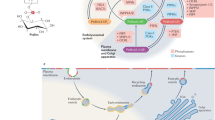
Abstract
The lipid composition of cellular organelles is tailored to suit their specialized tasks. A fundamental transition in the lipid landscape divides the secretory pathway in early and late membrane territories, allowing an adaptation from biogenic to barrier functions. Defending the contrasting features of these territories against erosion by vesicular traffic poses a major logistical problem. To this end, cells evolved a network of lipid composition sensors and pipelines along which lipids are moved by non-vesicular mechanisms. We review recent insights into the molecular basis of this regulatory network and consider examples in which malfunction of its components leads to system failure and disease.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Wenk, M. R. The emerging field of lipidomics. Nature Rev. Drug Discov. 4, 594–610 (2005).
Shevchenko, A. & Simons, K. Lipidomics: coming to grips with lipid diversity. Nature Rev. Mol. Cell Biol. 11, 593–598 (2010).
Sharpe, H. J., Stevens, T. J. & Munro, S. A comprehensive comparison of transmembrane domains reveals organelle-specific properties. Cell 142, 158–169 (2010). This comprehensive computational analysis reveals that membrane proteins show dichotomy in TMD length and composition that correlates with major differences in lipid composition and asymmetry between early and late secretory organelles.
Bigay, J. & Antonny, B. Curvature, lipid packing, and electrostatics of membrane organelles: defining cellular territories in determining specificity. Dev. Cell 23, 886–895 (2012).
Mesmin, B., Antonny, B. & Drin, G. Insights into the mechanisms of sterol transport between organelles. Cell. Mol. Life Sci. 70, 3405–3421 (2013).
de Kroon, A. I. P. M., Rijken, P. J. & De Smet, C. H. Checks and balances in membrane phospholipid class and acyl chain homeostasis, the yeast perspective. Prog. Lipid Res. 52, 374–394 (2013).
Koynova, R. & Caffrey, M. Phases and phase transitions of the phosphatidylcholines. Biochim. Biophys. Acta 1376, 91–145 (1998).
Marsh, D. Lateral pressure profile, spontaneous curvature frustration, and the incorporation and conformation of proteins in membranes. Biophys. J. 93, 3884–3899 (2007).
Frolov, V. A., Shnyrova, A. V. & Zimmerberg, J. Lipid polymorphisms and membrane shape. Cold Spring Harb. Perspect. Biol. 3, a004747 (2011).
Magalhaes, M. A. O. & Glogauer, M. Pivotal advance: phospholipids determine net membrane surface charge resulting in differential localization of active Rac1 and Rac2. J. Leukoc. Biol. 87, 545–555 (2010).
Grinstein, S. Imaging signal transduction during phagocytosis: phospholipids, surface charge, and electrostatic interactions. Am. J. Physiol. Cell Physiol. 299, C876–C881 (2010).
van Meer, G., Voelker, D. R. & Feigenson, G. W. Membrane lipids: where they are and how they behave. Nature Rev. Mol. Cell Biol. 9, 112–124 (2008).
Slotte, J. P. Biological functions of sphingomyelins. Prog. Lipid Res. 52, 424–437 (2013).
Koynova, R. & Caffrey, M. Phases and phase transitions of the sphingolipids. Biochim. Biophys. Acta 1255, 213–236 (1995).
Yeagle, P. The Membranes of Cells (Academic, 1993).
Brown, D. A. & London, E. Structure and origin of ordered lipid domains in biological membranes. J. Membr. Biol. 164, 103–114 (1998).
Boumann, H. A. et al. Depletion of phosphatidylcholine in yeast induces shortening and increased saturation of the lipid acyl chains: evidence for regulation of intrinsic membrane curvature in a eukaryote. Mol. Biol. Cell 17, 1006–1017 (2006).
Hikiji, T., Miura, K., Kiyono, K., Shibuya, I. & Ohta, A. Disruption of the CHO1 gene encoding phosphatidylserine synthase in Saccharomyces cerevisiae. J. Biochem. 104, 894–900 (1988).
Lester, R. L., Wells, G. B., Oxford, G. & Dickson, R. C. Mutant strains of Saccharomyces cerevisiae lacking sphingolipids synthesize novel inositol glycerophospholipids that mimic sphingolipid structures. J. Biol. Chem. 268, 845–856 (1993).
Park, E. & Rapoport, T. A. Mechanisms of Sec61/SecY-mediated protein translocation across membranes. Annu. Rev. Biophys. 41, 21–40 (2012).
