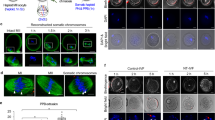
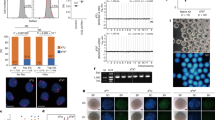
Abstract
Mitochondrial DNA mutations transmitted maternally within the oocyte cytoplasm often cause life-threatening disorders. Here we explore the use of nuclear genome transfer between unfertilized oocytes of two donors to prevent the transmission of mitochondrial mutations. Nuclear genome transfer did not reduce developmental efficiency to the blastocyst stage, and genome integrity was maintained provided that spontaneous oocyte activation was avoided through the transfer of incompletely assembled spindle–chromosome complexes. Mitochondrial DNA transferred with the nuclear genome was initially detected at levels below 1%, decreasing in blastocysts and stem-cell lines to undetectable levels, and remained undetectable after passaging for more than one year, clonal expansion, differentiation into neurons, cardiomyocytes or β-cells, and after cellular reprogramming. Stem cells and differentiated cells had mitochondrial respiratory chain enzyme activities and oxygen consumption rates indistinguishable from controls. These results demonstrate the potential of nuclear genome transfer to prevent the transmission of mitochondrial disorders in humans.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Jenuth, J. P., Peterson, A. C., Fu, K. & Shoubridge, E. A. Random genetic drift in the female germline explains the rapid segregation of mammalian mitochondrial DNA. Nature Genet. 14, 146–151 (1997)
Bolhuis, P. A. et al. Rapid shift in genotype of human mitochondrial DNA in a family with Leber’s hereditary optic neuropathy. Biochem. Biophys. Res. Commun. 170, 994–997 (1990)
Cree, L. M. et al. A reduction of mitochondrial DNA molecules during embryogenesis explains the rapid segregation of genotypes. Nature Genet. 40, 249–254 (2008)
Wai, T., Teoli, D. & Shoubridge, E. A. The mitochondrial DNA genetic bottleneck results from replication of a subpopulation of genomes. Nature Genet. 40, 1484–1488 (2008)
Steffann, J. et al. Analysis of mtDNA variant segregation during early human embryonic development: a tool for successful NARP preimplantation diagnosis. J. Med. Genet. 43, 244–247 (2006)
Nuffield Council on Bioethics. Novel Techniques for the Prevention of Mitochondrial DNA Disorders: an Ethical Reviewhttp://www.nuffieldbioethics.org/news/discussion-event-novel-techniques-prevention-mitochondrial-dna-disorders-ethical-review (2012)
Sato, A. et al. Gene therapy for progeny of mito-mice carrying pathogenic mtDNA by nuclear transplantation. Proc. Natl Acad. Sci. USA 102, 16765–16770 (2005)
Tachibana, M. et al. Mitochondrial gene replacement in primate offspring and embryonic stem cells. Nature 461, 367–372 (2009)
Craven, L. et al. Pronuclear transfer in human embryos to prevent transmission of mitochondrial DNA disease. Nature 465, 82–85 (2010)
Egli, D. et al. Reprogramming within hours following nuclear transfer into mouse but not human zygotes. Nature Comm. 2, 488 (2011)
Ganem, N. J., Godinho, S. A. & Pellman, D. A mechanism linking extra centrosomes to chromosomal instability. Nature 460, 278–282 (2009)
Sathananthan, A. H. et al. Centrioles in the beginning of human development. Proc. Natl Acad. Sci. USA 88, 4806–4810 (1991)
Kaufman, M. H., Robertson, E. J., Handyside, A. H. & Evans, M. J. Establishment of pluripotent cell lines from haploid mouse embryos. J. Embryol. Exp. Morphol. 73, 249–261 (1983)
Draper, J. S. et al. Recurrent gain of chromosomes 17q and 12 in cultured human embryonic stem cells. Nature Biotechnol. 22, 53–54 (2004)
Kim, K. et al. Histocompatible embryonic stem cells by parthenogenesis. Science 315, 482–486 (2007)
Noggle, S. et al. Human oocytes reprogram somatic cells to a pluripotent state. Nature 478, 70–75 (2011)
