by a TNP-labelled protein. Renal disease is prevented in both active and passive immunization models by antigen-specific IgG1; other isotypes are less potent at preventing disease. These observations demonstrate the adaptive significance of Ig isotypes that poorly activate effector mechanisms, reveal an immune-complex-dependent, complement- and FcR-independent nephrotoxic mechanism, and suggest that isotypes that poorly activate effector mechanisms may be useful for inhibiting immune complex immunopathology.
Similar content being viewed by others
Main
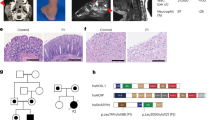
Immunization of wild-type BALB/c or C57BL/6 mice with a potent immunogen, goat anti-mouse IgD antiserum (GaMD), leads to a large, rapid, predominantly IgG1 antibody response to goat IgG (GIgG) and the generation of mouse IgG1–GIgG immune complexes9, but no noticeable disease. In contrast, GaMD-immunized γ1− BALB/c and C57BL/6 mice develop renal disease characterized by increased urinary protein, leukocyte esterase and erythrocytes (blood), starting 6–7 days after immunization, as well as increased blood concentration of urea (BUN) and decreased serum albumin, with anasarca (subcutaneous oedema) and peritoneal effusion (Fig. 1a–e and Extended Data Fig. 1a). Kidney colour in these mice changes from red/brown to yellow, reflecting dramatically decreased perfusion (Fig. 1f). Microscopically, glomerular capillaries contain IgG and complement deposits, but few inflammatory cells (Fig. 1g and Extended Data Fig. 1b, c). The microscopic damage is initially observed 6–7 days after GaMD immunization and is followed by disruption of glomerular structure and development of fibrosis (Fig. 1g and Extended Data Fig. 1c). Because no other organ damage was observed (data not shown), it is likely that renal insufficiency caused the death of 60–80% of γ1− mice by day 16–22 post-immunization (Fig. 1h).
a–h, Wild-type (WT) and γ1− mice (4 per group (a–g); or 8 or 10 per group (h)) were immunized with GaMD. a, Urine protein. b, Serum BUN and albumin. c–g, Representative photographs of mice 13 days after GaMD immunization demonstrate anasarca (c, d), ascites (e), kidney hypoperfusion (f) and glomerulopathy with PAS+ deposits and fibrosis (blue colour on Masson’s stain) (g) only in γ1− mice. H&E, haematoxylin and eosin; PAS, periodic acid–Schiff. Original magnification, ×400. h, Survival curves. All figures show means ± standard error of the mean (s.e.m.). Experiments depicted in all figures were repeated with similar results unless otherwise indicated. *P < 0.05, non-parametric Mann–Whitney two-tailed t-test.
Lack of the normally dominant IgG1 response in GaMD-immunized γ1− mice was accompanied by increased production of IgG3, IgM and, in some experiments, IgG2a (Fig. 2a and Extended Data Fig. 2a). Because these isotypes, unlike IgG1, strongly activate complement, and IgG2a potently activates all stimulatory IgGFcRs1,2,3, we expected renal disease in γ1− mice to be complement and possibly FcR dependent. However, severe renal disease still developed in GaMD-immunized γ1− mice that lacked both C3—the complement component that is generally required for all complement activation pathways2—and FcR γ-chain (FcRγ), a required component of all stimulatory FcRs in mice10 (Fig. 2b, c and Extended Data Fig. 2b). This was true even when these mice were also treated with C5a antagonists (Extended Data Fig. 3). Inhibition of IgG2a production with anti-interferon (IFN)-γ monoclonal antibody11 also failed to suppress kidney disease (Extended Data Fig. 2c, d). Additional studies eliminated the possibilities that renal disease in γ1− mice results from the persistence of circulating antigen or a decreased ratio of Ig to antigen that might form more inflammatory immune complexes (Extended Data Fig. 4).
a, Serum anti-goat IgG titres in wild-type (WT) and γ1− mice (4 per group) 8 days after GaMD immunization. b, c, Urine protein (b) and BUN (c) of GaMD-immunized γ1−, γ1−/FcRγ−, γ1−/C3−, C3−/FcRγ− and γ1−/C3−/FcγR− mice (5 per group). d, e, Serum cryoprecipitate protein and Ig isotype concentrations 6–7 days after GaMD immunization of wild-type and γ1− mice (7 or 8 per group). Only cryoprecipitates from γ1− mice contained detectable Ig. f, IgG3 (brown) in glomerular capillaries (arrows) of γ1− mice 8 days after GaMD (top, low magnification; bottom, high magnification). Original magnification, ×400. *P < 0.05, **P < 0.005. ND, none detected. Error bars show s.e.m.
These observations suggested that GaMD-induced kidney disease might be caused by a qualitative change in the immune complexes in immunized γ1− mice. Consistent with this, IgG3, the dominant isotype produced in these mice, generates large immune complexes by self-associating through Fc–Fc interactions7,8; these large immune complexes tend to reversibly precipitate at reduced temperature (that is, they are cryoglobulins) and at increased concentration (which occurs as plasma undergoes ultrafiltration in glomeruli). Indeed, large cryoglobulin concentrations were found in plasma from GaMD-immunized γ1−, but not wild-type mice (Fig. 2d); cryoglobulin analysis demonstrated that IgG3 was the dominant mouse Ig constituent, although cryoglobulins also contained IgM (Fig. 2e). In keeping with this, deposits within glomerular capillaries were rich in IgG3 (Fig. 2f). A dominant role for IgM in this kidney disease model is unlikely because glomerular IgM, unlike glomerular IgG3, does not persist (Extended Data Fig. 5). Severe disease still develops in immunized mice deficient in both IgG1 and J chain (Extended Data Fig. 6), which produce little pentameric IgM12, and mice that lack activation-induced cytidine deaminase (AID) and consequently secrete only IgM do not develop kidney disease after GaMD immunization (data not shown).