Radhakrishnan, A., Goldstein, J. L., McDonald, J. G. & Brown, M. S. Switch-like control of SREBP-2 transport triggered by small changes in ER cholesterol: a delicate balance. Cell Metab. 8, 512–521 (2008). This study determined the narrow range within which ER cholesterol levels control SREBP–SCAP export from the ER through cooperative interactions between cholesterol, SCAP and INSIG.
Holthuis, J. C., Pomorski, T., Raggers, R. J., Sprong, H. & Van Meer, G. The organizing potential of sphingolipids in intracellular membrane transport. Physiol. Rev. 81, 1689–1723 (2001).
Schneiter, R. et al. Electrospray ionization tandem mass spectrometry (ESI-MS/MS) analysis of the lipid molecular species composition of yeast subcellular membranes reveals acyl chain-based sorting/remodeling of distinct molecular species en route to the plasma membrane. J. Cell Biol. 146, 741–754 (1999).
Slotte, J. P. & Bierman, E. L. Depletion of plasma-membrane sphingomyelin rapidly alters the distribution of cholesterol between plasma membranes and intracellular cholesterol pools in cultured fibroblasts. Biochem. J. 250, 653–658 (1988).
Brügger, B. et al. Evidence for segregation of sphingomyelin and cholesterol during formation of COPI-coated vesicles. J. Cell Biol. 151, 507–518 (2000).
Munro, S. An investigation of the role of transmembrane domains in Golgi protein retention. EMBO J. 14, 4695–4704 (1995).
Quiroga, R., Trenchi, A., González Montoro, A., Valdez Taubas, J. & Maccioni, H. J. F. Short transmembrane domains with high-volume exoplasmic halves determine retention of Type II membrane proteins in the Golgi complex. J. Cell Sci. 126, 5344–5349 (2013).
Kaiser, H.-J. et al. Lateral sorting in model membranes by cholesterol-mediated hydrophobic matching. Proc. Natl Acad. Sci. USA 108, 16628–16633 (2011).
Fairn, G. D. et al. High-resolution mapping reveals topologically distinct cellular pools of phosphatidylserine. J. Cell Biol. 194, 257–275 (2011). This important study provides quantitative information on the transbilayer distribution of PtdSer in early and late secretory organelles with the aid of a genetically encoded lipid probe.
Sanyal, S. & Menon, A. K. Flipping lipids: why an' what's the reason for? ACS Chem. Biol. 4, 895–909 (2009).
Bartz, R. et al. Lipidomics reveals that adiposomes store ether lipids and mediate phospholipid traffic. J. Lipid Res. 48, 837–847 (2007).
Natarajan, P., Wang, J., Hua, Z. & Graham, T. R. Drs2p-coupled aminophospholipid translocase activity in yeast Golgi membranes and relationship to in vivo function. Proc. Natl Acad. Sci. USA 101, 10614–10619 (2004).
Alder-Baerens, N., Lisman, Q., Luong, L., Pomorski, T. & Holthuis, J. C. Loss of P4 ATPases Drs2p and Dnf3p disrupts aminophospholipid transport and asymmetry in yeast post-Golgi secretory vesicles. Mol. Biol. Cell 17, 1632–1642 (2006).
Kim, Y. J., Guzman-Hernandez, M. L. & Balla, T. A highly dynamic ER-derived phosphatidylinositol-synthesizing organelle supplies phosphoinositides to cellular membranes. Dev. Cell 21, 813–824 (2011).
Vanni, S. et al. Amphipathic lipid packing sensor motifs: probing bilayer defects with hydrophobic residues. Biophys. J. 104, 575–584 (2013).
Levine, T. Short-range intracellular trafficking of small molecules across endoplasmic reticulum junctions. Trends Cell Biol. 14, 483–490 (2004).
Lev, S. Non-vesicular lipid transport by lipid-transfer proteins and beyond. Nature Rev. Mol. Cell Biol. 11, 739–750 (2010).
Diakogiannaki, E., Welters, H. J. & Morgan, N. G. Differential regulation of the endoplasmic reticulum stress response in pancreatic β-cells exposed to long-chain saturated and monounsaturated fatty acids. J. Endocrinol. 197, 553–563 (2008).
Deguil, J. et al. Modulation of lipid-induced ER stress by fatty acid shape. Traffic 12, 349–362 (2011).
Cybulski, L. E., Martín, M., Mansilla, M. C., Fernández, A. & de Mendoza, D. Membrane thickness cue for cold sensing in a bacterium. Curr. Biol. 20, 1539–1544 (2010).
