Abstract
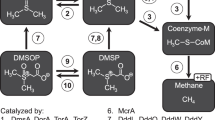
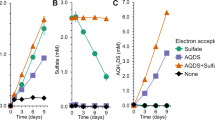
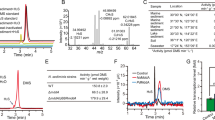
Emissions of methane, a potent greenhouse gas, from marine sediments are controlled by anaerobic oxidation of methane coupled primarily to sulphate reduction (AOM). Sulphate-coupled AOM is believed to be mediated by a consortium of methanotrophic archaea (ANME) and sulphate-reducing Deltaproteobacteria but the underlying mechanism has not yet been resolved. Here we show that zero-valent sulphur compounds (S0) are formed during AOM through a new pathway for dissimilatory sulphate reduction performed by the methanotrophic archaea. Hence, AOM might not be an obligate syntrophic process but may be carried out by the ANME alone. Furthermore, we show that the produced S0—in the form of disulphide—is disproportionated by the Deltaproteobacteria associated with the ANME. Our observations expand the diversity of known microbially mediated sulphur transformations and have significant implications for our understanding of the biogeochemical carbon and sulphur cycles.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Knittel, K. & Boetius, A. Anaerobic oxidation of methane: progress with an unknown process. Annu. Rev. Microbiol. 63, 311–334 (2009)
Hoehler, T. M., Alperin, M. J., Albert, D. B. & Martens, C. S. Field and laboratory studies of methane oxidation in an anoxic marine sediment: evidence for a methanogen-sulfate reducer consortium. Glob. Biogeochem. Cycles 8, 451–463 (1994)
Boetius, A. et al. A marine microbial consortium apparently mediating anaerobic oxidation of methane. Nature 407, 623–626 (2000)
Orphan, V. J., House, C. H., Hinrichs, K. U., McKeegan, K. D. & DeLong, E. F. Methane-consuming archaea revealed by directly coupled isotopic and phylogenetic analysis. Science 293, 484–487 (2001)
Alperin, M. & Hoehler, T. The ongoing mystery of sea-floor methane. Science 329, 288–289 (2010)
Schreiber, L., Holler, T., Knittel, K., Meyerdierks, A. & Amann, R. Identification of the dominant sulfate-reducing bacterial partner of anaerobic methanotrophs of the ANME-2 clade. Environ. Microbiol. 12, 2327–2340 (2010)
Nauhaus, K., Albrecht, M., Elvert, M., Boetius, A. & Widdel, F. In vitro cell growth of marine archaeal-bacterial consortia during anaerobic oxidation of methane with sulfate. Environ. Microbiol. 9, 187–196 (2007)
Wegener, G., Niemann, H., Elvert, M., Hinrichs, K. U. & Boetius, A. Assimilation of methane and inorganic carbon by microbial communities mediating the anaerobic oxidation of methane. Environ. Microbiol. 10, 2287–2298 (2008)
Meulepas, R. J. W., Jagersma, C. G., Khadem, A. F., Stams, A. J. M. & Lens, P. N. L. Effect of methanogenic substrates on anaerobic oxidation of methane and sulfate reduction by an anaerobic methanotrophic enrichment. Appl. Microbiol. Biotechnol. 87, 1499–1506 (2010)
Nauhaus, K., Boetius, A., Kruger, M. & Widdel, F. In vitro demonstration of anaerobic oxidation of methane coupled to sulphate reduction in sediment from a marine gas hydrate area. Environ. Microbiol. 4, 296–305 (2002)
Sørensen, K. B., Finster, K. & Ramsing, N. B. Thermodynamic and kinetic requirements in anaerobic methane oxidizing consortia exclude hydrogen, acetate, and methanol as possible electron shuttles. Microb. Ecol. 42, 1–10 (2001)
Orcutt, B. & Meile, C. Constraints on mechanisms and rates of anaerobic oxidation of methane by microbial consortia: process-based modeling of ANME-2 archaea and sulfate reducing bacteria interactions. Biogeosciences 5, 1587–1599 (2008)
Alperin, M. J. & Hoehler, T. M. Anaerobic methane oxidation by archaea/sulfate-reducing bacteria aggregates: 1. Thermodynamic and physical constraints. Am. J. Sci. 309, 869–957 (2009)
Moran, J. J. et al. Methyl sulfides as intermediates in the anaerobic oxidation of methane. Environ. Microbiol. 10, 162–173 (2008)
Meyerdierks, A. et al. Metagenome and mRNA expression analyses of anaerobic methanotrophic archaea of the ANME-1 group. Environ. Microbiol. 12, 422–439 (2010)
