Abstract
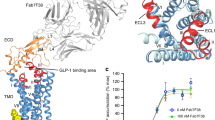
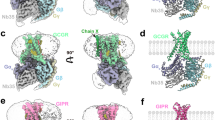
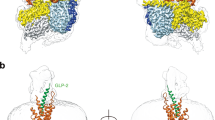
Binding of the glucagon peptide to the glucagon receptor (GCGR) triggers the release of glucose from the liver during fasting; thus GCGR plays an important role in glucose homeostasis. Here we report the crystal structure of the seven transmembrane helical domain of human GCGR at 3.4 Å resolution, complemented by extensive site-specific mutagenesis, and a hybrid model of glucagon bound to GCGR to understand the molecular recognition of the receptor for its native ligand. Beyond the shared seven transmembrane fold, the GCGR transmembrane domain deviates from class A G-protein-coupled receptors with a large ligand-binding pocket and the first transmembrane helix having a ‘stalk’ region that extends three alpha-helical turns above the plane of the membrane. The stalk positions the extracellular domain (∼12 kilodaltons) relative to the membrane to form the glucagon-binding site that captures the peptide and facilitates the insertion of glucagon’s amino terminus into the seven transmembrane domain.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Lagerström, M. C. & Schioth, H. B. Structural diversity of G protein-coupled receptors and significance for drug discovery. Nature Rev. Drug Discov. 7, 339–357 (2008)
Cho, Y. M., Merchant, C. E. & Kieffer, T. J. Targeting the glucagon receptor family for diabetes and obesity therapy. Pharmacol. Ther. 135, 247–278 (2012)
Katritch, V., Cherezov, V. & Stevens, R. C. Structure-function of the G protein-coupled receptor superfamily. Annu. Rev. Pharmacol. Toxicol. 53, 531–556 (2013)
Hoare, S. R. Mechanisms of peptide and nonpeptide ligand binding to class B G-protein-coupled receptors. Drug Discov. Today 10, 417–427 (2005)
Pal, K., Melcher, K. & Xu, H. E. Structure and mechanism for recognition of peptide hormones by Class B G-protein-coupled receptors. Acta Pharmacol. Sin. 33, 300–311 (2012)
Koth, C. M. et al. Molecular basis for negative regulation of the glucagon receptor. Proc. Natl Acad. Sci. USA 109, 14393–14398 (2012)
Parthier, C., Reedtz-Runge, S., Rudolph, R. & Stubbs, M. T. Passing the baton in class B GPCRs: peptide hormone activation via helix induction? Trends Biochem. Sci. 34, 303–310 (2009)
Underwood, C. R. et al. Crystal structure of glucagon-like peptide-1 in complex with the extracellular domain of the glucagon-like peptide-1 receptor. J. Biol. Chem. 285, 723–730 (2010)
Yaqub, T. et al. Identification of determinants of glucose-dependent insulinotropic polypeptide receptor that interact with N-terminal biologically active region of the natural ligand. Mol. Pharmacol. 77, 547–558 (2010)
Miller, L. J., Dong, M., Harikumar, K. G. & Gao, F. Structural basis of natural ligand binding and activation of the Class II G-protein-coupled secretin receptor. Biochem. Soc. Trans. 35, 709–712 (2007)
Dong, M. et al. Mapping spatial approximations between the amino terminus of secretin and each of the extracellular loops of its receptor using cysteine trapping. FASEB J. 26, 5092–5105 (2012)
Miller, L. J. et al. Refinement of glucagon-like peptide 1 docking to its intact receptor using mid-region photolabile probes and molecular modeling. J. Biol. Chem. 286, 15895–15907 (2011)
Dong, M. et al. Molecular basis of secretin docking to its intact receptor using multiple photolabile probes distributed throughout the pharmacophore. J. Biol. Chem. 286, 23888–23899 (2011)
Gensure, R. C., Shimizu, N., Tsang, J. & Gardella, T. J. Identification of a contact site for residue 19 of parathyroid hormone (PTH) and PTH-related protein analogs in transmembrane domain two of the type 1 PTH receptor. Mol. Endocrinol. 17, 2647–2658 (2003)