Inda, M. E. et al. A lipid-mediated conformational switch modulates the thermosensing activity of DesK. Proc. Natl Acad. Sci. USA 111, 3579–3584 (2014). This elegant study revealed the molecular principles by which a membrane-bound thermosensor allows bacteria to adjust lipid desaturation for optimal membrane fluidity.
Jonikas, M. C. et al. Comprehensive characterization of genes required for protein folding in the endoplasmic reticulum. Science 323, 1693–1697 (2009).
Surma, M. A. et al. A lipid E-MAP identifies Ubx2 as a critical regulator of lipid saturation and lipid bilayer stress. Mol. Cell 51, 519–530 (2013).
Schuck, S., Prinz, W. A., Thorn, K. S., Voss, C. & Walter, P. Membrane expansion alleviates endoplasmic reticulum stress independently of the unfolded protein response. J. Cell Biol. 187, 525–536 (2009).
Promlek, T. et al. Membrane aberrancy and unfolded proteins activate the endoplasmic reticulum stress sensor Ire1 in different ways. Mol. Biol. Cell 22, 3520–3532 (2011).
Volmer, R., van der Ploeg, K. & Ron, D. Membrane lipid saturation activates endoplasmic reticulum unfolded protein response transducers through their transmembrane domains. Proc. Natl Acad. Sci. USA 110, 4628–4633 (2013).
Nilsson, I., Ohvo-Rekilä, H., Slotte, J. P., Johnson, A. E. & von Heijne, G. Inhibition of protein translocation across the endoplasmic reticulum membrane by sterols. J. Biol. Chem. 276, 41748–41754 (2001).
Krumpe, K. et al. Ergosterol content specifies targeting of tail-anchored proteins to mitochondrial outer membranes. Mol. Biol. Cell 23, 3927–3935 (2012).
Walker, A. K. et al. A conserved SREBP-1/phosphatidylcholine feedback circuit regulates lipogenesis in metazoans. Cell 147, 840–852 (2011).
Lee, J., Taneva, S. G., Holland, B. W., Tieleman, D. P. & Cornell, R. B. Structural basis for autoinhibition of CTP:phosphocholine cytidylyltransferase (CCT), the regulatory enzyme in phosphatidylcholine synthesis, by its membrane-binding amphipathic helix. J. Biol. Chem. 289, 1742–1755 (2014).
Attard, G. S., Templer, R. H., Smith, W. S., Hunt, A. N. & Jackowski, S. Modulation of CTP:phosphocholine cytidylyltransferase by membrane curvature elastic stress. Proc. Natl Acad. Sci. USA 97, 9032–9036 (2000).
Krahmer, N. et al. Phosphatidylcholine synthesis for lipid droplet expansion is mediated by localized activation of CTP:phosphocholine cytidylyltransferase. Cell Metab. 14, 504–515 (2011).
Mesmin, B. et al. A four-step cycle driven by PI(4)P hydrolysis directs sterol/PI(4)P exchange by the ER-Golgi tether OSBP. Cell 155, 830–843 (2013).
Tatsuta, T., Scharwey, M. & Langer, T. Mitochondrial lipid trafficking. Trends Cell Biol. 24, 44–52 (2014).
Urbani, L. & Simoni, R. D. Cholesterol and vesicular stomatitis virus G protein take separate routes from the endoplasmic reticulum to the plasma membrane. J. Biol. Chem. 265, 1919–1923 (1990).
Baumann, N. A. et al. Transport of newly synthesized sterol to the sterol-enriched plasma membrane occurs via nonvesicular equilibration. Biochemistry 44, 5816–5826 (2005). Together with ref. 55 this paper shows that anterograde sterol transport from the ER to the plasma membrane occurs by a non-vesicular, equilibratory mechanism in mammalian cells and yeast.
Kaplan, M. R. & Simoni, R. D. Intracellular transport of phosphatidylcholine to the plasma membrane. J. Cell Biol. 101, 441–445 (1985).
Sleight, R. G. & Pagano, R. E. Rapid appearance of newly synthesized phosphatidylethanolamine at the plasma membrane. J. Biol. Chem. 258, 9050–9058 (1983).
Heino, S. et al. Dissecting the role of the golgi complex and lipid rafts in biosynthetic transport of cholesterol to the cell surface. Proc. Natl Acad. Sci. USA 97, 8375–8380 (2000).
Hanada, K. et al. Molecular machinery for non-vesicular trafficking of ceramide. Nature 426, 803–809 (2003). This milestone paper defines the molecular mechanism for non-vesicular transport of ceramide from the ER to the trans -Golgi.
Kobayashi, T. & Pagano, R. E. Lipid transport during mitosis. Alternative pathways for delivery of newly synthesized lipids to the cell surface. J. Biol. Chem. 264, 5966–5973 (1989).