A passive immunization model was used to further test the hypothesis that renal disease can be caused by IgG3–antigen immune complex precipitation in glomerular capillaries. Wild-type BALB/c mice were injected simultaneously with IgG3 anti-TNP monoclonal antibody intravenously and TNP-conjugated goat serum (TNP-GIgG) subcutaneously on days 0 and 1. These mice developed increased BUN, urine protein, leukocyte esterase and blood, and large deposits of amorphous material were observed in glomerular capillaries on day 2 (Fig. 3a–c and Extended Data Fig. 7a). Similar lesions developed in similarly treated C3-deficient mice (Fig. 3d and Extended Data Fig. 7b) and FcRγ-deficient mice, as well as in C57BL/6 mice and in BALB/c wild-type mice when TNP-conjugated bovine serum albumin (TNP-BSA) was substituted for TNP-GIgG (data not shown). Wild-type mice injected with TNP-GIgG plus IgG1, IgG2a or IgG2b anti-TNP monoclonal antibody failed to develop renal disease (Fig. 3a and Extended Data Fig. 7a). None of the monoclonal antibodies induced disease when injected without TNP (Fig. 3b and data not shown).
a, Wild-type (WT) mice (4 per group) were injected intravenously (i.v.) with 4 mg of mouse IgG1, IgG2a, IgG2b or IgG3 anti-TNP monoclonal antibody and subcutaneously (s.c.) with 100 µl of TNP-goat serum on days 0 and 1. Urine protein was measured before injections and on days 1 and 2. b, c, Wild-type mice (4 per group) were injected with mouse IgG3 anti-TNP monoclonal antibody plus or minus TNP-goat serum as in a. b, Day 2 mouse sera were analysed for BUN. c, Day 2 kidneys were stained with PAS (left, glomerulus from mouse that received only IgG3; right, glomerulus from mouse that received IgG3 plus TNP-goat serum). Representative of 3 mice per group. Original magnification, ×400. d, Urine protein for BALB/c wild-type and C3− mice (4 per group) injected i.v. with 4 mg of IgG3 anti-TNP monoclonal antibody and s.c. with 400 µl of TNP-goat serum on days 0 and 1. *P < 0.05.
The unique pathogenicity of IgG3 raised the possibility that the other IgG isotypes might be able to inhibit IgG3-mediated disease. Consistent with this, GaMD induced only transient renal disease in γ1+/− mice, which produced ∼50% as much IgG1 as wild-type (γ1+/+) mice, but similar amounts of IgG3 as γ1−/− mice (Extended Data Fig. 8). Similarly, development of proteinuria, hypoalbuminaemia and azotaemia in GaMD-immunized γ1−/− mice was suppressed by administration of the IgG1 anti-GIgG-rich serum from GaMD-immunized wild-type mice (GaMD immune WT serum). This suppression was antigen specific, because it was not observed with serum from rabbit anti-mouse IgD-immunized wild-type mice (RaMD immune WT serum) (Fig. 4a, b). Disease suppression by GaMD immune WT serum required initiation of treatment by day 5 after GaMD immunization (Extended Data Fig. 9a), when immunized mice first secrete IgG anti-GIgG. Importantly, injection of GaMD immune WT serum starting 4–5 days after GaMD immunization suppressed renal disease in γ1− mice without decreasing serum IgM, IgG2a or IgG3 levels and only modestly decreased production of any isotype by cultured spleen cells (Fig. 4c and Extended Data Fig. 9b). Thus, IgG1 primarily suppresses renal disease in our model by competing with IgG3 for antigen epitopes and/or changing the solubility of immune complexes rather than by decreasing IgG3 secretion; and the increased IgG3 secretion by GaMD-immunized γ1− mice results from blocked isotype switching rather than from a lack of IgG1.
