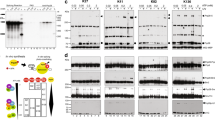
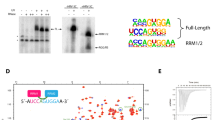
Abstract
Alternative splicing of pre-messenger RNAs diversifies gene products in eukaryotes and is guided by factors that enable spliceosomes to recognize particular splice sites. Here we report that alternative splicing of Saccharomyces cerevisiae SRC1 pre-mRNA is promoted by the conserved ubiquitin-like protein Hub1. Structural and biochemical data show that Hub1 binds non-covalently to a conserved element termed HIND, which is present in the spliceosomal protein Snu66 in yeast and mammals, and Prp38 in plants. Hub1 binding mildly alters spliceosomal protein interactions and barely affects general splicing in S. cerevisiae. However, spliceosomes that lack Hub1, or are defective in Hub1–HIND interaction, cannot use certain non-canonical 5′ splice sites and are defective in alternative SRC1 splicing. Hub1 confers alternative splicing not only when bound to HIND, but also when experimentally fused to Snu66, Prp38, or even the core splicing factor Prp8. Our study indicates a novel mechanism for splice site utilization that is guided by non-covalent modification of the spliceosome by an unconventional ubiquitin-like modifier.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Change history
09 June 2011
Figs 3c and 4b were corrected.
References
Hochstrasser, M. Origin and function of ubiquitin-like proteins. Nature 458, 422–429 (2009)
McNally, T. et al. Structural analysis of UBL5, a novel ubiquitin-like modifier. Protein Sci. 12, 1562–1566 (2003)
Ramelot, T. A. et al. Solution structure of the yeast ubiquitin-like modifier protein Hub1. J. Struct. Funct. Genomics 4, 25–30 (2003)
Wilkinson, C. R. et al. Ubiquitin-like protein Hub1 is required for pre-mRNA splicing and localization of an essential splicing factor in fission yeast. Curr. Biol. 14, 2283–2288 (2004)
Friedman, J. S., Koop, B. F., Raymond, V. & Walter, M. A. Isolation of a ubiquitin-like (UBL5) gene from a screen identifying highly expressed and conserved iris genes. Genomics 71, 252–255 (2001)
Dittmar, G. A., Wilkinson, C. R., Jedrzejewski, P. T. & Finley, D. Role of a ubiquitin-like modification in polarized morphogenesis. Science 295, 2442–2446 (2002)
Lüders, J., Pyrowolakis, G. & Jentsch, S. The ubiquitin-like protein HUB1 forms SDS-resistant complexes with cellular proteins in the absence of ATP. EMBO Rep. 4, 1169–1174 (2003)
Yashiroda, H. & Tanaka, K. Hub1 is an essential ubiquitin-like protein without functioning as a typical modifier in fission yeast. Genes Cells 9, 1189–1197 (2004)
Benedetti, C., Haynes, C. M., Yang, Y., Harding, H. P. & Ron, D. Ubiquitin-like protein 5 positively regulates chaperone gene expression in the mitochondrial unfolded protein response. Genetics 174, 229–239 (2006)
Hazbun, T. R. et al. Assigning function to yeast proteins by integration of technologies. Mol. Cell 12, 1353–1365 (2003)
Wahl, M. C., Will, C. L. & Luhrmann, R. The spliceosome: design principles of a dynamic RNP machine. Cell 136, 701–718 (2009)
Ramage, R. et al. Synthetic, structural and biological studies of the ubiquitin system: the total chemical synthesis of ubiquitin. Biochem. J. 299, 151–158 (1994)
Bayer, P. et al. Structure determination of the small ubiquitin-related modifier SUMO-1. J. Mol. Biol. 280, 275–286 (1998)
Dikic, I., Wakatsuki, S. & Walters, K. J. Ubiquitin-binding domains—from structures to functions. Nature Rev. Mol. Cell Biol. 10, 659–671 (2009)
