Abstract
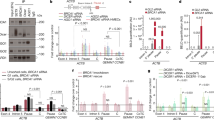
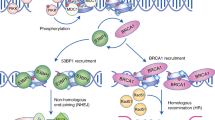
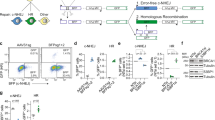
Non-coding RNAs (ncRNAs) are involved in an increasingly recognized number of cellular events1. Some ncRNAs are processed by DICER and DROSHA RNases to give rise to small double-stranded RNAs involved in RNA interference (RNAi)2. The DNA-damage response (DDR) is a signalling pathway that originates from a DNA lesion and arrests cell proliferation3. So far, DICER and DROSHA RNA products have not been reported to control DDR activation. Here we show, in human, mouse and zebrafish, that DICER and DROSHA, but not downstream elements of the RNAi pathway, are necessary to activate the DDR upon exogenous DNA damage and oncogene-induced genotoxic stress, as studied by DDR foci formation and by checkpoint assays. DDR foci are sensitive to RNase A treatment, and DICER- and DROSHA-dependent RNA products are required to restore DDR foci in RNase-A-treated cells. Through RNA deep sequencing and the study of DDR activation at a single inducible DNA double-strand break, we demonstrate that DDR foci formation requires site-specific DICER- and DROSHA-dependent small RNAs, named DDRNAs, which act in a MRE11–RAD50–NBS1-complex-dependent manner (MRE11 also known as MRE11A; NBS1 also known as NBN). DDRNAs, either chemically synthesized or in vitro generated by DICER cleavage, are sufficient to restore the DDR in RNase-A-treated cells, also in the absence of other cellular RNAs. Our results describe an unanticipated direct role of a novel class of ncRNAs in the control of DDR activation at sites of DNA damage.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Accession codes
Data deposits
Sequence data have been deposited in the DNA Data Bank of Japan under accession code DRA000540.
References
Esteller, M. Non-coding RNAs in human disease. Nature Rev. Genet. 12, 861–874 (2011)
Krol, J., Loedige, I. & Filipowicz, W. The widespread regulation of microRNA biogenesis, function and decay. Nature Rev. Genet. 11, 597–610 (2010)
Jackson, S. P. & Bartek, J. The DNA-damage response in human biology and disease. Nature 461, 1071–1078 (2009)
Clark, M. B. et al. The reality of pervasive transcription. PLoS Biol. 9, e1000625 (2011)
Wilusz, J. E., Sunwoo, H. & Spector, D. L. Long noncoding RNAs: functional surprises from the RNA world. Genes Dev. 23, 1494–1504 (2009)
Wang, X. et al. Induced ncRNAs allosterically modify RNA-binding proteins in cis to inhibit transcription. Nature 454, 126–130 (2008)
Zhao, J., Sun, B. K., Erwin, J. A., Song, J. J. & Lee, J. T. Polycomb proteins targeted by a short repeat RNA to the mouse X chromosome. Science 322, 750–756 (2008)
Mercer, T. R., Dinger, M. E. & Mattick, J. S. Long non-coding RNAs: insights into functions. Nature Rev. Genet. 10, 155–159 (2009)
Kim, V. N., Han, J. & Siomi, M. C. Biogenesis of small RNAs in animals. Nature Rev. Mol. Cell Biol. 10, 126–139 (2009)
Lukas, J., Lukas, C. & Bartek, J. More than just a focus: the chromatin response to DNA damage and its role in genome integrity maintenance. Nature Cell Biol. 13, 1161–1169 (2011)
d’Adda di Fagagna, F. Living on a break: cellular senescence as a DNA-damage response. Nature Rev. Cancer 8, 512–522 (2008)
Narita, M. et al. Rb-mediated heterochromatin formation and silencing of E2F target genes during cellular senescence. Cell 113, 703–716 (2003)
White, S. A. & Allshire, R. C. RNAi-mediated chromatin silencing in fission yeast. Curr. Top. Microbiol. Immunol. 320, 157–183 (2008)
Di Micco, R. et al. Oncogene-induced senescence is a DNA damage response triggered by DNA hyper-replication. Nature 444, 638–642 (2006)
Tritschler, F., Huntzinger, E. & Izaurralde, E. Role of GW182 proteins and PABPC1 in the miRNA pathway: a sense of deja vu. Nature Rev. Mol. Cell Biol. 11, 379–384 (2010)
Zhang, H., Kolb, F. A., Jaskiewicz, L., Westhof, E. & Filipowicz, W. Single processing center models for human Dicer and bacterial RNase III. Cell 118, 57–68 (2004)
Nicoli, S. et al. MicroRNA-mediated integration of haemodynamics and Vegf signalling during angiogenesis. Nature 464, 1196–1200 (2010)
Cummins, J. M. et al. The colorectal microRNAome. Proc. Natl Acad. Sci. USA 103, 3687–3692 (2006)
Wienholds, E. et al. MicroRNA expression in zebrafish embryonic development. Science 309, 310–311 (2005)
Maison, C. et al. Higher-order structure in pericentric heterochromatin involves a distinct pattern of histone modification and an RNA component. Nature Genet. 30, 329–334 (2002)
Pryde, F. et al. 53BP1 exchanges slowly at the sites of DNA damage and appears to require RNA for its association with chromatin. J. Cell Sci. 118, 2043–2055 (2005)
Berkovich, E., Monnat, R. J., Jr & Kastan, M. B. Roles of ATM and NBS1 in chromatin structure modulation and DNA double-strand break repair. Nature Cell Biol. 9, 683–690 (2007)