a–f, BALB/c γ1− mice (5 per group) were injected with GaMD on day 0 and/or GaMD or rabbit anti-mouse IgD (RaMD) immune WT serum daily on days 4–7. a, b, Urine protein (a) and day 12 serum albumin and BUN levels (b). *P < 0.05, **P < 0.005. NS, not significant. c, BALB/c γ1− mice (5 per group) were injected with GaMD on day 0 ± GaMD immune WT serum daily on days 4–7 or 5–7. Spleens were harvested and cultured on day 8; 24 h culture supernatant IgG2a, IgG3 and IgM anti-goat IgG titres were determined. No anti-goat IgG antibody was detected in culture supernatants from non-immune spleen cells. d, Wild-type (WT) and C3-deficient (C3−) mice (4 per group) were injected s.c. with 100 µl of TNP-goat serum and i.v. with 4 mg of mouse IgG3 anti-TNP plus or minus 5 mg of IgG1 anti-TNP monoclonal antibody on days 0 and 1. Urine was analysed on day 0, 1 and 2. e, Wild-type and FcγRIIB-deficient (FcγRIIB−) mice (4 per group) were injected s.c. with 100 µl of TNP-goat serum and i.v. with 4 mg of IgG3 anti-TNP plus or minus 5 mg of IgG1 anti-TNP on days 0 and 1. Urine was analysed on days 0, 1 and 2. f, BALB/c mice were injected i.v. with 4 mg IgG3 anti-TNP and s.c. with 1.4 mg of TNP-BSA on days 0 and 1. Some mice were also injected with 0.625, 1.25, 2.5 or 5 mg of switch variant IgG1, IgG2a or IgG2b anti-TNP monoclonal antibodies on days 0 and 1. Urine protein was determined on day 0 (data not shown), day 1 (top) and day 2 (bottom). Results are pooled from a total of seven experiments. Group sizes were as follows. IgG3 alone: 19 mice; 0.625 mg of IgG1, IgG2a or IgG2b: 4 mice; 1.25 mg of IgG1, IgG2a or IgG2b: 8 mice; 2.5 mg of IgG1, IgG2a or IgG2b: 6 mice; 5 mg of IgG1, IgG2a or IgG2b: 8 or 9 mice. The significance of differences between treatment groups was determined with a normal regression model with the covariates: group; dose; and group–dose interaction. A t-test was used to evaluate the significance of the difference in least square means for each of these effects. This procedure was used to evaluate urine protein, measured at day 1 and day 2. A P value less than or equal to 0.05 after applying the Tukey adjustment for multiple comparisons was judged to be significant. *P < 0.05 as compared to IgG3 alone; †P < 0.05 as compared to IgG2b plus IgG3; ‡P < 0.05 as compared to IgG2a plus IgG3. Error bars show s.e.m.
Consistent with our conclusion that IgG1 suppresses IgG3-induced renal disease by competing with IgG3 for antigen binding and/or changing immune complex solubility, IgG1 anti-TNP monoclonal antibody suppressed glomerular IgG3 deposition and disordered renal function when mice were injected with IgG3 anti-TNP monoclonal antibody plus TNP-BSA or TNP-goat serum (Fig. 4d, e and Extended Data Fig. 10). Very little IgG1 was found in the glomeruli of mice injected with both isotypes (Extended Data Fig. 10b), suggesting that the presence of IgG1 in an immune complex with IgG3 prevents glomerular immune complex deposition and/or increases its clearance. IgG1 suppression of IgG3-associated renal disease did not depend on C3 or FcγRIIB (Fig. 4d, e and Extended Data Fig. 7b, c) and isotype control mouse IgG1 monoclonal antibody had no effect on IgG3-mediated disease (data not shown). IgG1 anti-TNP was more potent than IgG2a anti-TNP—and IgG2a anti-TNP was more potent than IgG2b anti-TNP—at preventing IgG3-mediated disease (Fig. 4f and Extended Data Fig. 7d), even though isotype switch variants of IgG1, IgG2a and IgG2b were used that had identical antigen-binding V regions13, avidity for TNP (Extended Data Fig. 7e) and non-specific binding to IgG3 (Extended Data Fig. 7f), and similar non-specific binding to themselves (Extended Data Fig. 7g). Preferential inhibition by IgG1 over IgG2a and IgG2b was also seen in studies with a second set of monoclonal antibodies that were not switch variants (data not shown).
The increased ability of IgG1 to inhibit IgG3-mediated renal disease may be a consequence of its short hinge region length and consequent low segmental flexibility. This may limit immune complex formation by decreasing the ability of IgG1 to bind bivalently to a ligand and by increasing the likelihood that it will sterically block binding of IgG3 (refs 14, 15), which could separate IgG3 molecules sufficiently to inhibit their self-association. Consistent with this possibility, IgG2a, which has a hinge region length and segmental flexibility that are intermediate between IgG1 and IgG2b14, had an intermediate ability to suppress IgG3-mediated renal disease (Fig. 4f and Extended Data Fig. 7d). Thus, IgG1 may limit antibody-mediated disease in our model by suppressing the formation of immune complexes that become insoluble when they are concentrated by glomerular filtration. We cannot, however, eliminate the possibility that the addition of IgG1 to IgG3–antigen immune complexes facilitates their elimination by the reticulo-endothelial system, which could limit nephrotoxicity.
Our observations lead us to two important conclusions. First, we show that immune complexes can destroy kidney function by precipitating in glomerular capillaries, even in the absence of complement and FcR activation. The rapidity of capillary obstruction and the lack of an anaphylatoxin gradient in our model, as well as the ability of complement to increase immune complex solubility and elimination16, may explain the failure of complement to exacerbate disease despite its deposition in glomeruli. Second, we show that Ig isotypes that poorly activate effector mechanisms protect against disease caused by more proinflammatory isotypes. In this regard, isotypes such as mouse IgG1 and human IgG4 seem to act like partial agonists, which can cause immunopathology under some conditions, but prevent it by blocking the effects of other, more proinflammatory isotypes, in other circumstances. Indeed, functional similarities between mouse IgG1 and human IgG4 suggest that our observations in mice are applicable to humans. Mouse IgG1-mediated protection against immune complex deposition in our model is probably facilitated by its short hinge region, which limits antigen crosslinking by decreasing segmental flexibility14. Human IgG4 is likely to have an even greater ability to suppress immune complex development because, in addition to its short hinge region14, its labile inter-heavy-chain disulphide bond allows it to dissociate into univalent half molecules17.