Maeda, K. et al. Interactome map uncovers phosphatidylserine transport by oxysterol-binding proteins. Nature 501, 257–261 (2013). This study shows that Osh6 and Osh7 transport PtdSer but not sterol between vesicle populations in the test tube and between the ER and plasma membrane in cells at ER–plasma-membrane contact sites.
Schnabl, M., Daum, G. & Pichler, H. Multiple lipid transport pathways to the plasma membrane in yeast. Biochim. Biophys. Acta 1687, 130–140 (2005).
Halter, D. et al. Pre- and post-Golgi translocation of glucosylceramide in glycosphingolipid synthesis. J. Cell Biol. 179, 101–115 (2007).
D'Angelo, G. et al. Vesicular and non-vesicular transport feed distinct glycosylation pathways in the Golgi. Nature 501, 116–120 (2013).
Silvius, J. R. & Leventis, R. Spontaneous interbilayer transfer of phospholipids: dependence on acyl chain composition. Biochemistry 32, 13318–13326 (1993).
Estronca, L. M. B. B., Moreno, M. J. & Vaz, W. L. C. Kinetics and thermodynamics of the association of dehydroergosterol with lipid bilayer membranes. Biophys. J. 93, 4244–4253 (2007).
Wirtz, K. W. Phospholipid transfer proteins. Annu. Rev. Biochem. 60, 73–99 (1991).
Loewen, C. J. R., Roy, A. & Levine, T. P. A conserved ER targeting motif in three families of lipid binding proteins and in Opi1p binds VAP. EMBO J. 22, 2025–2035 (2003).
Kawano, M., Kumagai, K., Nishijima, M. & Hanada, K. Efficient trafficking of ceramide from the endoplasmic reticulum to the Golgi apparatus requires a VAMP-associated protein-interacting FFAT motif of CERT. J. Biol. Chem. 281, 30279–30288 (2006).
Kumagai, K., Kawano-Kawada, M. & Hanada, K. Phosphoregulation of the ceramide transport protein CERT at serine 315 in the interaction with VAMP-associated protein (VAP) for inter-organelle trafficking of ceramide in mammalian cells. J. Biol. Chem. 289, 10748–10760 (2014).
Maxfield, F. R. & Menon, A. K. Intracellular sterol transport and distribution. Curr. Opin. Cell Biol. 18, 379–385 (2006).
Ngo, M. & Ridgway, N. D. Oxysterol binding protein-related protein 9 (ORP9) is a cholesterol transfer protein that regulates Golgi structure and function. Mol. Biol. Cell 20, 1388–1399 (2009).
Ridgway, N. D. Oxysterol-binding proteins. Subcell. Biochem. 51, 159–182 (2010).
de Saint-Jean, M. et al. Osh4p exchanges sterols for phosphatidylinositol 4-phosphate between lipid bilayers. J. Cell Biol. 195, 965–978 (2011). Together with ref. 53, this study shows that a mammalian OSBP and yeast Osh4 bind PtdIns(4)P and sterols in a mutually exclusive manner to facilitate heterotypic lipid exchange between the ER and trans -Golgi.
Banerji, S. et al. Oxysterol binding protein-dependent activation of sphingomyelin synthesis in the golgi apparatus requires phosphatidylinositol 4-kinase IIα. Mol. Biol. Cell 21, 4141–4150 (2010).
Funato, K. & Riezman, H. Vesicular and nonvesicular transport of ceramide from ER to the Golgi apparatus in yeast. J. Cell Biol. 155, 949–959 (2001).
Breslow, D. K. Sphingolipid homeostasis in the endoplasmic reticulum and beyond. Cold Spring Harb. Perspect. Biol. 5, a013326 (2013).
Breslow, D. K. et al. Orm family proteins mediate sphingolipid homeostasis. Nature 463, 1048–1053 (2010).
Han, S., Lone, M. A., Schneiter, R. & Chang, A. Orm1 and Orm2 are conserved endoplasmic reticulum membrane proteins regulating lipid homeostasis and protein quality control. Proc. Natl Acad. Sci. USA 107, 5851–5856 (2010). Together with ref. 79, this study identified Orm proteins as homeostatic regulators of sphingolipid biosynthesis through formation of a complex with the rate-limiting enzyme SPT.
Roelants, F. M., Breslow, D. K., Muir, A., Weissman, J. S. & Thorner, J. Protein kinase Ypk1 phosphorylates regulatory proteins Orm1 and Orm2 to control sphingolipid homeostasis in Saccharomyces cerevisiae. Proc. Natl Acad. Sci. USA 108, 19222–19227 (2011).