Shima, S. & Thauer, R. K. Methyl-coenzyme M reductase and the anaerobic oxidation of methane in methanotrophic Archaea. Curr. Opin. Microbiol. 8, 643–648 (2005)
Holmkvist, L. et al. Sulfate reduction below the sulfate-methane transition in Black Sea sediments. Deep Sea Res. Part I Oceanogr. Res. Pap. 58, 493–504 (2011)
Joye, S. B. et al. The anaerobic oxidation of methane and sulfate reduction in sediments from Gulf of Mexico cold seeps. Chem. Geol. 205, 219–238 (2004)
Hansen, L. B., Finster, K., Fossing, H. & Iversen, N. Anaerobic methane oxidation in sulfate depleted sediments: effects of sulfate and molybdate additions. Aquat. Microb. Ecol. 14, 195–204 (1998)
Kamyshny, A., Ekeltchik, I., Gun, J. & Lev, O. Method for the determination of inorganic polysulfide distribution in aquatic systems. Anal. Chem. 78, 2631–2639 (2006)
Pasteris, J. D., Freeman, J. J., Goffredi, S. K. & Buck, K. R. Raman spectroscopic and laser scanning confocal microscopic analysis of sulfur in living sulfur-precipitating marine bacteria. Chem. Geol. 180, 3–18 (2001)
Trofimov, B. A., Sinegovskaya, L. M. & Gusarova, N. K. Vibrations of the S-S bond in elemental sulfur and organic polysulfides: a structural guide. J. Sulfur Chem. 30, 518–554 (2009)
Holler, T. et al. Carbon and sulfur back flux during anaerobic microbial oxidation of methane and coupled sulfate reduction. Proc. Natl Acad. Sci. USA 108, E1484–E1490 (2011)
Basen, M. et al. Bacterial enzymes for dissimilatory sulfate reduction in a marine microbial mat (Black Sea) mediating anaerobic oxidation of methane. Environ. Microbiol. 13, 1370–1379 (2011)
Thauer, R. K. Biochemistry of methanogenesis: a tribute to Marjory Stephenson. Microbiology 144, 2377–2406 (1998)
Widdel, F., Musat, F., Knittel, K. & Galushko, A. in Sulphate-Reducing Bacteria: Environmental and Engineered Systems (eds Barton, L. et al.) 265–303 (Cambridge Univ. Press, 2007)
Johnson, E. F. & Mukhopadhyay, B. in Microbial Sulfur Metabolism. (eds Dahl, C. et al.) 202–216 (Springer, 2008)
Thauer, R. K. & Shima, S. in Incredible Anaerobes: from Physiology to Genomics to Fuels Vol. 1125 158–170 (Wiley-Blackwell, 2008)
Thauer, R. K. Anaerobic oxidation of methane with sulfate: on the reversibility of the reactions that are catalyzed by enzymes also involved in methanogenesis from CO2 . Curr. Opin. Microbiol. 14, 292–299 (2011)
Orphan, V. J., House, C. H., Hinrichs, K. U., McKeegan, K. D. & DeLong, E. F. Multiple archaeal groups mediate methane oxidation in anoxic cold seep sediments. Proc. Natl Acad. Sci. USA 99, 7663–7668 (2002)
Krämer, M. & Cypionka, H. Sulfate formation via ATP sulfurylase in thiosulfate-disproportionating and sulfite-disproportionating bacteria. Arch. Microbiol. 151, 232–237 (1989)
Trüper, H. G. & Fischer, U. Anaerobic oxidation of sulfur compounds as electron donors for bacterial photosynthesis. Phil. Trans. R. Soc. Lond. B 298, 529–542 (1982)
House, C. H., Beal, E. J. & Orphan, V. J. The apparent involvement of ANMEs in mineral dependent methane oxidation, as an analog for possible Martian methanotrophy. Life 1, 19–33 (2011)
Treude, T. et al. Consumption of methane and CO2 by methanotrophic microbial mats from gas seeps of the anoxic Black Sea. Appl. Environ. Microbiol. 73, 2271–2283 (2007)
Rickard, D. & Luther, G. W. Chemistry of iron sulfides. Chem. Rev. 107, 514–562 (2007)
Niemann, H. et al. Novel microbial communities of the Haakon Mosby mud volcano and their role as a methane sink. Nature 443, 854–858 (2006)
Lösekann, T. et al. Diversity and abundance of aerobic and anaerobic methane oxidizers at the Haakon Mosby mud volcano, Barents Sea. Appl. Environ. Microbiol. 73, 3348–3362 (2007)
Pernthaler, A. et al. Diverse syntrophic partnerships from deep-sea methane vents revealed by direct cell capture and metagenomics. Proc. Natl Acad. Sci. USA 105, 7052–7057 (2008)
Lovley, D. R. & Phillips, E. J. P. Novel processes for anaerobic sulfate production from elemental sulfur by sulfate-reducing bacteria. Appl. Environ. Microbiol. 60, 2394–2399 (1994)