de Graaf, C., Rein, C., Piwnica, D., Giordanetto, F. & Rognan, D. Structure-based discovery of allosteric modulators of two related class B G-protein-coupled receptors. ChemMedChem 6, 2159–2169 (2011)
Chun, E. et al. Fusion partner toolchest for the stabilization and crystallization of G protein-coupled receptors. Structure 20, 967–976 (2012)
Ballesteros, J. A. & Weinstein, H. in Methods in Neurosciences Vol. 25 (ed. Sealfon, S. C. ) 366–428 (Academic Press, 1995)
Wootten, D., Simms, J., Miller, L. J., Christopoulos, A. & Sexton, P. M. Polar transmembrane interactions drive formation of ligand-specific and signal pathway-biased family B G protein-coupled receptor conformations. Proc. Natl Acad. Sci. USA 110, 5211–5216 (2013)
Unson, C. G. et al. Roles of specific extracellular domains of the glucagon receptor in ligand binding and signaling. Biochemistry 41, 11795–11803 (2002)
Xiao, Q., Jeng, W. & Wheeler, M. B. Characterization of glucagon-like peptide-1 receptor-binding determinants. J. Mol. Endocrinol. 25, 321–335 (2000)
Roberts, D. J., Vertongen, P. & Waelbroeck, M. Analysis of the glucagon receptor first extracellular loop by the substituted cysteine accessibility method. Peptides 32, 1593–1599 (2011)
Wu, H. et al. Structure of the human κ-opioid receptor in complex with JDTic. Nature 485, 327–332 (2012)
Manglik, A. et al. Crystal structure of the µ-opioid receptor bound to a morphinan antagonist. Nature 485, 321–326 (2012)
Fredriksson, R., Lagerstrom, M. C., Lundin, L. G. & Schioth, H. B. The G-protein-coupled receptors in the human genome form five main families. Phylogenetic analysis, paralogon groups, and fingerprints. Mol. Pharmacol. 63, 1256–1272 (2003)
Venkatakrishnan, A. J. et al. Molecular signatures of G-protein-coupled receptors. Nature 494, 185–194 (2013)
Inooka, H. et al. Conformation of a peptide ligand bound to its G-protein coupled receptor. Nature Struct. Biol. 8, 161–165 (2001)
Prévost, M. et al. Mutational and cysteine scanning analysis of the glucagon receptor N-terminal domain. J. Biol. Chem. 285, 30951–30958 (2010)
Ahn, J. M., Medeiros, M., Trivedi, D. & Hruby, V. J. Development of potent truncated glucagon antagonists. J. Med. Chem. 44, 1372–1379 (2001)
Coopman, K. et al. Residues within the transmembrane domain of the glucagon-like peptide-1 receptor involved in ligand binding and receptor activation: modelling the ligand-bound receptor. Mol. Endocrinol. 25, 1804–1818 (2011)
Di Paolo, E. et al. Contribution of the second transmembrane helix of the secretin receptor to the positioning of secretin. FEBS Lett. 424, 207–210 (1998)
Perret, J. et al. Mutational analysis of the glucagon receptor: similarities with the vasoactive intestinal peptide (VIP)/pituitary adenylate cyclase-activating peptide (PACAP)/secretin receptors for recognition of the ligand's third residue. Biochem. J. 362, 389–394 (2002)
Runge, S. et al. Three distinct epitopes on the extracellular face of the glucagon receptor determine specificity for the glucagon amino terminus. J. Biol. Chem. 278, 28005–28010 (2003)
Neumann, J. M. et al. Class-B GPCR activation: is ligand helix-capping the key? Trends Biochem. Sci. 33, 314–319 (2008)
Cascieri, M. A. et al. Characterization of a novel, non-peptidyl antagonist of the human glucagon receptor. J. Biol. Chem. 274, 8694–8697 (1999)
Di Paolo, E. et al. Mutations of aromatic residues in the first transmembrane helix impair signalling by the secretin receptor. Receptors Channels 6, 309–315 (1999)
Koole, C. et al. Second extracellular loop of human glucagon-like peptide-1 receptor (GLP-1R) has a critical role in GLP-1 peptide binding and receptor activation. J. Biol. Chem. 287, 3642–3658 (2012)
Tseng, C. C. & Lin, L. A point mutation in the glucose-dependent insulinotropic peptide receptor confers constitutive activity. Biochem. Biophys. Res. Commun. 232, 96–100 (1997)
