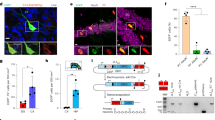
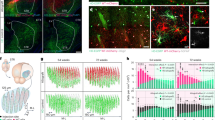
Abstract
Human pluripotent stem cells (PSCs) are a promising source of cells for applications in regenerative medicine. Directed differentiation of PSCs into specialized cells such as spinal motoneurons1 or midbrain dopamine (DA) neurons2 has been achieved. However, the effective use of PSCs for cell therapy has lagged behind. Whereas mouse PSC-derived DA neurons have shown efficacy in models of Parkinson’s disease3,4, DA neurons from human PSCs generally show poor in vivo performance5. There are also considerable safety concerns for PSCs related to their potential for teratoma formation or neural overgrowth6,7. Here we present a novel floor-plate-based strategy for the derivation of human DA neurons that efficiently engraft in vivo, suggesting that past failures were due to incomplete specification rather than a specific vulnerability of the cells. Midbrain floor-plate precursors are derived from PSCs 11 days after exposure to small molecule activators of sonic hedgehog (SHH) and canonical WNT signalling. Engraftable midbrain DA neurons are obtained by day 25 and can be maintained in vitro for several months. Extensive molecular profiling, biochemical and electrophysiological data define developmental progression and confirm identity of PSC-derived midbrain DA neurons. In vivo survival and function is demonstrated in Parkinson’s disease models using three host species. Long-term engraftment in 6-hydroxy-dopamine-lesioned mice and rats demonstrates robust survival of midbrain DA neurons derived from human embryonic stem (ES) cells, complete restoration of amphetamine-induced rotation behaviour and improvements in tests of forelimb use and akinesia. Finally, scalability is demonstrated by transplantation into parkinsonian monkeys. Excellent DA neuron survival, function and lack of neural overgrowth in the three animal models indicate promise for the development of cell-based therapies in Parkinson’s disease.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Change history
21 December 2011
In the online-only Methods, in the "Neural induction" subsection, the concentration of DAPT appeared incorrectly as "10 nM" in the AOP PDF version. This has been corrected.
References
Li, X. J. et al. Specification of motoneurons from human embryonic stem cells. Nature Biotechnol. 23, 215–221 (2005)
Perrier, A. L. et al. From the cover: derivation of midbrain dopamine neurons from human embryonic stem cells. Proc. Natl Acad. Sci. USA 101, 12543–12548 (2004)
Tabar, V. et al. Therapeutic cloning in individual parkinsonian mice. Nature Med. 14, 379–381 (2008)
Wernig, M. et al. Neurons derived from reprogrammed fibroblasts functionally integrate into the fetal brain and improve symptoms of rats with Parkinson’s disease. Proc. Natl Acad. Sci. USA 105, 5856–5861 (2008)
Lindvall, O. & Kokaia, Z. Stem cells in human neurodegenerative disorders–time for clinical translation? J. Clin. Invest. 120, 29–40 (2010)
Elkabetz, Y. et al. Human ES cell-derived neural rosettes reveal a functionally distinct early neural stem cell stage. Genes Dev. 22, 152–165 (2008)
Roy, N. S. et al. Functional engraftment of human ES cell-derived dopaminergic neurons enriched by coculture with telomerase-immortalized midbrain astrocytes. Nature Med. 12, 1259–1268 (2006)
Kittappa, R., Chang, W. W., Awatramani, R. B. & McKay, R. D. The foxa2 gene controls the birth and spontaneous degeneration of dopamine neurons in old age. PLoS Biol. 5, e325 (2007)
Ferri, A. L. et al. Foxa1 and Foxa2 regulate multiple phases of midbrain dopaminergic neuron development in a dosage-dependent manner. Development 134, 2761–2769 (2007)