Iacovoni, J. S. et al. High-resolution profiling of γH2AX around DNA double strand breaks in the mammalian genome. EMBO J. 29, 1446–1457 (2010)
Soutoglou, E. et al. Positional stability of single double-strand breaks in mammalian cells. Nature Cell Biol. 9, 675–682 (2007)
Stracker, T. H. & Petrini, J. H. The MRE11 complex: starting from the ends. Nature Rev. Mol. Cell Biol. 12, 90–103 (2011)
Dupré, A. et al. A forward chemical genetic screen reveals an inhibitor of the Mre11–Rad50–Nbs1 complex. Nature Chem. Biol. 4, 119–125 (2008)
Duchaine, T. F. et al. Functional proteomics reveals the biochemical niche of C. elegans DCR-1 in multiple small-RNA-mediated pathways. Cell 124, 343–354 (2006)
Sidi, S. et al. Chk1 suppresses a caspase-2 apoptotic response to DNA damage that bypasses p53, Bcl-2, and caspase-3. Cell 133, 864–877 (2008)
Wienholds, E., Koudijs, M. J., van Eeden, F. J., Cuppen, E. & Plasterk, R. H. The microRNA-producing enzyme Dicer1 is essential for zebrafish development. Nature Genet. 35, 217–218 (2003)
Kawano, M. et al. Reduction of non-insert sequence reads by dimer eliminator LNA oligonucleotide for small RNA deep sequencing. Biotechniques 49, 751–755 (2010)
Acknowledgements
We thank E. Soutoglou, W. C. Hahn, M. Kastan, V. Orlando, R. Shiekhattar, J. Amatruda, T. Halazonetis, E. Dejana, P. Ng and F. Nicassio for sharing reagents, M. Fumagalli and F. Rossiello for reading the manuscript, M. Dobreva, V. Matti and F. Pezzimenti for technical support, G. D’Ario for help with statistical analyses, B. Amati, M. Foiani, V. Costanzo and F.d.A.d.F. group members for help and discussions. The F.d.A.d.F. laboratory was supported by Fondazione Italiana Ricerca Sul Cancro (FIRC), Associazione Italiana Ricerca sul Cancro (AIRC) European Community’s 7th Framework Programme (FP7/2007-2013) under grant agreement no. 202230, acronym “GENINCA”, HFSP, AICR, the EMBO Young Investigator Program. The initial part of this project was supported by Telethon grant no. GGP08183. P.C. was supported by 7th Framework of the European Union commission to the Dopaminet consortium, a Grant-in-Aids for Scientific Research (A) no. 20241047, Funding Program for the Next Generation World-Leading Researchers (NEXT Program) to P.C. and a Research Grant to RIKEN Omics Science Center from MEXT. S.F. is supported by Center for Genomic Science of IIT@SEMM (Scuola Europea di Medicina Molecolare) and AIRC. M.M. was supported by Cariplo (grant no. 2007-5500) and AIRC. A.S. is supported by a JSPS fellowship P09745 and grant in aid by JSPS, and D.T. is supported by the European Union 7th Framework Programme under grant agreement FP7-People-ITN-2008-238055 (“BrainTrain” project) to P.C.
Author information
Authors and Affiliations
Contributions
A.S., D.T. and P.C. planned, generated and analysed the genomics data presented in Supplementary Figs 20a–e, 21, 22b and 23. M.d.H. performed statistical analysis of the genomics data. A.S. and P.C. also edited the manuscript. M.M. and V.A. generated the data presented in Supplementary Figs 14 and 15. F.M. generated the data shown in Figs 2b, 3d, e, 4b and Supplementary Figs 2b, e, 3e, 4b, 5f, g, 6b–d, 7d, 9, 13d–f, 14d, f, 17f, g, 18a, b, 19, 20g, h and 22a and generated RNA for deep sequencing; contributed to: Supplementary Figs 16a, 5d, e, 11c, d and edited the manuscript. S.F. generated the data shown in remaining figures, contributed to experimental design and edited the manuscript. F.d.A.d.F. conceived the study, designed the experiments and wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
This file contains Supplementary Introduction, Supplementary Figures 1-23, a Supplementary Discussion and additional references. (PDF 9693 kb)
Rights and permissions
About this article
Cite this article
Francia, S., Michelini, F., Saxena, A. et al. Site-specific DICER and DROSHA RNA products control the DNA-damage response. Nature 488, 231–235 (2012). https://doi.org/10.1038/nature11179
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature11179
This article is cited by
-
Death Induced by Survival gene Elimination (DISE) correlates with neurotoxicity in Alzheimer’s disease and aging
Nature Communications (2024)
-
Integrative modeling of lncRNA-chromatin interaction maps reveals diverse mechanisms of nuclear retention
BMC Genomics (2023)
-
ATR-binding lncRNA ScaRNA2 promotes cancer resistance through facilitating efficient DNA end resection during homologous recombination repair
Journal of Experimental & Clinical Cancer Research (2023)
-
DNA double-strand break end synapsis by DNA loop extrusion
Nature Communications (2023)
-
The RNA m5C modification in R-loops as an off switch of Alt-NHEJ
Nature Communications (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.