Hyun, C. S. et al. Optimal ICSI timing after the first polar body extrusion in in vitro matured human oocytes. Hum. Reprod. 22, 1991–1995 (2007)
Brinkley, B. R. & Cartwright, J., Jr Cold-labile and cold-stable microtubules in the mitotic spindle of mammalian cells. Ann. NY Acad. Sci. 253, 428–439 (1975)
van Oven, M. & Kayser, M. Updated comprehensive phylogenetic tree of global human mitochondrial DNA variation. Hum. Mutat. 30, E386–E394 (2009)
Mimaki, M. et al. Reversible infantile respiratory chain deficiency: a clinical and molecular study. Ann. Neurol. 68, 845–854 (2010)
Wang, J., Venegas, V., Li, F. & Wong, L.-J. Analysis of mitochondrial DNA point mutation heteroplasmy by ARMS quantitative PCR. Curr. Protocols Human Genet. Chapter 19, Unit–19.16 (2011)
Bai, R.-K. & Wong, L.-J. C. Detection and quantification of heteroplasmic mutant mitochondrial DNA by real-time amplification refractory mutation system quantitative PCR analysis: a single-step approach. Clinical Chem. 50, 996–1001 (2004)
Lee, H. S. et al. Rapid mitochondrial DNA segregation in primate preimplantation embryos precedes somatic and germline bottleneck. Cell Rep. 1, 506–515 (2012)
Meirelles, F. V. & Smith, L. C. Mitochondrial genotype segregation during preimplantation development in mouse heteroplasmic embryos. Genetics 148, 877–883 (1998)
Jenuth, J. P., Peterson, A. C. & Shoubridge, E. A. Tissue-specific selection for different mtDNA genotypes in heteroplasmic mice. Nature Genet. 16, 93–95 (1997)
Sharpley, M. S. et al. Heteroplasmy of mouse mtDNA is genetically unstable and results in altered behavior and cognition. Cell 151, 333–343 (2012)
Fujikura, J. et al. Induced pluripotent stem cells generated from diabetic patients with mitochondrial DNA A3243G mutation. Diabetologia 55, 1689–1698 (2012)
Birket, M. J. et al. A reduction in ATP demand and mitochondrial activity with neural differentiation of human embryonic stem cells. J. Cell Sci. 124, 348–358 (2011)
Cowan, C. A. et al. Derivation of embryonic stem-cell lines from human blastocysts. New Engl. J. Med. 350, 1353–1356 (2004)
Inoue, S., Fuseler, J., Salmon, E. D. & Ellis, G. W. Functional organization of mitotic microtubules. Physical chemistry of the in vivo equilibrium system. Biophys. J. 15, 725–744 (1975)
Bianchi, V., Coticchio, G., Fava, L., Flamigni, C. & Borini, A. Meiotic spindle imaging in human oocytes frozen with a slow freezing procedure involving high sucrose concentration. Human Reprod. 20, 1078–1083 (2005)
Larman, M. G., Minasi, M. G., Rienzi, L. & Gardner, D. K. Maintenance of the meiotic spindle during vitrification in human and mouse oocytes. Reprod. Biomed. Online 15, 692–700 (2007)
Forman, E. J. et al. Oocyte vitrification does not increase the risk of embryonic aneuploidy or diminish the implantation potential of blastocysts created after intracytoplasmic sperm injection: a novel, paired randomized controlled trial using DNA fingerprinting. Fertil. Steril. 98, 644–649 (2012)
Gook, D. A., Osborn, S. M., Bourne, H. & Johnston, W. I. Fertilization of human oocytes following cryopreservation; normal karyotypes and absence of stray chromosomes. Hum. Reprod. 9, 684–691 (1994)
ASRM. Mature oocyte cryopreservation: a guideline. Fertil. Steril.http://dx.doi.org/10.1016/j.fertnstert.2012.09.028 (18 October 2012)
Tachibana, M. et al. Towards germline gene therapy of inherited mitochondrial diseases. Naturehttp://dx.doi.org/10.1038/nature11647 (this issue)
Tachibana, M., Sparman, M. & Mitalipov, S. Chromosome transfer in mature oocytes. Nature Protoc. 5, 1138–1147 (2010)
Liu, D., Vader, G., Vromans, M. J., Lampson, M. A. & Lens, S. M. Sensing chromosome bi-orientation by spatial separation of aurora B kinase from kinetochore substrates. Science 323, 1350–1353 (2009)
A mother’s gift, minus mitochondria. Nature Med. 16, 645 (2010)