Song, J., Zhang, Z., Hu, W. & Chen, Y. Small ubiquitin-like modifier (SUMO) recognition of a SUMO binding motif: a reversal of the bound orientation. J. Biol. Chem. 280, 40122–40129 (2005)
Makarova, O. V., Makarov, E. M. & Luhrmann, R. The 65 and 110 kDa SR-related proteins of the U4/U6.U5 tri-snRNP are essential for the assembly of mature spliceosomes. EMBO J. 20, 2553–2563 (2001)
Stevens, S. W. & Abelson, J. Purification of the yeast U4/U6.U5 small nuclear ribonucleoprotein particle and identification of its proteins. Proc. Natl Acad. Sci. USA 96, 7226–7231 (1999)
Stevens, S. W. et al. Composition and functional characterization of the yeast spliceosomal penta-snRNP. Mol. Cell 9, 31–44 (2002)
Fabrizio, P. et al. The evolutionarily conserved core design of the catalytic activation step of the yeast spliceosome. Mol. Cell 36, 593–608 (2009)
Clark, T. A., Sugnet, C. W. & Ares, M., Jr Genomewide analysis of mRNA processing in yeast using splicing-specific microarrays. Science 296, 907–910 (2002)
Burckin, T. et al. Exploring functional relationships between components of the gene expression machinery. Nature Struct. Mol. Biol. 12, 175–182 (2005)
Keren, H., Lev-Maor, G. & Ast, G. Alternative splicing and evolution: diversification, exon definition and function. Nature Rev. Genet. 11, 345–355 (2010)
Jacquier, A., Rodriguez, J. R. & Rosbash, M. A quantitative analysis of the effects of 5′ junction and TACTAAC box mutants and mutant combinations on yeast mRNA splicing. Cell 43, 423–430 (1985)
Lesser, C. F. & Guthrie, C. Mutational analysis of pre-mRNA splicing in Saccharomyces cerevisiae using a sensitive new reporter gene, CUP1 . Genetics 133, 851–863 (1993)
Chen, M. & Manley, J. L. Mechanisms of alternative splicing regulation: insights from molecular and genomics approaches. Nature Rev. Mol. Cell Biol. 10, 741–754 (2009)
Davis, C. A., Grate, L., Spingola, M. & Ares, M. Test of intron predictions reveals novel splice sites, alternatively spliced mRNAs and new introns in meiotically regulated genes of yeast. Nucleic Acids Res. 28, 1700–1706 (2000)
Rodríguez-Navarro, S., Igual, J. C. & Pérez-Ortín, J. E. SRC1: an intron-containing yeast gene involved in sister chromatid segregation. Yeast 19, 43–54 (2002)
Grund, S. E. et al. The inner nuclear membrane protein Src1 associates with subtelomeric genes and alters their regulated gene expression. J. Cell Biol. 182, 897–910 (2008)
Kuhn, A. N. & Kaufer, N. F. Pre-mRNA splicing in Schizosaccharomyces pombe: regulatory role of a kinase conserved from fission yeast to mammals. Curr. Genet. 42, 241–251 (2003)
Okazaki, K. & Niwa, O. mRNAs encoding zinc finger protein isoforms are expressed by alternative splicing of an in-frame intron in fission yeast. DNA Res. 7, 27–30 (2000)
Kress, T. L., Krogan, N. J. & Guthrie, C. A single SR-like protein, Npl3, promotes pre-mRNA splicing in budding yeast. Mol. Cell 32, 727–734 (2008)
Knop, M. et al. Epitope tagging of yeast genes using a PCR-based strategy: more tags and improved practical routines. Yeast 15, 963–972 (1999)
Janke, C. et al. A versatile toolbox for PCR-based tagging of yeast genes: new fluorescent proteins, more markers and promoter substitution cassettes. Yeast 21, 947–962 (2004)
Hagan, I. M. & Ayscough, K. R. in Protein Localization by Fluorescence Microscopy: A Practical Approach (ed Allan, V. J.) 179–205 (Oxford Univ. Press, 2000)
Ansari, A. & Schwer, B. SLU7 and a novel activity, SSF1, act during the PRP16-dependent step of yeast pre-mRNA splicing. EMBO J. 14, 4001–4009 (1995)