Similarities between mouse IgG1 and human IgG4 in their ability to suppress disease caused by other isotypes may extend further. Although not shown in this paper, our preliminary observations, with additional collaborators, demonstrate that the absence of IgG1 promotes the development and severity of complement- and FcγR-mediated diseases in mice, including collagen-induced arthritis and experimental myasthenia gravis. Thus, mouse IgG1 probably suppresses disease mediated by complement and FcγRs, as well as disease mediated by excessive intravascular formation of insoluble immune complexes. The inability of human IgG4 to activate complement2,3 and its poor binding to FcγRs4 suggest that it can similarly limit organ damage in complement- and FcγR-mediated diseases. These considerations raise the possibility of using human IgG4 antibodies to suppress autoimmune and immune complex disorders that are mediated by other isotypes, an approach that might be amplified by making IgG4 antibodies even more immunosuppressive by increasing their sialylation18, galactosylation19 and/or affinity for FcγRIIB20.
Methods
Mice
All mice were bred and maintained in the specific pathogen-free (SPF) facility at the Cincinnati Children’s Research Foundation and all experiments were done with the approval of and in accordance with regulatory guidelines and standards set by the Institutional Animal Care and Use Committee of Cincinnati Children’s Hospital Medical Center. Male and female mice were used between the ages of 8 and 20 weeks. BALB/c and/or C57BL/6 background γ1-deficient mice5, FcRγ-deficient (Taconic), C3-deficient mice (a gift from M. Wills-Karp), J-chain deficient mice (a gift from D. Metzger), FcγRIIB-deficient, AID-deficient (a gift from M. Muramatsu)21 and wild-type control mice were bred in our colony. Double and triple gene-deficient mice, made by crossbreeding the single gene-deficient mice, were typed by PCR. Typing for γ1 and C3 deficiency was confirmed by gel double diffusion assay of serum. Wild-type littermates of the double and triple gene-deficient mice were used as controls. Mice of the appropriate genotype were randomly assigned to groups, but a specific randomization program was not used. Studies were not blinded.
Active model for induction of immune complex renal disease
Mice were injected s.c. with 0.2 ml (BALB/c) or 0.4 ml (C57BL/6) of GaMD on day 0. In some experiments mice were also injected intraperitoneally (i.p.) daily with pooled day 12 serum from GaMD or RaMD immunized WT mice (GaMD immune WT serum and RaMD immune WT serum, respectively). Spontaneously micturated urine was collected from mice on specific days and assayed for protein, leukocyte esterase and blood content by urine dipstick. Serum was collected by tail vein puncture and kidneys were preserved in formalin or gluteraldehyde or frozen in OCT for histological evaluation.
Passive model for immune complex induction of renal disease
Mice were simultaneously injected with mouse IgG3 anti-TNP monoclonal antibody i.v. and TNP-goat serum or TNP-BSA s.c. on days 0 and 1. Some mice also received mouse IgG1, IgG2a or IgG2b anti-TNP monoclonal antibody isotype switch variants or in some cases non-switch variant monoclonal antibodies i.v. on days 0 and 1.
Reagents
Hybridomas were obtained from the following sources: 9A6 (mouse IgG3 anti-TNP monoclonal antibody), a gift from S. Izui22; 1B7.11 (mouse IgG1 anti-TNP monoclonal antibody), from the American Type Culture Collection; HY1.2 and C1040 (mouse IgG2a anti-TNP monoclonal antibodies) and GORK (mouse IgG2b anti-TNP monoclonal antibody), a gift from B. Heyman; switch variant mouse IgG1, IgG2a and IgG2b anti-TNP monoclonal antibodies, a gift from M. Robson23; and XMG-6 (rat IgG1 anti-mouse IFN-γ)24, from DNAX. A hybridoma that secretes mouse IgG1 anti-FITC monoclonal antibody was produced in house. Hybridomas were grown as ascites in Pristane-primed athymic nude mice and monoclonal antibodies were purified from ascites by ammonium sulphate precipitation (25–50% for all IgGs except 20–30% for IgG3), followed by DE-52 cation exchange chromatography for the IgG isotypes. IgG2a anti-TNP monoclonal antibody was also purchased from Bio X Cell. GaMD and RaMD antisera were made as described25. Mouse hyperimmune antisera to goat (GaMD immune WT serum) and rabbit (RaMD immune WT serum) were made by injecting wild-type mice s.c. every 14 days for several injections with GaMD or RaMD, respectively, and pooling serum collected 10–12 days after each immunization. Non-immune mouse serum was collected by tail vein bleeding from untreated BALB/c or C57BL/6 wild-type mice and pooled. The C5aR antagonists JPE 1275 (ref. 26) (a gift from J. Lambris) and A8δ71-73 (ref. 27) were injected as 20 µg or 10 µM doses, respectively, i.p. every 12 h starting on day 4 and ending on day 8 after GaMD immunization.
Urine and serum measurements
Urine protein, leukocyte esterase and erythrocyte levels were measured on fresh, freely excreted urine using Multistik 10 (Becton-Dickson). Measurements were on a colorimetric scale, ranging from 0 to 4 for protein and 0 to 3 for leukocyte esterase and erythrocytes, as per the manufacturer’s instructions. Serum albumin and BUN were measured using a Beckman DXC courtesy of R. Louderbeck, Veterans Administration Medical Center, Cincinnati, Ohio. Serum, splenic supernatants and re-suspended cryoglobulin pellets were analysed for total and GIgG-specific IgG1, IgG2a/c, IgG2b, IgG3, IgE, IgA and IgM content, using standard sandwich ELISA with paired anti-Ig isotype monoclonal antibodies for each Ig isotype (BD-Pharmingen and eBioscience). Sera, splenic supernatants and re-suspended cryoglobulin solutions were titred for GIgG-specific antibody levels by ELISA, as previously described25, using wells coated with 5 μg ml−1 of goat IgG. Gel double diffusion was used to identify mouse IgG1, mouse C3 and GIgG in mouse serum with antibodies purchased from Bethyl.