Fuseler, K. & Cypionka, H. Elemental sulfur as an intermediate of sulfide oxidation with oxygen by Desulfobulbus propionicus . Arch. Microbiol. 164, 104–109 (1995)
Parkes, R. J. et al. Deep sub-seafloor prokaryotes stimulated at interfaces over geological time. Nature 436, 390–394 (2005)
Lipp, J. S., Morono, Y., Inagaki, F. & Hinrichs, K. U. Significant contribution of Archaea to extant biomass in marine subsurface sediments. Nature 454, 991–994 (2008)
Teske, A. & Sørensen, K. B. Uncultured archaea in deep marine subsurface sediments: have we caught them all? ISME J. 2, 3–18 (2008)
Kubo, K. et al. Archaea of the Miscellaneous Crenarchaeotal Group are abundant, diverse and widespread in marine sediments. ISME J. 6, 1949–1965 (2012)
Meysman, F. J. R. & Middelburg, J. J. Acid-volatile sulfide (AVS) — a comment. Mar. Chem. 97, 206–212 (2005)
Holmkvist, L., Ferdelman, T. G. & Jørgensen, B. B. A cryptic sulfur cycle driven by iron in the methane zone of marine sediment (Aarhus Bay, Denmark). Geochim. Cosmochim. Acta 75, 3581–3599 (2011)
Philippot, P. et al. Early Archaean microorganisms preferred elemental sulfur, not sulfate. Science 317, 1534–1537 (2007)
Stumm, W. & Morgan, J. J. in Aquatic Chemistry: Chemical Equilibria and Rates in Natural Waters 3rd edn 990–1003 (Wiley-Interscience, 1996)
Kamyshny, A., Goifma, A., Gun, J., Rizkov, D. & Lev, O. Equilibrium distribution of polysulfide ions in aqueous solutions at 25°C: a new approach for the study of polysulfide’s equilibrium. Environ. Sci. Technol. 38, 6633–6644 (2004)
Mastalerz, V., de Lange, G. J., Dählmann, A. & Feseker, T. Active venting at the Isis mud volcano, offshore Egypt: origin and migration of hydrocarbons. Chem. Geol. 246, 87–106 (2007)
Widdel, F. & Bak, F. in The Prokaryotes (eds. Balows, A. T. et al.) Vol. 4 3352–3378 (Springer, 1992)
Cord-Ruwisch, R. A quick method for the determination of dissolved and precipitated sulfides in cultures of sulfate-reducing bacteria. J. Microbiol. Methods 4, 33–36 (1985)
Saleh, A. M., Macpherson, R. & Miller, J. D. A. The effect of inhibitors on sulphate reducing bacteria: a compilation. J. Appl. Bacteriol. 27, 281–293 (1964)
Cline, J. D. Spectrophotometric determination of hydrogen sulfide in natural waters. Limnol. Oceanogr. 14, 454–458 (1969)
Steudel, R., Göbel, T. & Holdt, G. The molecular composition of hydrophilic sulfur sols prepared by acid decomposition of thiosulfate. Z. Naturforsch. 43b, 203–218 (1988)
Musat, N. et al. A single-cell view on the ecophysiology of anaerobic phototrophic bacteria. Proc. Natl Acad. Sci. USA 105, 17861–17866 (2008)
Tokuyasu, K. T. Technique for ultracryotomy of cell suspensions and tissues. J. Cell Biol. 57, 551–565 (1973)
Milucka, J., Widdel, F. & Shima, S. Immunological detection of enzymes for sulfate reduction in anaerobic methane-oxidizing consortia. Environ. Microbiol. http://dx.doi.org/10.1111/1462-2920.12003 (28 September 2012)
Huang, W. E. et al. Raman-FISH: combining stable-isotope Raman spectroscopy and fluorescence in situ hybridization for the single cell analysis of identity and function. Environ. Microbiol. 9, 1878–1889 (2007)
Stahl, D. A. & Amann, R. in Nucleic Acid Techniques in Bacterial Systematics (eds. Stackebrandt, E. & Goodfellow, M. ) 205–248 (John Wiley, 1991)
Daims, H., Brühl, A., Amann, R., Schleifer, K. H. & Wagner, M. Probe EUB338 is insufficient for the detection of all Bacteria: development and evaluation of a more comprehensive probe set. Syst. Appl. Microbiol. 22, 434–444 (1999)
Stoecker, K., Dorninger, C., Daims, H. & Wagner, M. Double-labeling of oligonucleotide probes for fluorescence in situ hybridization (DOPE-FISH) improves signal intensity and increases rRNA accessibility. Appl. Environ. Microbiol. 76, 922–926 (2010)
Loy, A., Maixner, F., Wagner, M. & Horn, M. probeBase—an online resource for rRNA-targeted oligonucleotide probes: new features 2007. Nucleic Acids Res. 35, D800–D804 (2007)