Steen, H. & Mann, M. The abc’s (and xyz’s) of peptide sequencing. Nature Rev. Mol. Cell Biol. 5, 699–711 (2004)
Kabsch, W. Automatic processing of rotation diffraction data from crystals of initially unknown symmetry and cell constants. J. Appl. Cryst. 26, 795–800 (1993)
Collaborative Computational Project 4. The CCP4 suite: programs for protein crystallography. Acta Crystallogr. D 50, 760–763 (1994)
McRee, D. E. XtalView/Xfit—a versatile program for manipulating atomic coordinates and electron density. J. Struct. Biol. 125, 156–165 (1999)
Lamzin, V. S. Automated refinement of protein models. Acta Crystallogr. D 49, 129–147 (1993)
Liu, M. et al. Improved WATERGATE pulse sequences for solvent suppresion in NMR spectroscopy. J. Magn. Reson. 132, 125–129 (1998)
Mori, S., Abeygunawardana, C., Johnson, M. O. & van Zijl, P. C. Improved sensitivity of HSQC spectra of exchanging protons at short interscan delays using a new fast HSQC (FHSQC) detection scheme that avoids water saturation. J. Magn. Reson. B. 108, 94–98 (1995)
Zhu, G., Kong, X. M. & Sze, K. H. Gradient and sensitivity enhancement of 2D TROSY with water flip-back, 3D NOESY-TROSY and TOCSY-TROSY experiments. J. Biomol. NMR 13, 77–81 (1999)
Pervushin, K. V., Wider, G. & Wüthrich, K. Single transition-to-single transition polarization transfer (ST2-PT) in [15N,1H]-TROSY. J. Biomol. NMR 12, 345–348 (1998)
Acknowledgements
We thank U. Cramer and M. Kost for technical assistance, G. Dittmar, C. Guthrie, M. Konarska, K. Matuschewski, O. Nielsen, M. Rosbash and H. Yashiroda for materials, S. Uebel and C. Boulegue for mass spectrometric analysis and help, K. Hofmann for pointing out putative HIND elements in Prp38 proteins of plants, and M. Singh for initiating structural work. S.J. is supported by the Max Planck Society, Deutsche Forschungsgemeinschaft, Fonds der chemischen Industrie, Center for Integrated Protein Science Munich and RUBICON EU Network of Excellence; T.A.H. by the Max Planck Society; M.A. by NIH (GM040478).
Author information
Authors and Affiliations
Contributions
S.K.M. (S. cerevisiae, S. pombe), T.A. (mammalian) and S.J. designed, obtained and analysed the genetic, biochemical and functional data; G.M.P, M.K. and T.A.H. the structural data; R.J.N. and M.A. Jr the splicing array data. S.J. and S.K.M. wrote the paper, and all authors contributed to the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
This file contains Supplementary Figures 1-14 with legends, Supplementary Tables 1-3 and additional references. (PDF 7396 kb)
Rights and permissions
About this article
Cite this article
Mishra, S., Ammon, T., Popowicz, G. et al. Role of the ubiquitin-like protein Hub1 in splice-site usage and alternative splicing. Nature 474, 173–178 (2011). https://doi.org/10.1038/nature10143
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature10143
This article is cited by
-
Metolazone upregulates mitochondrial chaperones and extends lifespan in Caenorhabditis elegans
Biogerontology (2021)
-
Post-translational modification and protein sorting to small extracellular vesicles including exosomes by ubiquitin and UBLs
Cellular and Molecular Life Sciences (2019)
-
Crystal structure and mutation analysis revealed that DREP2 CIDE forms a filament-like structure with features differing from those of DREP4 CIDE
Scientific Reports (2018)
-
Structures of the human pre-catalytic spliceosome and its precursor spliceosome
Cell Research (2018)
-
Intron specificity in pre-mRNA splicing
Current Genetics (2018)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.