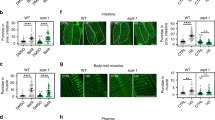
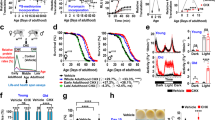
Abstract
Organisms that protect their germ-cell lineages from damage often do so at considerable cost: limited metabolic resources become partitioned away from maintenance of the soma, leaving the ageing somatic tissues to navigate survival amid an environment containing damaged and poorly functioning proteins. Historically, experimental paradigms that limit reproductive investment result in lifespan extension. We proposed that germline-deficient animals might exhibit heightened protection from proteotoxic stressors in somatic tissues. We find that the forced re-investment of resources from the germ line to the soma in Caenorhabditis elegans results in elevated somatic proteasome activity, clearance of damaged proteins and increased longevity. This activity is associated with increased expression of rpn-6, a subunit of the 19S proteasome, by the FOXO transcription factor DAF-16. Ectopic expression of rpn-6 is sufficient to confer proteotoxic stress resistance and extend lifespan, indicating that rpn-6 is a candidate to correct deficiencies in age-related protein homeostasis disorders.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Kirkwood, T. B. Evolution of ageing. Nature 270, 301–304 (1977)
Kenyon, C. A pathway that links reproductive status to lifespan in Caenorhabditis elegans . Ann. NY Acad. Sci. 1204, 156–162 (2010)
Partridge, L., Gems, D. & Withers, D. J. Sex and death: what is the connection? Cell 120, 461–472 (2005)
Shanley, D. P. & Kirkwood, T. B. Calorie restriction and aging: a life-history analysis. Evolution 54, 740–750 (2000)
Arantes-Oliveira, N., Apfeld, J., Dillin, A. & Kenyon, C. Regulation of life-span by germ-line stem cells in Caenorhabditis elegans . Science 295, 502–505 (2002)
Hsin, H. & Kenyon, C. Signals from the reproductive system regulate the lifespan of C. elegans . Nature 399, 362–366 (1999)
Wang, M. C., O’Rourke, E. J. & Ruvkun, G. Fat metabolism links germline stem cells and longevity in C. elegans . Science 322, 957–960 (2008)
Lin, K., Hsin, H., Libina, N. & Kenyon, C. Regulation of the Caenorhabditis elegans longevity protein DAF-16 by insulin/IGF-1 and germline signaling. Nature Genet. 28, 139–145 (2001)
Berman, J. R. & Kenyon, C. Germ-cell loss extends C. elegans life span through regulation of DAF-16 by kri-1 and lipophilic-hormone signaling. Cell 124, 1055–1068 (2006)
Gerisch, B., Weitzel, C., Kober-Eisermann, C., Rottiers, V. & Antebi, A. A hormonal signaling pathway influencing C. elegans metabolism, reproductive development, and life span. Dev. Cell 1, 841–851 (2001)
Bence, N. F., Sampat, R. M. & Kopito, R. R. Impairment of the ubiquitin-proteasome system by protein aggregation. Science 292, 1552–1555 (2001)
Bennett, E. J., Bence, N. F., Jayakumar, R. & Kopito, R. R. Global impairment of the ubiquitin-proteasome system by nuclear or cytoplasmic protein aggregates precedes inclusion body formation. Mol. Cell 17, 351–365 (2005)
Powers, E. T., Morimoto, R. I., Dillin, A., Kelly, J. W. & Balch, W. E. Biological and chemical approaches to diseases of proteostasis deficiency. Annu. Rev. Biochem. 78, 959–991 (2009)
Lakowski, B. & Hekimi, S. The genetics of caloric restriction in Caenorhabditis elegans . Proc. Natl Acad. Sci. USA 95, 13091–13096 (1998)
Dillin, A. et al. Rates of behavior and aging specified by mitochondrial function during development. Science 298, 2398–2401 (2002)
Hamer, G., Matilainen, O. & Holmberg, C. I. A photoconvertible reporter of the ubiquitin-proteasome system in vivo . Nature Methods 7, 473–478 (2010)