Nakano, K., Yamamoto, T., Kishimoto, T., Noji, T. & Tanaka, K. Protein kinases Fpk1p and Fpk2p are novel regulators of phospholipid asymmetry. Mol. Biol. Cell 19, 1783–1797 (2008).
Roelants, F. M., Baltz, A. G., Trott, A. E., Fereres, S. & Thorner, J. A protein kinase network regulates the function of aminophospholipid flippases. Proc. Natl Acad. Sci. USA 107, 34–39 (2010).
Sanyal, A. J. Mechanisms of disease: pathogenesis of nonalcoholic fatty liver disease. Nature Clin. Pract. Gastroenterol. Hepatol. 2, 46–53 (2005).
Kusminski, C. M., Shetty, S., Orci, L., Unger, R. H. & Scherer, P. E. Diabetes and apoptosis: lipotoxicity. Apoptosis 14, 1484–1495 (2009).
Zámbó, V. et al. Lipotoxicity in the liver. World J. Hepatol. 5, 550–557 (2013).
Li, Y. et al. Enrichment of endoplasmic reticulum with cholesterol inhibits sarcoplasmic-endoplasmic reticulum calcium ATPase-2b activity in parallel with increased order of membrane lipids: implications for depletion of endoplasmic reticulum calcium stores and apoptosis in cholesterol-loaded macrophages. J. Biol. Chem. 279, 37030–37039 (2004).
Fu, S. et al. Aberrant lipid metabolism disrupts calcium homeostasis causing liver endoplasmic reticulum stress in obesity. Nature 473, 528–531 (2011).
Fang, D.-L. et al. Endoplasmic reticulum stress leads to lipid accumulation through upregulation of SREBP-1c in normal hepatic and hepatoma cells. Mol. Cell. Biochem. 381, 127–137 (2013).
Paulusma, C. C. et al. Atp8b1 deficiency in mice reduces resistance of the canalicular membrane to hydrophobic bile salts and impairs bile salt transport. Hepatology 44, 195–204 (2006).
Kurbegov, A. C. et al. Biliary diversion for progressive familial intrahepatic cholestasis: improved liver morphology and bile acid profile. Gastroenterology 125, 1227–1234 (2003).
Paulusma, C. C., de Waart, D. R., Kunne, C., Mok, K. S. & Elferink, R. P. J. O. Activity of the bile salt export pump (ABCB11) is critically dependent on canalicular membrane cholesterol content. J. Biol. Chem. 284, 9947–9954 (2009).
Bankaitis, V. A. et al. Thoughts on Sec14-like nanoreactors and phosphoinositide signaling. Adv. Biol. Regul. 52, 115–121 (2012).
Alpy, F. & Tomasetto, C. START ships lipids across interorganelle space. Biochimie 96, 85–95 (2014).
Georgiev, A. G. et al. Osh proteins regulate membrane sterol organization but are not required for sterol movement between the ER and PM. Traffic 12, 1341–1355 (2011). This paper shows that the OSBP protein family in yeast is not required for sterol transport between the ER and plasma membrane.
Saito, S. et al. Protein phosphatase 2Cε is an endoplasmic reticulum integral membrane protein that dephosphorylates the ceramide transport protein CERT to enhance its association with organelle membranes. J. Biol. Chem. 283, 6584–6593 (2008).
Acknowledgements
Work in the authors laboratories is supported by the European Union (MC-ITN Sphingonet Project 289278 to J.C.H.), the Deutsche Forschungsgemeinschaft (SFB944-P14 to J.C.H.) and the National Institutes of Health (grant GM071041 to A.K.M.).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Additional information
Reprints and permissions information is available at www.nature.com/reprints.
Rights and permissions
About this article
Cite this article
Holthuis, J., Menon, A. Lipid landscapes and pipelines in membrane homeostasis. Nature 510, 48–57 (2014). https://doi.org/10.1038/nature13474
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature13474
This article is cited by
-
How ceramides affect the development of colon cancer: from normal colon to carcinoma
Pflügers Archiv - European Journal of Physiology (2024)
-
Exocytosis of the silicified cell wall of diatoms involves extensive membrane disintegration
Nature Communications (2023)
-
De novo lipogenesis fuels adipocyte autophagosome and lysosome membrane dynamics
Nature Communications (2023)
-
The expanding organelle lipidomes: current knowledge and challenges
Cellular and Molecular Life Sciences (2023)
-
HRG-9 homologues regulate haem trafficking from haem-enriched compartments
Nature (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.