Ganguli, S. C. et al. Protean effects of a natural peptide agonist of the G protein-coupled secretin receptor demonstrated by receptor mutagenesis. J. Pharmacol. Exp. Ther. 286, 593–598 (1998)
Solano, R. M. et al. Two basic residues of the h-VPAC1 receptor second transmembrane helix are essential for ligand binding and signal transduction. J. Biol. Chem. 276, 1084–1088 (2001)
Ceraudo, E. et al. Spatial proximity between the VPAC1 receptor and the amino terminus of agonist and antagonist peptides reveals distinct sites of interaction. FASEB J. 26, 2060–2071 (2012)
Tan, Y. V., Couvineau, A. & Laburthe, M. Diffuse pharmacophoric domains of vasoactive intestinal peptide (VIP) and further insights into the interaction of VIP with the N-terminal ectodomain of human VPAC1 receptor by photoaffinity labeling with [Bpa6]-VIP. J. Biol. Chem. 279, 38889–38894 (2004)
Caffrey, M. & Cherezov, V. Crystallizing membrane proteins using lipidic mesophases. Nature Protocols 4, 706–731 (2009)
Di Paolo, E. et al. Role of charged amino acids conserved in the vasoactive intestinal polypeptide/secretin family of receptors on the secretin receptor functionality. Peptides 20, 1187–1193 (1999)
Lomize, M. A., Pogozheva, I. D., Joo, H., Mosberg, H. I. & Lomize, A. L. OPM database and PPM web server: resources for positioning of proteins in membranes. Nucleic Acids Res. 40, D370–D376 (2012)
Hanson, M. A. et al. Profiling of membrane protein variants in a baculovirus system by coupling cell-surface detection with small-scale parallel expression. Protein Expr. Purif. 56, 85–92 (2007)
Misquitta, Y. & Caffrey, M. Detergents destabilize the cubic phase of monoolein: implications for membrane protein crystallization. Biophys. J. 85, 3084–3096 (2003)
Cherezov, V., Peddi, A., Muthusubramaniam, L., Zheng, Y. F. & Caffrey, M. A robotic system for crystallizing membrane and soluble proteins in lipidic mesophases. Acta Crystallogr. D 60, 1795–1807 (2004)
Xu, F., Liu, W., Hanson, M. A., Stevens, R. C. & Cherezov, V. Development of an automated high throughput LCP-FRAP assay to guide membrane protein crystallization in lipid mesophases. Cryst. Growth Des. 11, 1193–1201 (2011)
Cherezov, V. et al. Rastering strategy for screening and centring of microcrystal samples of human membrane proteins with a sub-10 µm size X-ray synchrotron beam. J. R. Soc. Interface 6 (Suppl 5). S587–S597 (2009)
Otwinowski, Z. & Minor, W. Processing of X-ray diffraction data collected in oscillation mode. Methods Enzymol. 276, 307–326 (1997)
McCoy, A. J. et al. Phaser crystallographic software. J. Appl. Crystalogr. 40, 658–674 (2007)
Terwilliger, T. C. et al. Decision-making in structure solution using Bayesian estimates of map quality: the PHENIX AutoSol wizard. Acta Crystallogr. D 65, 582–601 (2009)
Terwilliger, T. C. et al. phenix. mr_rosetta: molecular replacement and model rebuilding with Phenix and Rosetta. J. Struct. Funct. Genomics 13, 81–90 (2012)
Schwarzenbacher, R., Godzik, A. & Jaroszewski, L. The JCSG MR pipeline: optimized alignments, multiple models and parallel searches. Acta Crystallogr. D 64, 133–140 (2008)
Murshudov, G. N., Vagin, A. A. & Dodson, E. J. Refinement of macromolecular structures by the maximum-likelihood method. Acta Crystallogr. D 53, 240–255 (1997)
BUSTER. v. 2.8.0 (Global Phasing, Cambridge, UK, 2009)
Terwilliger, T. C. et al. Iterative model building, structure refinement and density modification with the PHENIX AutoBuild wizard. Acta Crystallogr. D 64, 61–69 (2008)
Emsley, P., Lohkamp, B., Scott, W. G. & Cowtan, K. Features and development of Coot. Acta Crystallogr. D 66, 486–501 (2010)
Bhat, T. Calculation of an OMIT map. J. Appl. Crystallogr. 21, 279–281 (1988)
ICM. Manual v. 3.0 (MolSoft, La Jolla, California, 2012)
Arnautova, Y. A., Abagyan, R. A. & Totrov, M. Development of a new physics-based internal coordinate mechanics force field and its application to protein loop modeling. Proteins 79, 477–498 (2011)