Roelink, H. et al. Floor plate and motor neuron induction by vhh-1, a vertebrate homolog of hedgehog expressed by the notochord. Cell 76, 761–775 (1994)
Liem, K. F., Tremml, G., Roelink, H. & Jessell, T. M. Dorsal differentiation of neural plate cells induced by BMP-mediated signals from epidermal ectoderm. Cell 82, 969–979 (1995)
Fasano, C. A., Chambers, S. M., Lee, G., Tomishima, M. J. & Studer, L. Efficient derivation of functional floor plate tissue from human embryonic stem cells. Cell Stem Cell 6, 336–347 (2010)
Chambers, S. M. et al. Highly efficient neural conversion of human ES and iPS cells by dual inhibition of SMAD signaling. Nature Biotechnol. 27, 275–280 (2009)
Muroyama, Y., Fujihara, M., Ikeya, M., Kondoh, H. & Takada, S. Wnt signaling plays an essential role in neuronal specification of the dorsal spinal cord. Genes Dev. 16, 548–553 (2002)
Joksimovic, M. et al. Wnt antagonism of Shh facilitates midbrain floor plate neurogenesis. Nature Neurosci. 12, 125–131 (2009)
Lyashenko, N. et al. Differential requirement for the dual functions of beta-catenin in embryonic stem cell self-renewal and germ layer formation. Nature Cell Biol. 13, 753–761 (2011)
VanDunk, C., Hunter, L. A. & Gray, P. A. Development, maturation, and necessity of transcription factors in the mouse suprachiasmatic nucleus. J. Neurosci. 31, 6457–6467 (2011)
Huang, W., Sherman, B. T. & Lempicki, R. A. Systematic and integrative analysis of large gene lists using DAVID bioinformatics resources. Nature Protocols 4, 44–57 (2009)
Costa, R. H., Grayson, D. R. & Darnell, J. E., Jr Multiple hepatocyte-enriched nuclear factors function in the regulation of transthyretin and alpha 1-antitrypsin genes. Mol. Cell. Biol. 9, 1415–1425 (1989)
Soldner, F. et al. Parkinson’s disease patient-derived induced pluripotent stem cells free of viral reprogramming factors. Cell 136, 964–977 (2009)
Guzman, J. N., Sanchez-Padilla, J., Chan, C. S. & Surmeier, D. J. Robust pacemaking in substantia nigra dopaminergic neurons. J. Neurosci. 29, 11011–11019 (2009)
Nedergaard, S., Flatman, J. A. & Engberg, I. Nifedipine- and omega-conotoxin-sensitive Ca2+ conductances in guinea-pig substantia nigra pars compacta neurones. J. Physiol. (Lond.) 466, 727–747 (1993)
Ferrari, D., Sanchez-Pernaute, R., Lee, H., Studer, L. & Isacson, O. Transplanted dopamine neurons derived from primate ES cells preferentially innervate DARPP-32 striatal progenitors within the graft. Eur. J. Neurosci. 24, 1885–1896 (2006)
Olanow, C. W., Kordower, J. H. & Freeman, T. B. Fetal nigral transplantation as a therapy for Parkinson’s disease. Trends Neurosci. 19, 102–109 (1996)
Zetterström, R. H. et al. Dopamine neuron agenesis in Nurr1-deficient mice. Science 276, 248–250 (1997)
Quintana, E. et al. Efficient tumour formation by single human melanoma cells. Nature 456, 593–598 (2008)
Kim, H. et al. miR-371-3 expression predicts neural differentiation propensity in human pluripotent stem cells. Cell Stem Cell 8, 695–706 (2011)
Hargus, G. et al. Differentiated Parkinson patient-derived induced pluripotent stem cells grow in the adult rodent brain and reduce motor asymmetry in Parkinsonian rats. Proc. Natl Acad. Sci. USA 107, 15921–15926 (2010)
Aubry, L. et al. Striatal progenitors derived from human ES cells mature into DARPP32 neurons in vitro and in quinolinic acid-lesioned rats. Proc. Natl Acad. Sci. USA 105, 16707–16712 (2008)
Ban, H. et al. Efficient generation of transgene-free human induced pluripotent stem cells (iPSCs) by temperature-sensitive Sendai virus vectors. Proc. Natl. Acad. Sci. USA 108, 14234–14239 (2011)
Studer, L., Tabar, V. & McKay, R. D. Transplantation of expanded mesencephalic precursors leads to recovery in parkinsonian rats. Nature Neurosci. 1, 290–295 (1998)