TNP-goat serum and TNP-BSA
Goat serum or BSA were conjugated to TNP as previously described28.
Immunofluorescence microscopy
Kidneys were harvested from mice and immediately placed in OCT and snap frozen in liquid nitrogen. OCT-embedded kidneys were stored at −80 °C. Frozen tissue sections were cut, mounted on glass slides, fixed in acetone and air-dried. After rehydration and blocking, immunofluorescence microscopy was performed with FITC-labelled anti-C3 and anti-mouse IgG antibodies (ICN Biomedicals/Cappel). After washing, coverslips were applied to slides after addition of anti-fade medium that contained 4′,6-diamidino-2-phenylindole (DAPI) (Prolong Gold; Invitrogen). Slides were assessed microscopically and photographed at an original magnification of ×400 using an RT Slider digital camera (Diagnostic Instruments) mounted on an E600 fluorescent microscope (Nikon Instruments).
Immunostaining microscopy
Kidneys were harvested from mice and immediately placed in formalin for a minimum of 3 days before embedding in paraffin. For IgG3 staining, de-paraffinized sections were incubated with goat anti-mouse IgG3 antibody (Jackson ImmunoResearch Laboratories) for 1 h, then incubated for 12 min with biotin-donkey anti-goat IgG antibody (Jackson ImmunoResearch Laboratories) and visualized with an iVIEW Plus Detection Kit (Ventana Medical Systems). Staining for IgG1 was performed with a rabbit anti-mouse IgG1 antibody (Novus Biologicals), followed by biotin-donkey anti-rabbit antibody (Jackson ImmunoResearch Laboratories). All antibodies were used at 1:100 dilutions and all staining was performed with a Discovery XT (Ventana Medical Systems).
Quantitation of splenic Ig production
Spleens were diced into 1–2 mm cubes, washed in ice-cold PBS, then transferred to a Petri dish with 5 ml of RPMI medium 1640 supplemented with fetal bovine serum, penicillin, gentamicin, HEPES, sodium pyruvate, essential amino acids and 2-mercaptoethanol and cultured at 37 °C, 5% CO2 for 24 h. Supernatants were collected, separated into aliquots, frozen and stored at −80 °C until analysed.
Cryoprecipitant collection
Blood was collected in a polystyrene centrifuge tube and immediately incubated for 4 h at 37 °C. The tube was centrifuged and solid material was removed. Sera were incubated at 4 °C for 7 days. Precipitates were obtained by centrifugation, washed 3 times in ice-cold saline, then re-suspended in a volume of saline equal to that of the initial serum sample and heated to 37 °C for 2 h to dissolve cryoprecipitates. Samples were then re-centrifuged at room temperature and supernatants were collected.
Anti-TNP monoclonal antibody ELISA
For avidity measurement ELISA plates were coated with 10 µg TNP-OVA per ml overnight followed by varying concentrations of the monoclonal antibodies. This was followed by horseradish peroxidase (HRP)- or biotin-labelled anti-mouse Ig isotype-specific monoclonal antibody purchased from BD bioscience, streptavidin-HRP if needed and SuperSignal ELISA substrate from Pierce. For IgG3 binding or self-association measurement, ELISA plates were coated with 10 µg mouse IgG1, IgG2a, IgG2b or IgG3 anti-TNP monoclonal antibody per ml overnight followed by varying concentrations of biotin-labelled mouse IgG1, IgG2a, IgG2b or IgG3 anti-TNP monoclonal antibodies. This was followed by streptavidin-HRP and SuperSignal substrate from Pierce.
Statistics
The non-parametric Mann–Whitney two-tailed t-test (GraphPad Prism 5.0; GraphPad Software) was used to compare Ig levels, BUN and albumin concentrations between different groups of mice. A P value < 0.05 was considered significant. A more complex test was used to compare the multiple samples in Fig. 4f and Extended Data Fig. 7d (see figure legends for details).
Sample size was calculated with a tool for comparing two independent samples on the website http://www.stat.ubc.ca/~rollin/stats/ssize/n2.html. Sample size calculations for initial studies were based on the assumptions that a one-sided test would be used to test the hypothesis that the mean for the normal (usually wild-type) group would be three times as large (or one-third as large) as the mean for the abnormal (usually γ1-deficient) group, that the common standard deviation would be the same size as the smaller mean, that the type I error rate would be 0.05 and that the desired power would be 0.80. These assumptions suggested a sample size of 4 for each sample. In practice, we often used the results of our initial studies to determine whether this sample size was sufficient to yield the desired power or indicate the larger sample size that would yield a significant result if the trend observed in the initial study predicted the results obtained with the larger sample size.
Change history
21 January 2015
Spelling of author S.K.S. was corrected.
