Abstract
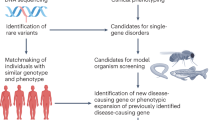
Since the publication of the human reference genome, the identities of specific genes associated with human diseases are being discovered at a rapid rate. A central problem is that the biological activity of these genes is often unclear. Detailed investigations in model vertebrate organisms, typically mice, have been essential for understanding the activities of many orthologues of these disease-associated genes. Although gene-targeting approaches1,2,3 and phenotype analysis have led to a detailed understanding of nearly 6,000 protein-coding genes3,4, this number falls considerably short of the more than 22,000 mouse protein-coding genes5. Similarly, in zebrafish genetics, one-by-one gene studies using positional cloning6, insertional mutagenesis7,8,9, antisense morpholino oligonucleotides10, targeted re-sequencing11,12,13, and zinc finger and TAL endonucleases14,15,16,17 have made substantial contributions to our understanding of the biological activity of vertebrate genes, but again the number of genes studied falls well short of the more than 26,000 zebrafish protein-coding genes18. Importantly, for both mice and zebrafish, none of these strategies are particularly suited to the rapid generation of knockouts in thousands of genes and the assessment of their biological activity. Here we describe an active project that aims to identify and phenotype the disruptive mutations in every zebrafish protein-coding gene, using a well-annotated zebrafish reference genome sequence18,19, high-throughput sequencing and efficient chemical mutagenesis. So far we have identified potentially disruptive mutations in more than 38% of all known zebrafish protein-coding genes. We have developed a multi-allelic phenotyping scheme to efficiently assess the effects of each allele during embryogenesis and have analysed the phenotypic consequences of over 1,000 alleles. All mutant alleles and data are available to the community and our phenotyping scheme is adaptable to phenotypic analysis beyond embryogenesis.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Accession codes
Accessions
European Nucleotide Archive
Data deposits
Detailed information on alleles and their availability can be found online (http://www.sanger.ac.uk/Projects/D_rerio/zmp/). All sequencing data are deposited in the European Molecular Biology Laboratory (EMBL) European Nucleotide Archive under accession ERP000426.
References
Gossler, A., Joyner, A. L., Rossant, J. & Skarnes, W. C. Mouse embryonic stem cells and reporter constructs to detect developmentally regulated genes. Science 244, 463–465 (1989)
Skarnes, W. C., Auerbach, B. A. & Joyner, A. L. A gene trap approach in mouse embryonic stem cells: the lacZ reported is activated by splicing, reflects endogenous gene expression, and is mutagenic in mice. Genes Dev. 6, 903–918 (1992)
Ringwald, M. et al. The IKMC web portal: a central point of entry to data and resources from the International Knockout Mouse Consortium. Nucleic Acids Res. 39, D849–D855 (2011)
Church, D. M. et al. Lineage-specific biology revealed by a finished genome assembly of the mouse. PLoS Biol. 7, e1000112 (2009)
Chinwalla, A. T. et al. Initial sequencing and comparative analysis of the mouse genome. Nature 420, 520–562 (2002)
Zhang, J., Talbot, W. S. & Schier, A. F. Positional cloning identifies zebrafish one-eyed pinhead as a permissive EGF-related ligand required during gastrulation. Cell 92, 241–251 (1998)
Golling, G. et al. Insertional mutagenesis in zebrafish rapidly identifies genes essential for early vertebrate development. Nature Genet. 31, 135–140 (2002)
Amsterdam, A. et al. Identification of 315 genes essential for early zebrafish development. Proc. Natl Acad. Sci. USA 101, 12792–12797 (2004)
Sun, Z. et al. A genetic screen in zebrafish identifies cilia genes as a principal cause of cystic kidney. Development 131, 4085–4093 (2004)
Nasevicius, A. & Ekker, S. C. Effective targeted gene 'knockdown' in zebrafish. Nature Genet. 26, 216–220 (2000)
Kettleborough, R. N., Bruijn, E., Eeden, F., Cuppen, E. & Stemple, D. L. High-throughput target-selected gene inactivation in zebrafish. Methods Cell Biol. 104, 121–127 (2011)
Sood, R. et al. Methods for reverse genetic screening in zebrafish by resequencing and TILLING. Methods 39, 220–227 (2006)
Wienholds, E. et al. Efficient target-selected mutagenesis in zebrafish. Genome Res. 13, 2700–2707 (2003)
Meng, X., Noyes, M. B., Zhu, L. J., Lawson, N. D. & Wolfe, S. A. Targeted gene inactivation in zebrafish using engineered zinc-finger nucleases. Nature Biotechnol. 26, 695–701 (2008)
Doyon, Y. et al. Heritable targeted gene disruption in zebrafish using designed zinc-finger nucleases. Nature Biotechnol. 26, 702–708 (2008)
Huang, P. et al. Heritable gene targeting in zebrafish using customized TALENs. Nature Biotechnol. 29, 699–700 (2011)
Sander, J. D. et al. Targeted gene disruption in somatic zebrafish cells using engineered TALENs. Nature Biotechnol. 29, 697–698 (2011)
