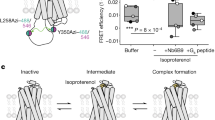
Abstract
G-protein-coupled receptors (GPCRs) are seven-transmembrane proteins that mediate most cellular responses to hormones and neurotransmitters. They are the largest group of therapeutic targets for a broad spectrum of diseases. Recent crystal structures of GPCRs1,2,3,4,5 have revealed structural conservation extending from the orthosteric ligand-binding site in the transmembrane core to the cytoplasmic G-protein-coupling domains. In contrast, the extracellular surface (ECS) of GPCRs is remarkably diverse and is therefore an ideal target for the discovery of subtype-selective drugs. However, little is known about the functional role of the ECS in receptor activation, or about conformational coupling of this surface to the native ligand-binding pocket. Here we use NMR spectroscopy to investigate ligand-specific conformational changes around a central structural feature in the ECS of the β2 adrenergic receptor: a salt bridge linking extracellular loops 2 and 3. Small-molecule drugs that bind within the transmembrane core and exhibit different efficacies towards G-protein activation (agonist, neutral antagonist and inverse agonist) also stabilize distinct conformations of the ECS. We thereby demonstrate conformational coupling between the ECS and the orthosteric binding site, showing that drugs targeting this diverse surface could function as allosteric modulators with high subtype selectivity. Moreover, these studies provide a new insight into the dynamic behaviour of GPCRs not addressable by static, inactive-state crystal structures.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Rosenbaum, D. M. et al. GPCR engineering yields high-resolution structural insights into β2-adrenergic receptor function. Science 318, 1266–1273 (2007)
Cherezov, V. et al. High-resolution crystal structure of an engineered human β2-adrenergic G protein-coupled receptor. Science 318, 1258–1265 (2007)
Rasmussen, S. G. et al. Crystal structure of the human β2 adrenergic G-protein-coupled receptor. Nature 450, 383–387 (2007)
Jaakola, V. P. et al. The 2.6 angstrom crystal structure of a human A2A adenosine receptor bound to an antagonist. Science 322, 1211–1217 (2008)
Warne, T. et al. Structure of a β1-adrenergic G-protein-coupled receptor. Nature 454, 486–491 (2008)
Kobilka, B. K. & Deupi, X. Conformational complexity of G-protein-coupled receptors. Trends Pharmacol. Sci. 28, 397–406 (2007)
Farrens, D. L., Altenbach, C., Yang, K., Hubbell, W. L. & Khorana, H. G. Requirement of rigid-body motion of transmembrane helices for light activation of rhodopsin. Science 274, 768–770 (1996)
Altenbach, C., Kusnetzow, A. K., Ernst, O. P., Hofmann, K. P. & Hubbell, W. L. High-resolution distance mapping in rhodopsin reveals the pattern of helix movement due to activation. Proc. Natl Acad. Sci. USA 105, 7439–7444 (2008)
Park, J. H., Scheerer, P., Hofmann, K. P., Choe, H. W. & Ernst, O. P. Crystal structure of the ligand-free G-protein-coupled receptor opsin. Nature 454, 183–187 (2008)
Scheerer, P. et al. Crystal structure of opsin in its G-protein-interacting conformation. Nature 455, 497–502 (2008)
Ghanouni, P. et al. Functionally different agonists induce distinct conformations in the G protein coupling domain of the β2 adrenergic receptor. J. Biol. Chem. 276, 24433–24436 (2001)
Swaminath, G. et al. Probing the β2 adrenoceptor binding site with catechol reveals differences in binding and activation by agonists and partial agonists. J. Biol. Chem. 280, 22165–22171 (2005)
Yao, X. et al. Coupling ligand structure to specific conformational switches in the β2-adrenoceptor. Nature Chem. Biol. 2, 417–422 (2006)
Ahuja, S. et al. Helix movement is coupled to displacement of the second extracellular loop in rhodopsin activation. Nature Struct. Mol. Biol. 16, 168–175 (2009)
Conn, P. J., Christopoulos, A. & Lindsley, C. W. Allosteric modulators of GPCRs: a novel approach for the treatment of CNS disorders. Nature Rev. Drug Discov. 8, 41–54 (2009)
Ballesteros, J. A. & Weinstein, H. Integrated methods for the construction of three-dimensional models and computational probing of structure-function relations in G-protein coupled receptors. Methods Neurosci. 25, 366–428 (1995)
Nygaard, R., Frimurer, T. M., Holst, B., Rosenkilde, M. M. & Schwartz, T. W. Ligand binding and micro-switches in 7TM receptor structures. Trends Pharmacol. Sci. 30, 249–259 (2009)
Zhang, M. & Vogel, H. J. Determination of the side chain pK a values of the lysine residues in calmodulin. J. Biol. Chem. 268, 22420–22428 (1993)
Tugarinov, V., Hwang, P. M., Ollerenshaw, J. E. & Kay, L. E. Cross-correlated relaxation enhanced 1H–13C NMR spectroscopy of methyl groups in very high molecular weight proteins and protein complexes. J. Am. Chem. Soc. 125, 10420–10428 (2003)
Jentoft, J. E., Jentoft, N., Gerken, T. A. & Dearborn, D. G. 13C NMR studies of ribonuclease A methylated with [13C]formaldehyde. J. Biol. Chem. 254, 4366–4370 (1979)