Polerecky, L. et al. Look@NanoSIMS – a tool for the analysis of nanoSIMS data in environmental microbiology Environ. Microbiol. 14, 1009–1023 (2012)
Ploug, H. et al. Carbon and nitrogen fluxes associated with the cyanobacterium Aphanizomenon sp. in the Baltic Sea. ISME J. 4, 1215–1223 (2010)
Zopfi, J., Ferdelman, T. G. & Fossing, H. in Sulfur Biogeochemistry - Past and Present (eds. Amend, J., Edwards, K. J. & Lyons, T. W. ) Vol. 379 97–116 (The Geological Society of America Special Paper, 2004)
Kamyshny, A. Solubility of cyclooctasulfur in pure water and sea water at different temperatures. Geochim. Cosmochim. Acta 73, 6022–6028 (2009)
Kamyshny, A., Gun, J., Rizkov, D., Voitsekovski, T. & Lev, O. Equilibrium distributions of polysulfide ions in aqueous solutions at different temperatures by rapid phase derivitization. Environ. Sci. Technol. 41, 2395–2400 (2007)
Kamyshny, A., Borkenstein, C. G. & Ferdelman, T. G. Protocol for quantitative detection of elemental sulfur and polysulfide zero-valent sulfur distribution in natural aquatic samples. Geostand. Geoanal. Res. 33, 415–435 (2009)
Thode, H. G., Monster, J. & Dunford, H. B. Sulphur isotope geochemistry. Geochim. Cosmochim. Acta 25, 159–174 (1961)
Kallmeyer, J., Ferdelman, T. G., Weber, A., Fossing, H. & Jørgensen, B. B. A cold chromium distillation procedure for radiolabeled sulfide applied to sulfate reduction measurements. Limnol. Oceanogr. Methods 2, 171–180 (2004)
Jørgensen, B. B. & Fenchel, T. Sulfur cycle of a marine sediment model system. Mar. Biol. 24, 189–201 (1974)
Treude, T., Boetius, A., Knittel, K., Wallmann, K. & Jørgensen, B. B. Anaerobic oxidation of methane above gas hydrates at Hydrate Ridge, NE Pacific Ocean. Mar. Ecol. Prog. Ser. 264, 1–14 (2003)
Acknowledgements
We want to thank G. Klockgether, K. Imhoff, S. Littmann and T. Vagner for technical support, G. Lavik and M. Formolo for analytical support and discussions, M. Schüler and T. Keil for assistance with cryosectioning, A. Boetius for providing samples, and T. Holler for the initial enrichment and maintenance of the Isis culture. This work was financially supported by the Max Planck Society and the ERC Advanced Grant 294343 (to M.W.).
Author information
Authors and Affiliations
Contributions
J.M., T.G.F., F.W. and M.M.M.K. designed experiments. J.M., T.G.F. and M.M.M.K. performed experiments and analysed data. J.M. performed immunolabeling and microscopy. D.F. performed CARD-FISH and nanoSIMS. L.P. analysed nanoSIMS data. M.S. and M.W. performed Raman measurements, I.L. performed energy-dispersive X-ray spectroscopy analyses, and G.W. contributed new analytical tools. J.M., T.G.F. and M.M.M.K. wrote the manuscript with contributions of all co-authors.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
This file contains Supplementary Tables 1-5, a Supplementary Discussion, Supplementary Figures 1-15 and Supplementary References. (PDF 2668 kb)
Rights and permissions
About this article
Cite this article
Milucka, J., Ferdelman, T., Polerecky, L. et al. Zero-valent sulphur is a key intermediate in marine methane oxidation. Nature 491, 541–546 (2012). https://doi.org/10.1038/nature11656
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature11656
This article is cited by
-
Geochemical transformations of sulfur and their role in the formation of different types and subtypes of saline lakes in Southeastern Transbaikalia
Applied Water Science (2024)
-
Vertically stratified methane, nitrogen and sulphur cycling and coupling mechanisms in mangrove sediment microbiomes
Microbiome (2023)
-
Evidence of Arctic methane emissions across the mid-Pleistocene
Communications Earth & Environment (2023)
-
Opportunities and challenges of using metagenomic data to bring uncultured microbes into cultivation
Microbiome (2022)
-
Sulfate differentially stimulates but is not respired by diverse anaerobic methanotrophic archaea
The ISME Journal (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.