Society for Assisted Reproductive Technology, American Society for Reproductive Medicine. Assisted reproductive technology in the United States: 2000 results generated from the American Society for Reproductive Medicine/Society for Assisted Reproductive Technology Registry. Fertil. Steril. 81, 1207–1220 (2004)
The Ethics Committee of the American Society for Reproductive Medicine. Financial compensation of oocyte donors. Fertil. Steril. 88, 305–309 (2007)
Chen, A. E. et al. Optimal timing of inner cell mass isolation increases the efficiency of human embryonic stem cell derivation and allows generation of sibling cell lines. Cell Stem Cell 4, 103–106 (2009)
Lin, D. P.-C. et al. Comparison of mitochondrial DNA contents in human embryos with good or poor morphology at the 8-cell stage. Fertil. Steril. 81, 73–79 (2004)
D’Amour, K. A. et al. Production of pancreatic hormone–expressing endocrine cells from human embryonic stem cells. Nature Biotechnol. 24, 1392–1401 (2006)
Chambers, S. M. et al. Highly efficient neural conversion of human ES and iPS cells by dual inhibition of SMAD signaling. Nature Biotechnol. 27, 275–280 (2009)
Burridge, P. W. et al. A universal system for highly efficient cardiac differentiation of human induced pluripotent stem cells that eliminates interline variability. PloS ONE 6, e18293 (2011)
Lin, T. et al. A chemical platform for improved induction of human iPSCs. Nature Methods 6, 805–808 (2009)
DiMauro, S. et al. Cytochrome c oxidase deficiency in Leigh syndrome. Ann. Neurol. 22, 498–506 (1987)
Acknowledgements
We thank S. Chang and K. Eggan for discussions, Z. Hall for critical reading of the manuscript, and L. Yu and O. Nahum for SNP-array preparation. We thank anonymous oocyte donors for participating in research, and M. Spencer for a Lykos laser system. This work was supported by the New York Stem Cell Foundation, the New York State Stem Cell Science award C026184, and the Bernard and Anne Spitzer Fund.
Author information
Authors and Affiliations
Contributions
M.V.S. consented oocyte donors and retrieved oocytes. R.P. contributed IVF developmental data. R.S.G. and M.V.S. wrote institutional review board and consent documents. D.E., D.P. and S.N. designed and performed experiments with oocytes. D.P. and V.E. determined heteroplasmy. N.T. performed array analysis of single cells. D.E., D.P., V.E., L.S., K.A.W., H.H., M.Z. and D.J.K characterized stem-cell lines. D.E., D.P., V.E. and M.H. wrote the paper.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
This file contains Supplementary Figures 1-9, Supplementary Tables 1-8 and Supplementary Karyotypes. (PDF 41490 kb)
SwaPS1 cardiomyocytes
This video shows a contracting embryoid body that had undergone directed differentiation toward a cardiac (mesodermal) lineage. (MOV 949 kb)
Rights and permissions
About this article
Cite this article
Paull, D., Emmanuele, V., Weiss, K. et al. Nuclear genome transfer in human oocytes eliminates mitochondrial DNA variants. Nature 493, 632–637 (2013). https://doi.org/10.1038/nature11800
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature11800
This article is cited by
-
Mitochondria and the eye—manifestations of mitochondrial diseases and their management
Eye (2023)
-
Genome transfer technique for bovine embryo production using the metaphase plate and polar body
Journal of Assisted Reproduction and Genetics (2023)
-
Mitochondria: emerging therapeutic strategies for oocyte rescue
Reproductive Sciences (2022)
-
Reduction of mtDNA heteroplasmy in mitochondrial replacement therapy by inducing forced mitophagy
Nature Biomedical Engineering (2022)
-
Identifying regulators of parental imprinting by CRISPR/Cas9 screening in haploid human embryonic stem cells
Nature Communications (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.