Kordower, J. H. et al. Neurodegeneration prevented by lentiviral vector delivery of GDNF in primate models of Parkinson’s disease. Science 290, 767–773 (2000)
Paxinos, G., Huang, X.-F. & Toga, A. W. The Rhesus Monkey Brain in Stereotaxic Coordinates (Academic Press, 2000)
Blume, S. R., Cass, D. K. & Tseng, K. Y. Stepping test in mice: a reliable approach in determining forelimb akinesia in MPTP-induced Parkinsonism. Exp. Neurol. 219, 208–211 (2009)
Crawley, J. N. What’s Wrong With My Mouse: Behavioral Phenotyping of Transgenic and Knockout Mice (Wiley, 2000)
Studer, L. et al. Noninvasive dopamine determination by reversed phase HPLC in the medium of free-floating roller tube cultures of rat fetal ventral mesencephalon: A tool to assess dopaminergic tissue prior to grafting. Brain Res. Bull. 41, 143–150 (1996)
Tabar, V. et al. Migration and differentiation of neural precursors derived from human embryonic stem cells in the rat brain. Nature Biotechnol. 23, 601–606 (2005)
Acknowledgements
We thank K. Manova, M. Tomishima and A. Viale for excellent technical support, and R. McKay for the anti-nestin antibody. The work was supported by NIH/NINDS grant NS052671, the European Commission project NeuroStemcell, the Starr foundation and NYSTEM contract C024414 to L.S.; by NYSTEM contract C024413, the Michael T. McCarthy Foundation and the Elkus Family Foundation to V.T.; by the Consolidated Anti-Aging Foundation to J.H.K.; by NIH/NINDS grant P50 NS047085, and support from Falk Medical Research Trust to D.J.S.; J.-W.S. was supported by NYSCF (Druckenmiller fellowship) and S.K. by a Starr stem cell scholar fellowship.
Author information
Authors and Affiliations
Contributions
S.K. and J.-W.S.: conception and study design, maintenance and directed differentiation of PSCs, cellular/molecular assays, histological analyses, mouse behavioural assays, data interpretation and writing of manuscript. J.P., G.A., C.A. and A.B.: rat transplantation, histological analyses and behavioural assays. Y.M.G.: mouse transplantation and histological analyses. D.R.W. and J.H.K.: monkey transplantation, histological analysis and data interpretation. L.Y. and M.F.B.: HPLC analysis and data interpretation. L.C.-R., Z.X and D.J.S.: electrophysiological analyses and data interpretation. V.T.: study design, data analysis and writing of manuscript. L.S.: conception and study design, data analysis and interpretation, and writing of manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
The file contains Supplementary Figures 1-11 with legends and Supplementary Tables 1-6.s (PDF 0 kb)
Rights and permissions
About this article
Cite this article
Kriks, S., Shim, JW., Piao, J. et al. Dopamine neurons derived from human ES cells efficiently engraft in animal models of Parkinson’s disease. Nature 480, 547–551 (2011). https://doi.org/10.1038/nature10648
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature10648
This article is cited by
-
Intracellular delivery of Parkin-RING0-based fragments corrects Parkin-induced mitochondrial dysfunction through interaction with SLP-2
Journal of Translational Medicine (2024)
-
Disruption of lysosomal proteolysis in astrocytes facilitates midbrain organoid proteostasis failure in an early-onset Parkinson’s disease model
Nature Communications (2024)
-
An epigenetic barrier sets the timing of human neuronal maturation
Nature (2024)
-
Unravelling the Parkinson’s puzzle, from medications and surgery to stem cells and genes: a comprehensive review of current and future management strategies
Experimental Brain Research (2024)
-
Identifying secreted biomarkers of dopaminergic ventral midbrain progenitor cells
Stem Cell Research & Therapy (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.