Gerken, T. A., Jentoft, J. E., Jentoft, N. & Dearborn, D. G. Intramolecular interactions of amino groups in 13C reductively methylated hen egg-white lysozyme. J. Biol. Chem. 257, 2894–2900 (1982)
Sherry, A. D. & Teherani, J. Physical studies of 13C-methylated concanavalin A. pH- and Co2+-induced nuclear magnetic resonance shifts. J. Biol. Chem. 258, 8663–8669 (1983)
Abraham, S. J., Hoheisel, S. & Gaponenko, V. Detection of protein-ligand interactions by NMR using reductive methylation of lysine residues. J. Biomol. NMR 42, 143–148 (2008)
Hanson, M. A. et al. A specific cholesterol binding site is established by the 2.8 Å structure of the human β2-adrenergic receptor. Structure 16, 897–905 (2008)
Kumar, S. & Nussinov, R. Relationship between ion pair geometries and electrostatic strengths in proteins. Biophys. J. 83, 1595–1612 (2002)
Baneres, J. L. et al. Molecular characterization of a purified 5-HT4 receptor: a structural basis for drug efficacy. J. Biol. Chem. 280, 20253–20260 (2005)
Wieland, K., Zuurmond, H. M., Krasel, C., Ijzerman, A. P. & Lohse, M. J. Involvement of Asn-293 in stereospecific agonist recognition and in activation of the β2-adrenergic receptor. Proc. Natl Acad. Sci. USA 93, 9276–9281 (1996)
Elling, C. E. et al. Metal ion site engineering indicates a global toggle switch model for seven-transmembrane receptor activation. J. Biol. Chem. 281, 17337–17346 (2006)
Schwartz, T. W., Frimurer, T. M., Holst, B., Rosenkilde, M. M. & Elling, C. E. Molecular mechanism of 7TM receptor activation—a global toggle switch model. Annu. Rev. Pharmacol. Toxicol. 46, 481–519 (2006)
Hubbard, S. J., Campbell, S. F. & Thornton, J. M. Molecular recognition. Conformational analysis of limited proteolytic sites and serine proteinase protein inhibitors. J. Mol. Biol. 220, 507–530 (1991)
Kobilka, B. K. Amino and carboxyl terminal modifications to facilitate the production and purification of a G protein-coupled receptor. Anal. Biochem. 231, 269–271 (1995)
Yao, X. J. et al. The effect of ligand efficacy on the formation and stability of a GPCR–G protein complex. Proc. Natl Acad. Sci. USA 106, 9501–9506 (2009)
Tota, M. R. & Strader, C. D. Characterization of the binding domain of the β-adrenergic receptor with the fluorescent antagonist carazolol. Evidence for a buried ligand binding site. J. Biol. Chem. 265, 16891–16897 (1990)
Baker, J. G. The selectivity of β-adrenoceptor antagonists at the human β1, β2 and β3 adrenoceptors. Br. J. Pharmacol. 144, 317–322 (2005)
Otwinowski, Z. & Minor, W. Processing of X-ray diffraction data collected in oscillation mode. Macromol. Crystallogr. A 276, 307–326 (1997)
Brunger, A. T. et al. Crystallography & NMR system: a new software suite for macromolecular structure determination. Acta Crystallogr. D 54, 905–921 (1998)
Adams, P. D. et al. PHENIX: building new software for automated crystallographic structure determination. Acta Crystallogr. D 58, 1948–1954 (2002)
Acknowledgements
We acknowledge support from National Institutes of Health, grants NS028471 (B.K.K.) and GM56169 (W.I.W.), the Stanford Medical Scientist Training Program (M.P.B.), the Lundbeck Foundation (S.G.F.R.), the University of Copenhagen and 7TM Pharma (R.N.), and the Instituto de Salud Carlos III (L.P.).
Author Contributions M.P.B. designed experiments, purified, labelled and functionally characterized β2AR, collected and analysed NMR data and wrote the paper. Y.Z. made, expressed and purified β2AR lysine mutants and collected NMR data. S.G.F.R. expressed and purified β2AR for NMR and crystallized the 13C-methylated β2AR–Fab complex. C.W.L. designed, optimized and supervised NMR experiments and collected NMR data. D.M.R., H.-J.C. and W.I.W. collected diffraction data and refined the structure of the 13C-methylated β2AR–Fab complex. R.N. collected and analysed NMR data and optimized data processing. J.J.F. performed G-protein-coupling assays on labelled β2AR. F.S.T. prepared insect cell cultures and purified β2AR. T.S.K. purified β2AR. J.D.P. advised on NMR spectroscopy experiments. L.P. performed molecular modelling and molecular dynamics simulations. R.S.P. designed and optimized NMR experiments, wrote NMR pulse sequences and collected data. L.M. conceived of lysine methylation of the β2AR, wrote NMR pulse sequences and designed NMR experiments. B.K.K. supervised the overall project, designed experiments, collected diffraction data and wrote the paper.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
This file contains Supplementary Figures 1-17 with Legends, Supplementary Tables 1-3 and Supplementary References. (PDF 14619 kb)
Rights and permissions
About this article
Cite this article
Bokoch, M., Zou, Y., Rasmussen, S. et al. Ligand-specific regulation of the extracellular surface of a G-protein-coupled receptor. Nature 463, 108–112 (2010). https://doi.org/10.1038/nature08650
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nature08650
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.