Mitchell, D. H., Stiles, J. W., Santelli, J. & Sanadi, D. R. Synchronous growth and aging of Caenorhabditis elegans in the presence of fluorodeoxyuridine. J. Gerontol. 34, 28–36 (1979)
Priess, J. R., Schnabel, H. & Schnabel, R. The glp-1 locus and cellular interactions in early C. elegans embryos. Cell 51, 601–611 (1987)
Dillin, A., Crawford, D. K. & Kenyon, C. Timing requirements for insulin/IGF-1 signaling in C. elegans . Science 298, 830–834 (2002)
Hansen, M., Hsu, A. L., Dillin, A. & Kenyon, C. New genes tied to endocrine, metabolic, and dietary regulation of lifespan from a Caenorhabditis elegans genomic RNAi screen. PLoS Genet. 1, e17 (2005)
Cohen, E., Bieschke, J., Perciavalle, R. M., Kelly, J. W. & Dillin, A. Opposing activities protect against age-onset proteotoxicity. Science 313, 1604–1610 (2006)
Hsu, A. L., Murphy, C. T. & Kenyon, C. Regulation of aging and age-related disease by DAF-16 and heat-shock factor. Science 300, 1142–1145 (2003)
Alam, J. et al. Nrf2, a Cap’n’Collar transcription factor, regulates induction of the heme oxygenase-1 gene. J. Biol. Chem. 274, 26071–26078 (1999)
An, J. H. & Blackwell, T. K. SKN-1 links C. elegans mesendodermal specification to a conserved oxidative stress response. Genes Dev. 17, 1882–1893 (2003)
Goudeau, J. et al. Fatty acid desaturation links germ cell loss to longevity through NHR-80/HNF4 in C. elegans . PLoS Biol. 9, e1000599 (2011)
Finley, D. Recognition and processing of ubiquitin-protein conjugates by the proteasome. Annu. Rev. Biochem. 78, 477–513 (2009)
Kisselev, A. F. & Goldberg, A. L. Monitoring activity and inhibition of 26S proteasomes with fluorogenic peptide substrates. Methods Enzymol. 398, 364–378 (2005)
Köhler, A. et al. The axial channel of the proteasome core particle is gated by the Rpt2 ATPase and controls both substrate entry and product release. Mol. Cell 7, 1143–1152 (2001)
Pathare, G. R. et al. The proteasomal subunit Rpn6 is a molecular clamp holding the core and regulatory subcomplexes together. Proc. Natl Acad. Sci. USA 109, 149–154 (2012)
Santamaria, P. G., Finley, D., Ballesta, J. P. & Remacha, M. Rpn6p, a proteasome subunit from Saccharomyces cerevisiae, is essential for the assembly and activity of the 26 S proteasome. J. Biol. Chem. 278, 6687–6695 (2003)
Furuyama, T., Nakazawa, T., Nakano, I. & Mori, N. Identification of the differential distribution patterns of mRNAs and consensus binding sequences for mouse DAF-16 homologues. Biochem. J. 349, 629–634 (2000)
Celniker, S. E. et al. Unlocking the secrets of the genome. Nature 459, 927–930 (2009)
Ghazi, A., Henis-Korenblit, S. & Kenyon, C. Regulation of Caenorhabditis elegans lifespan by a proteasomal E3 ligase complex. Proc. Natl Acad. Sci. USA 104, 5947–5952 (2007)
Li, X. J. &. L. i. S. Proteasomal dysfunction in aging and Huntington disease. Neurobiol. Dis. 43, 4–8 (2011)
Brignull, H. R., Moore, F. E., Tang, S. J. & Morimoto, R. I. Polyglutamine proteins at the pathogenic threshold display neuron-specific aggregation in a pan-neuronal Caenorhabditis elegans model. J. Neurosci. 26, 7597–7606 (2006)
Melendez, A. et al. Autophagy genes are essential for dauer development and life-span extension in C. elegans . Science 301, 1387–1391 (2003)
Morley, J. F. & Morimoto, R. I. Regulation of longevity in Caenorhabditis elegans by heat shock factor and molecular chaperones. Mol. Biol. Cell 15, 657–664 (2004)
Hansen, M. et al. A role for autophagy in the extension of lifespan by dietary restriction in C. elegans . PLoS Genet. 4, e24 (2008)
Lapierre, L. R., Melendez, A. & Hansen, M. Autophagy links lipid metabolism to longevity in C. elegans . Autophagy 8, 144–146 (2012)