References
Nimmerjahn, F. & Ravetch, J. V. Fcγ receptors as regulators of immune responses. Nature Rev. Immunol. 8, 34–47 (2008)
Barrington, R., Zhang, M., Fischer, M. & Carroll, M. C. The role of complement in inflammation and adaptive immunity. Immunol. Rev. 180, 5–15 (2001)
Snapper, C. & Finkelman, F. in Fundamental Immunology (ed. Paul, W. E. ) 837–863 (Lippincott-Raven, 1999)
Bruhns, P. et al. Specificity and affinity of human Fcγ receptors and their polymorphic variants for human IgG subclasses. Blood 113, 3716–3725 (2009)
Jung, S., Rajewsky, K. & Radbruch, A. Shutdown of class switch recombination by deletion of a switch region control element. Science 259, 984–987 (1993)
Roccatello, D. et al. Multicenter study on hepatitis C virus-related cryoglobulinemic glomerulonephritis. Am. J. Kidney Dis. 49, 69–82 (2007)
Nishimura, Y. & Nakamura, H. Human monoclonal cryoimmunoglobulins. I. Molecular properties of IgG3κ (Jir protein) and the cryo-coprecipitability of its molecular fragments by papain. J. Biochem. 95, 255–265 (1984)
Abdelmoula, M. et al. IgG3 is the major source of cryoglobulins in mice. J. Immunol. 143, 526–532 (1989)
Finkelman, F. D. et al. Polyclonal activation of the murine immune system by an antibody to IgD. II. Generation of polyclonal antibody production and cells with surface IgG. J. Immunol. 129, 638–646 (1982)
Ravetch, J. V. & Bolland, S. IgG Fc receptors. Annu. Rev. Immunol. 19, 275–290 (2001)
Finkelman, F. D., Katona, I. M., Mosmann, T. R. & Coffman, R. L. IFN-γ regulates the isotypes of Ig secreted during in vivo humoral immune responses. J. Immunol. 140, 1022–1027 (1988)
Erlandsson, L., Andersson, K., Sigvardsson, M., Lycke, N. & Leanderson, T. Mice with an inactivated joining chain locus have perturbed IgM secretion. Eur. J. Immunol. 28, 2355–2365 (1998)
Boot, J. H., Geerts, M. E., De Groot, E. R. & Aarden, L. A. Murine monoclonal isotype switch variants. Detection with rat monoclonal antibodies in ELISA and isolation by sequential sublining. J. Immunol. Methods 106, 195–202 (1988)
Dangl, J. L. et al. Segmental flexibility and complement fixation of genetically engineered chimeric human, rabbit and mouse antibodies. EMBO J. 7, 1989–1994 (1988)
Schneider, W. P., Wensel, T. G., Stryer, L. & Oi, V. T. Genetically engineered immunoglobulins reveal structural features controlling segmental flexibility. Proc. Natl Acad. Sci. USA 85, 2509–2513 (1988)
Sheerin, N. S., Springall, T., Abe, K. & Sacks, S. H. Protection and injury: the differing roles of complement in the development of glomerular injury. Eur. J. Immunol. 31, 1255–1260 (2001)
van der Neut Kolfschoten, M. et al. Anti-inflammatory activity of human IgG4 antibodies by dynamic Fab arm exchange. Science 317, 1554–1557 (2007)
Anthony, R. M., Kobayashi, T., Wermeling, F. & Ravetch, J. V. Intravenous gammaglobulin suppresses inflammation through a novel TH2 pathway. Nature 475, 110–113 (2011)
Karsten, C. M. et al. Anti-inflammatory activity of IgG1 mediated by Fc galactosylation and association of FcγRIIB and dectin-1. Nature Med. 18, 1401–1406 (2012)
Chu, S. Y. et al. Reduction of total IgE by targeted coengagement of IgE B-cell receptor and FcγRIIb with Fc-engineered antibody. J. Allergy Clin. Immunol. 129, 1102–1115 (2012)
Muramatsu, M. et al. Class switch recombination and hypermutation require activation-induced cytidine deaminase (AID), a potential RNA editing enzyme. Cell 102, 553–563 (2000)
Fulpius, T. et al. Glomerulopathy induced by IgG3 anti-trinitrophenyl monoclonal cryoglobulins derived from non-autoimmune mice. Kidney Int. 45, 962–971 (1994)
Giorgini, A. et al. FcγRIII and FcγRIV are indispensable for acute glomerular inflammation induced by switch variant monoclonal antibodies. J. Immunol. 181, 8745–8752 (2008)
Finkelman, F. D., Katona, I. M., Mosmann, T. R. & Coffman, R. L. IFN-γ regulates the isotypes of Ig secreted during in vivo humoral immune responses. J. Immunol. 140, 1022–1027 (1988)
Finkelman, F. D. et al. Polyclonal activation of the murine immune system by an antibody to IgD. II. Generation of polyclonal antibody production and cells with surface IgG. J. Immunol. 129, 638–646 (1982)
Boor, P. et al. Complement C5 mediates experimental tubulointerstitial fibrosis. J. Am. Soc. Nephrol. 18, 1508–1515 (2007)
Köhl, J. et al. A regulatory role for the C5a anaphylatoxin in type 2 immunity in asthma. J. Clin. Invest. 116, 783–796 (2006)
Strait, R. T., Morris, S. C. & Finkelman, F. D. IgG-blocking antibodies inhibit IgE-mediated anaphylaxis in vivo through both antigen interception and FcγRIIb cross-linking. J. Clin. Invest. 116, 833–841 (2006)
Acknowledgements
We thank M. Wills-Karp for C3-deficient mice, D. Metzger for J-chain-deficient mice, M. Muramatsu for AID-deficient mice, S. Izui for a hybridoma that secretes mouse IgG1 anti-TNP monoclonal antibody, M. Robson and L. Aarden for switch variant hybridomas that secrete mouse IgG1, IgG2a and IgG2b anti-TNP monoclonal antibodies, B. DiPasquale for histological staining, and J. Lambris for C5aR antagonist. Research was supported by a US Department of Veterans Affairs Merit Award, National Institutes of Health R01 AI072040, and the University of Cincinnati and Cincinnati Children’s Hospital, all to F.D.F.