Acknowledgements
This work was supported by NIH Roadmap grant P50 GM073197 for technology development (V.C. and R.C.S.), and PSI:Biology grant U54 GM094618 for biological studies and structure production (target GPCR-49) (V.K., V.C. and R.C.S.); PSI:Biology grant U54 GM094586 for structure QC; The Ministry of Health grants 2012ZX09304-011 and 2013ZX09507002 (M.-W.W.), Shanghai Science and Technology Development Fund 11DZ2292200 (M.-W.W.); Novo Nordisk-Chinese Academy of Sciences Research Fund NNCAS-2011-7 (M.-W.W.); Thousand Talents Program in China (R.C.S. and M.-W.W.); NIH Postdoctoral Training Grant (NRSA) F32 DK088392 (F.Y.S.); The Netherlands Organization for Scientific Research (NWO) through a VENI grant (Grant 700.59.408 to C.d.G.); COST Action CM1207, GLISTEN (C.d.G). We also thank V. Hruby and M. Cai for advice with the glucagon binding assay and general discussions; J. Velasquez for help with molecular biology; T. Trinh and M. Chu for help with baculovirus expression; K. Kadyshevskaya for assistance with figure preparation; X. Q. Cai, J. Wang, Y. Feng, A. T. Dai, Y. Zhou, J. J. Deng, Y. B. Dai and J. W. Zhao for technical assistance in mutation studies; A. Walker for assistance with manuscript preparation; and J. Smith and R. Fischetti for assistance in development and use of the minibeam and beamtime at GM/CA-CAT beamline 23-ID at the Advanced Photon Source, which is supported by National Cancer Institute grant Y1-CO-1020 and National Institute of General Medical Sciences grant Y1-GM-1104.
Author information
Authors and Affiliations
Contributions
F.Y.S. designed, expressed, characterized and screened constructs and ligands for crystallization. F.Y.S. purified and crystallized the receptor in LCP, optimized crystallization conditions, grew crystals, collected diffraction data and prepared the manuscript. G.W.H. and Q.X. solved and refined the structure, and prepared the manuscript. V.C. collected and processed diffraction data, and prepared the manuscript. M.H., D.Y., Z.Z. and C.Z. expressed the receptor, and performed the mutagenesis and ligand-binding assay. V.K. and C.d.G. designed and analysed the receptor mutagenesis studies, constructed the receptor–ligand model and prepared the manuscript. D.W. and J.S.J. collected and processed SAD data and determined an initial electron density map from experimental phases. W.L. and V.C. trained and assisted in LCP crystallization. J.L. provided ligands for GCGR and prepared the manuscript. R.C.S., F.Y.S., M.-W.W., V.K., V.C. and C.d.G. were responsible for the overall project strategy and management and wrote the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
This file contains Supplementary Tables 1-6, Supplementary Figures 1-10 and Supplementary References. (PDF 1596 kb)
Rights and permissions
About this article
Cite this article
Siu, F., He, M., de Graaf, C. et al. Structure of the human glucagon class B G-protein-coupled receptor. Nature 499, 444–449 (2013). https://doi.org/10.1038/nature12393
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature12393
This article is cited by
-
Effects of site-directed mutagenesis of GLP-1 and glucagon receptors on signal transduction activated by dual and triple agonists
Acta Pharmacologica Sinica (2023)
-
Dynamics of GLP-1R peptide agonist engagement are correlated with kinetics of G protein activation
Nature Communications (2022)
-
GPCR Intracellular Loop Regulation of Beta-Arrestin-Mediated Endosomal Signaling Dynamics
Journal of Molecular Neuroscience (2022)
-
Structural basis for activation of the growth hormone-releasing hormone receptor
Nature Communications (2020)
-
Advances in therapeutic peptides targeting G protein-coupled receptors
Nature Reviews Drug Discovery (2020)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.