Zabel, C. et al. Proteasome and oxidative phoshorylation changes may explain why aging is a risk factor for neurodegenerative disorders. J. Proteomics 73, 2230–2238 (2010)
Brenner, S. The genetics of Caenorhabditis elegans . Genetics 77, 71–94 (1974)
Fire, A. et al. Potent and specific genetic interference by double-stranded RNA in Caenorhabditis elegans . Nature 391, 806–811 (1998)
Chai, Y., Shao, J., Miller, V. M., Williams, A. & Paulson, H. L. Live-cell imaging reveals divergent intracellular dynamics of polyglutamine disease proteins and supports a sequestration model of pathogenesis. Proc. Natl Acad. Sci. USA 99, 9310–9315 (2002)
Mello, C. C., Kramer, J. M., Stinchcomb, D. & Ambros, V. Efficient gene transfer in C.elegans: extrachromosomal maintenance and integration of transforming sequences. EMBO J. 10, 3959–3970 (1991)
Vazquez-Manrique, R. P. et al. Reduction of Caenorhabditis elegans frataxin increases sensitivity to oxidative stress, reduces lifespan, and causes lethality in a mitochondrial complex II mutant. FASEB J. 20, 172–174 (2006)
Wolff, S. et al. SMK-1, an essential regulator of DAF-16-mediated longevity. Cell 124, 1039–1053 (2006)
Hoogewijs, D., Houthoofd, K., Matthijssens, F., Vandesompele, J. & Vanfleteren, J. R. Selection and validation of a set of reliable reference genes for quantitative sod gene expression analysis in C. elegans . BMC Mol. Biol. 9, 9 (2008)
Acknowledgements
We thank S. Panowski for help with the generation of transgenic strains. We thank D. Joyce for proteasome activity assays and S. Wolff for comments on the manuscript. This work was supported by HHMI and the NIA. D.V. was a recipient of the F.M. Kirby, Inc. Foundation Postdoctoral Scholar Award and Beatriu de Pinós (AGAUR) fellowship.
Author information
Authors and Affiliations
Contributions
D.V. and A.D. planned and supervised the project. D.V. performed the experiments, data analysis and interpretation. I.M. performed biochemistry experiments and contributed to other assays. Z.L. performed UPS reporter experiment, lifespans and injections. P.M.D. performed the filter trap assay. C.M. performed immunoblots. A.P.C.R. and G.M. performed the transcription factor binding site analysis. The manuscript was written by D.V. and A.D. and edited by I.M. and C.M. All authors discussed the results and commented on the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
This file contains Supplementary Figures 1-22, Supplementary Tables 1-4 and 6-7. (PDF 12476 kb)
Supplementary Data
This file contains Supplementary Table 5. (XLS 26 kb)
Rights and permissions
About this article
Cite this article
Vilchez, D., Morantte, I., Liu, Z. et al. RPN-6 determines C. elegans longevity under proteotoxic stress conditions. Nature 489, 263–268 (2012). https://doi.org/10.1038/nature11315
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature11315
This article is cited by
-
Endogenous DAF-16 spatiotemporal activity quantitatively predicts lifespan extension induced by dietary restriction
Communications Biology (2023)
-
Cold temperature extends longevity and prevents disease-related protein aggregation through PA28γ-induced proteasomes
Nature Aging (2023)
-
piRNAs regulate a Hedgehog germline-to-soma pro-aging signal
Nature Aging (2023)
-
The RPN12a proteasome subunit is essential for the multiple hormonal homeostasis controlling the progression of leaf senescence
Communications Biology (2022)
-
The membrane domain of respiratory complex I accumulates during muscle aging in Drosophila melanogaster
Scientific Reports (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.