Author information
Authors and Affiliations
Contributions
R.T.S. planned, performed, interpreted and directed all experiments and participated in all drafts of the manuscript; M.K., A.M. and N.B. performed experiments; C.O.J. provided mice; J.K. provided mice and reagents and participated in planning of all complement-related studies; M.E. participated in the planning and interpretation of isotype switch variant studies; S.K.S., K.S. and D.W. performed and interpreted histological studies; M.T.P. and A.B.H. contributed to investigation of the importance of antibody hinge region length; M.M.H. performed statistical analyses; and F.D.F. designed the study, analysed data and wrote the initial draft of the paper. All authors discussed the results and commented on the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Extended data figures and tables
Extended Data Figure 1 GaMD immunization of γ1− mice induces renal dysfunction and glomerular deposition of PAS+ material that includes IgG and complement.
a, Wild-type (WT) and γ1− mice (4 per group) were immunized with GaMD. Urine leukocyte esterase and blood were obtained. b, Representative photomicrographs of glomeruli stained for C3 (top) or total mouse IgG (bottom) from wild-type (right) and γ1−/− mice (left), 12 days after GaMD immunization (3 mice per group). c, Deposition of amorphous PAS+ material in glomeruli of γ1−, but not wild type begins ∼7 days after GaMD immunization and leads to glomerular destruction by day 9. Note the scarcity of inflammatory cells in glomeruli. Representative data from 6 mice per group. Original magnification, ×400 for b, ×200 for c.
Extended Data Figure 2 The development of kidney disease in GaMD-immunized γ1− mice is independent of IFN-γ, IgG2a, C3 and FcRγ.
a–d, BALB/c wild-type (WT) and γ1− mice (5 per group) were immunized with GaMD on day 0 and injected with 1 mg of either anti-IFN-γ or control monoclonal antibody (mAb) on days 0 and 5. a, Total levels of all Ig isotypes were determined in 24 h culture supernatants of spleen cells harvested on the days shown. b, GaMD-immunized γ1−, γ1−/FcRγ−, γ1−/C3−, C3−/FcRγ− and γ1−/C3−/FcγR− mice (5 per group) had their urine tested for leukocyte esterase and blood on the days shown. c, d, BALB/c wild-type and γ1− mice (5 per group) were immunized with GaMD on day 0 and injected with 1 mg of either anti-IFN-γ or control monoclonal antibody on days 0 and 5. c, Urine obtained on days indicated was assayed for protein, leukocyte esterase and blood. d, BUN levels were determined before and 10 days after GaMD immunization. *P < 0.05.
Extended Data Figure 3 Neither complement nor stimulatory FcRs are required for renal disease development in GaMD-immunized γ1− mice.
Mixed background γ1− and γ1−/C3−/FcRγ− mice (4 per group) were immunized with GaMD plus or minus C5aR antagonist. Urinalyses were obtained at baseline and daily starting on day 6. LE, leukocyte esterase.
Extended Data Figure 4 Delayed antigen elimination does not account for renal disease in GaMD-immunized γ1− mice.
a, BALB/c wild-type (WT) and γ1− mice (10 per group) were immunized s.c. with GaMD. Sera obtained 5, 6, 7 and 9 days later were evaluated by gel double diffusion for the presence of goat IgG. b–e, BALB/c wild-type mice (4 or 5 per group) were injected s.c. with a total of 0.2 ml of different mixtures of GaMD and goat anti-KLH antisera. b, c, Mouse sera collected 9 days later were assayed for BUN (b) and IgG1 anti-goat IgG antibody (c). d, Sera obtained 6–13 days post-immunization were evaluated by gel double diffusion for the presence of goat IgG. e, Urine samples collected 4–12 days post-immunization were analysed for protein. *P < 0.05, **P < 0.005.
Extended Data Figure 5 IgG3 immune complexes persist and accumulate in the glomeruli of GaMD-immunized γ1− mice.
BALB/c wild-type (WT) and γ1− mice were left untreated or were immunized with GaMD. Kidney sections were stained for mouse IgG1, IgG2a, IgG2b, IgG3 and IgM and 12 days after immunization. Representative photomicrographs from three GaMD-immunized mice are shown. Insets show magnified views. No staining was observed with sections from unimmunized mice (data not shown). Original magnification, ×400.
Extended Data Figure 6 Severe renal disease develops in GaMD-immunized γ−/J-chain− mice.
a–c, BALB/c γ1− (12 mice), J-chain− (9 mice) and γ1−/J-chain− (12 mice) mice were injected s.c. with GaMD. a, Urinalysis was performed on the indicated days. b, BUN levels on day 0 and 11. The difference between γ1− and γ1− × J-chain− mice was not consistently observed. c, Survival of GaMD-immunized mice. *P < 0.05.