Howe, K. et al. The zebrafish reference genome sequence and its relationship to the human genome. Nature (in the press)
Collins, J. E., White, S., Searle, S. M. & Stemple, D. L. Incorporating RNA-seq data into the zebrafish ensembl gene build. Genome Res. 22, 2067–2078 (2012)
Stemple, D. L. TILLING–a high-throughput harvest for functional genomics. Nature Rev. Genet. 5, 145–150 (2004)
The 1000 Genome Project Consortium A map of human genome variation from population-scale sequencing. Nature 467, 1061–1073 (2011)
The 1000 Genome Project Consortium An integrated map of genetic variation from 1,092 human genomes. Nature 491, 56–65 (2012)
Ayadi, A. et al. Mouse large-scale phenotyping initiatives: overview of the European Mouse Disease Clinic (EUMODIC) and of the Wellcome Trust Sanger Institute Mouse Genetics Project. Mamm. Genome 23, 600–610 (2012)
Seiler, C. et al. Myosin VI is required for structural integrity of the apical surface of sensory hair cells in zebrafish. Dev. Biol. 272, 328–338 (2004)
Manfroid, I. et al. Zebrafish sox9b is crucial for hepatopancreatic duct development and pancreatic endocrine cell regeneration. Dev. Biol. 366, 268–278 (2012)
Steffen, L. S. et al. The zebrafish runzel muscular dystrophy is linked to the titin gene. Dev. Biol. 309, 180–192 (2007)
Haffter, P. et al. The identification of genes with unique and essential functions in the development of the zebrafish, Danio rerio. Development 123, 1–36 (1996)
Driever, W. et al. A genetic screen for mutations affecting embryogenesis in zebrafish. Development. 123, 37–46 (1996)
Nielsen, R., Paul, J. S., Albrechtsen, A. & Song, Y. S. Genotype and SNP calling from next-generation sequencing data. Nature Rev. Genet. 12, 443–451 (2011)
McLaren, W. et al. Deriving the consequences of genomic variants with the Ensembl API and SNP Effect Predictor. Bioinformatics 26, 2069–2070 (2010)
Acknowledgements
We thank P. Ellis, E. Markham, H. van Roekel, P. Toonen, J. van de Belt, J. Mudde and S. Widaa for technical assistance, and B. Novak, Y. Yi and E. LeProust from Agilent Technologies. We thank everyone at The Zebrafish International Resource Center and the European Zebrafish Resource Center for stocking and distributing alleles. We thank members of the Wellcome Trust Sanger Institute RSF and DNA pipelines. We also thank G. Powell, J. Collins and F. L. Marlow for critical reading of the manuscript. This work was funded through a core grant to the Sanger Institute by the Wellcome Trust (grant number 098051), the US National Institutes of Health (5R01HG004819), the EU Sixth Framework Programme (ZF-MODELS, contract number LSHG-CT-2003-503496) and the EU Seventh Framework Programme (ZF-HEALTH). F.v.E. is supported by the UK Medical Research Council (grant number G0777791) and E.C. is supported by the SmartMix program (SSM06010) from the Dutch government.
Author information
Authors and Affiliations
Contributions
R.N.W.K., E.M.B.-N. and S.A.H. initiated, organized and executed the work (equal contributions). R.N.W.K., E.M.B.-N., S.A.H. and D.S. designed the experiments. R.N.W.K., E.M.B.-N. and S.A.H. wrote the manuscript with assistance from I.S., R.J.W., C.M.D. and D.L.S. Mutagenesis was carried out by R.N.W.K., F.v.E. and E.d.B. The mutation analysis pipeline was developed by I.S. and I.J.N., and maintained by R.J.W. C.H. and F.F. implemented and improved genotyping procedures, F.F. and E.M.B.-N. developed the cryopreservation procedure, S.A.H., S.M., C.S., C.M.D. and N.W. carried out the phenotyping, J.Y. designed and tested the first Agilent SureSelect exome set, R.G. helped to maintain and distribute alleles. S.C. and A.H. provided assistance for cryopreservation and genotyping. E.C. and D.L.S. collaborated in the initiation, design and process development of the project. All authors read the manuscript and provided comments.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
This file contains Supplementary Tables 1-3 and guidance notes for the Zebrafish Mutation Project website. (PDF 346 kb)
Rights and permissions
About this article
Cite this article
Kettleborough, R., Busch-Nentwich, E., Harvey, S. et al. A systematic genome-wide analysis of zebrafish protein-coding gene function. Nature 496, 494–497 (2013). https://doi.org/10.1038/nature11992
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature11992
This article is cited by
-
Pla2g12b drives expansion of triglyceride-rich lipoproteins
Nature Communications (2024)
-
Digenic inheritance involving a muscle-specific protein kinase and the giant titin protein causes a skeletal muscle myopathy
Nature Genetics (2024)
-
Oxidative pentose phosphate pathway controls vascular mural cell coverage by regulating extracellular matrix composition
Nature Metabolism (2022)
-
Rear traction forces drive adherent tissue migration in vivo
Nature Cell Biology (2022)
-
Zebrafish: A Promising Real-Time Model System for Nanotechnology-Mediated Neurospecific Drug Delivery
Nanoscale Research Letters (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.