Extended Data Figure 7 IgG1 inhibits IgG3-induced cryoglobulin kidney disease independent of complement and FcγRIIB and better than IgG2a and IgG2b.
a, Wild-type (WT) mice (4 per group) were injected i.v. with 4 mg of mouse IgG1, IgG2a, IgG2b or IgG3 anti-TNP monoclonal antibody and s.c. with 100 µl of TNP-goat serum on days 0 and 1. Urine leukocyte esterase and blood was measured before injections and on day 1 and day 2. b, Urine leukocyte esterase (LE) and blood for BALB/c wild-type and C3− mice (4 per group) injected i.v. with 4 mg of IgG3 anti-TNP monoclonal antibody and s.c. with 400 µl of TNP-goat serum on day 0 and day 1. c, Wild-type and FcγRIIB-deficient (FcγRIIB−) mice (4 per group) were injected s.c with 100 µl of TNP-goat serum and i.v. with 4 mg of IgG3 anti-TNP plus or minus 5 mg of IgG1 anti-TNP on day 0 and day 1. Urinalysis on days 0, 1 and 2. d, BALB/c mice were injected i.v. with 4 mg of IgG3 anti-TNP and s.c. with 1.4 mg of TNP-BSA on days 0 and 1. Some mice were also injected with 0.625, 1.25, 2.5 or 5 mg of switch variants of IgG1, IgG2a or IgG2b anti-TNP monoclonal antibodies on days 0 and 1. Urine protein was determined on day 0 (data not shown), day 1 (top) and day 2 (bottom). Results are pooled from a total of seven experiments. Group size was as follows. IgG3 alone: 19 mice; 0.625 mg of IgG1, IgG2a or IgG2b: 4 mice; 1.25 mg of IgG1, IgG2a or IgG2b: 8 mice; 2.5 mg of IgG1, IgG2a or IgG2b: 6 mice; 5 mg of IgG1, IgG2a or IgG2b: 8 or 9 mice. The significance of differences between treatment groups was determined as described in the legend to Fig. 4f. *P < 0.05 as compared to IgG3 alone; †P < 0.05 as compared to IgG2b plus IgG3; e, Binding of the Ig isotype switch variants to ELISA wells coated with TNP-BSA, reported as percentage of maximal binding. f, Binding of the Ig isotype switch variants to ELISA wells coated with IgG3 anti-TNP monoclonal antibody. g, Binding of IgG3 and the Ig isotype switch variants to ELISA wells coated with themselves.
Extended Data Figure 8 GaMD-immunized γ1+/− mice generate large IgG3 responses but develop mild renal disease.
a, b, BALB/c mice homozygous (γ1+/+), heterozygous (γ1+/−) and null (γ1−/−) for a functional γ1 allele (6 per group) were injected s.c. with GaMD. a, Sera were titred for goat IgG-specific IgG1, IgG2a and IgG3 0, 8 and 12 days later. Day 0 titres were zero for all Ig isotypes (data not shown). b, Urine samples from the same mice were assayed for protein and leukocyte esterase. ND, none detected.
Extended Data Figure 9 GaMD immune serum from wild-type mice inhibits GaMD-induced renal disease without decreasing other isotypes if injected into GaMD-immunized γ1− mice by 5 days after immunization.
a, BALB/c γ1− mice (4 or 8 per group) were injected s.c. with GaMD on day 0 and i.p. with 0.5 ml of pooled serum from GaMD-immunized wild-type mice (GaMD immune WT serum) or unimmunized wild-type mice (non-immune serum), starting 4, 5 or 6 days after GaMD immunization. Day 7 urine samples were analysed. LE, leukocyte esterase; ND, none detected. b, BALB/c γ1− mice (4 or 8 per group) were injected s.c. with GaMD on day 0 and i.p. with 0.5 ml of pooled serum from GaMD-immunized wild-type mice (GaMD immune WT serum) or unimmunized wild-type mice (non-immune serum), 5, 6 and 7 days after GaMD immunization. Sera were assayed for total IgG1, IgG2a, IgM and IgG3 on day 0 (unimmunized) and 8 days after GaMD immunization. ND, none detected. *P < 0.05, **P < 0.005 (both as compared to day 6 only in a and unimmunized in b).
Extended Data Figure 10 Antigen-specific IgG1 can prevent IgG3 immune complex glomerular deposition.
a, b, BALB/c wild-type (WT) mice were injected i.v. with mouse IgG1 and/or IgG3 anti-TNP monoclonal antibody with or without s.c. injection of TNP-BSA on days 0 and 1. a, Kidneys were stained with PAS on day 2. Representative micrographs from 3 mice per group are shown. b, Kidney serial sections were stained with PAS or for IgG3 or IgG1 (brown pigment). Representative micrographs from 4 mice per group are shown. Original magnification, ×200 for a, b.
Rights and permissions
About this article
Cite this article
Strait, R., Posgai, M., Mahler, A. et al. IgG1 protects against renal disease in a mouse model of cryoglobulinaemia. Nature 517, 501–504 (2015). https://doi.org/10.1038/nature13868
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature13868
This article is cited by
-
IgG subclasses in cryoglobulins: link to composition and clinical manifestations
Arthritis Research & Therapy (2020)
-
The complement system as a potential therapeutic target in rheumatic disease
Nature Reviews Rheumatology (2017)
-
IgG subclass co‐expression brings harmony to the quartet model of murine IgG function
Immunology & Cell Biology (2016)
-
Agglutinating mouse IgG3 compares favourably with IgMs in typing of the blood group B antigen: Functionality and stability studies
Scientific Reports (2016)
-
Erratum: IgG1 protects against renal disease in a mouse model of cryoglobulinaemia
Nature (2015)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.