Abstract
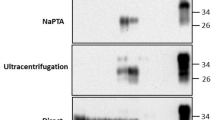
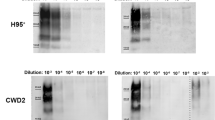
Infectious prion diseases1—scrapie of sheep2 and chronic wasting disease (CWD) of several species in the deer family3,4—are transmitted naturally within affected host populations. Although several possible sources of contagion have been identified in excretions and secretions from symptomatic animals5,6,7,8, the biological importance of these sources in sustaining epidemics remains unclear. Here we show that asymptomatic CWD-infected mule deer (Odocoileus hemionus) excrete CWD prions in their faeces long before they develop clinical signs of prion disease. Intracerebral inoculation of irradiated deer faeces into transgenic mice overexpressing cervid prion protein (PrP) revealed infectivity in 14 of 15 faecal samples collected from five deer at 7–11 months before the onset of neurological disease. Although prion concentrations in deer faeces were considerably lower than in brain tissue from the same deer collected at the end of the disease, the estimated total infectious dose excreted in faeces by an infected deer over the disease course may approximate the total contained in a brain. Prolonged faecal prion excretion by infected deer provides a plausible natural mechanism that might explain the high incidence and efficient horizontal transmission of CWD within deer herds3,4,9, as well as prion transmission among other susceptible cervids.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Prusiner, S. B. in Fields Virology (eds Knipe, D. M. et al.) 3059—3092 (Lippincott Williams & Wilkins, 2007)
Detwiler, L. A. & Baylis, M. The epidemiology of scrapie. Rev. Sci. Tech. 22, 121—143 (2003)
Williams, E. S. & Young, S. Chronic wasting disease of captive mule deer: a spongiform encephalopathy. J. Wildl. Dis. 16, 89—98 (1980)
Williams, E. S. Chronic wasting disease. Vet. Pathol. 42, 530—549 (2005)
Mathiason, C. K. et al. Infectious prions in the saliva and blood of deer with chronic wasting disease. Science 314, 133—136 (2006)
Angers, R. C. et al. Chronic wasting disease prions in elk antler velvet. Emerg. Infect. Dis. 15, 696—703 (2009)
Haley, N. J., Seelig, D. M., Zabel, M. D., Telling, G. C. & Hoover, E. A. Detection of CWD prions in urine and saliva of deer by transgenic mouse bioassay. PLoS ONE 4, e4848 (2009)
Konold, T., Moore, S. J., Bellworthy, S. J. & Simmons, H. A. Evidence of scrapie transmission via milk. BMC Vet. Res. 4, 14 (2008)
Miller, M. W. & Williams, E. S. Prion disease: horizontal prion transmission in mule deer. Nature 425, 35—36 (2003)
Baeten, L. A., Powers, B. E., Jewell, J. E., Spraker, T. R. & Miller, M. W. A natural case of chronic wasting disease in a free-ranging moose (Alces alces shirasi). J. Wildl. Dis. 43, 309—314 (2007)
Miller, M. W. et al. Lions and prions and deer demise. PLoS ONE 3, e4019 (2008)
Miller, M. W., Hobbs, N. T. & Tavener, S. J. Dynamics of prion disease transmission in mule deer. Ecol. Appl. 16, 2208—2214 (2006)
Sigurdson, C. J. et al. Oral transmission and early lymphoid tropism of chronic wasting disease PrPres in mule deer fawns (Odocoileus hemionus). J. Gen. Virol. 80, 2757—2764 (1999)
Fox, K. A., Jewell, J. E., Williams, E. S. & Miller, M. W. Patterns of PrPCWD accumulation during the course of chronic wasting disease infection in orally inoculated mule deer (Odocoileus hemionus). J. Gen. Virol. 87, 3451—3461 (2006)
Miller, M. W., Williams, E. S., Hobbs, N. T. & Wolfe, L. L. Environmental sources of prion transmission in mule deer. Emerg. Infect. Dis. 10, 1003—1006 (2004)
Angers, R. C. et al. Prions in skeletal muscles of deer with chronic wasting disease. Science 311, 1117 (2006)
Tamgüney, G. et al. Transmission of elk and deer prions to transgenic mice. J. Virol. 80, 9104—9114 (2006)
Dougherty, R. in Techniques in Experimental Virology (ed. Harris, R. J. C.) 169—224 (Academic, 1964)
Cox, D. R. Regression models and life-tables. J. R. Stat. Soc. A 34, 187—220 (1972)
Prusiner, S. B. et al. Measurement of the scrapie agent using an incubation time interval assay. Ann. Neurol. 11, 353—358 (1982)
Wolfe, L. L. et al. PrPCWD in rectal lymphoid tissue of deer (Odocoileus spp.). J. Gen. Virol. 88, 2078—2082 (2007)
Tamgüney, G. et al. Transmission of scrapie and sheep-passaged bovine spongiform encephalopathy prions to transgenic mice expressing elk prion protein. J. Gen. Virol. 90, 1035—1047 (2009)
Miekka, S. I. et al. Inactivation of viral and prion pathogens by gamma-irradiation under conditions that maintain the integrity of human albumin. Vox Sang. 84, 36—44 (2003)
Judge, J., Greig, A., Kyriazakis, I. & Hutchings, M. R. Ingestion of faeces by grazing herbivores—risk of inter-species disease transmission. Agric. Ecosyst. Environ. 107, 267—274 (2005)
Arthur, W. J. & Alldredge, A. W. Seasonal estimates of masses of mule deer fecal pellets and pellet groups. J. Wildl. Mgmt 44, 750—752 (1980)
Diringer, H., Roehmel, J. & Beekes, M. Effect of repeated oral infection of hamsters with scrapie. J. Gen. Virol. 79, 609—612 (1998)
Johnson, C. J., Pedersen, J. A., Chappell, R. J., McKenzie, D. & Aiken, J. M. Oral transmissibility of prion disease is enhanced by binding to soil particles. PLoS Pathog. 3, e93 (2007)
Arthur, W. J. & Alldredge, A. W. Soil ingestion by mule deer in northcentral Colorado. J. Range Mgmt 32, 67—71 (1979)
Hadlow, W. J., Kennedy, R. C. & Race, R. E. Natural infection of Suffolk sheep with scrapie virus. J. Infect. Dis. 146, 657—664 (1982)
Raymond, G. J. et al. Evidence of a molecular barrier limiting susceptibility of humans, cattle and sheep to chronic wasting disease. EMBO J. 19, 4425—4430 (2000)
Williams, E. S. et al. Epidemiology of Chronic Wasting Disease: Detection of PrPres, Shedding, and Environmental Contamination (United States Department of Defense National Prion Research Program, 2003)
Bellinger-Kawahara, C. G., Kempner, E., Groth, D. F., Gabizon, R. & Prusiner, S. B. Scrapie prion liposomes and rods exhibit target sizes of 55,000 Da. Virology 164, 537—541 (1988)
Büeler, H. et al. Normal development and behaviour of mice lacking the neuronal cell-surface PrP protein. Nature 356, 577—582 (1992)
Tamgüney, G. et al. Genes contributing to prion pathogenesis. J. Gen. Virol. 89, 1777—1788 (2008)
Carlson, G. A. et al. Genetics and polymorphism of the mouse prion gene complex: control of scrapie incubation time. Mol. Cell. Biol. 8, 5528—5540 (1988)
Scott, M. et al. Propagation of prions with artificial properties in transgenic mice expressing chimeric PrP genes. Cell 73, 979—988 (1993)
Cox, D. R. & Oakes, D. Analysis of Survival Data (Chapman & Hall and CRC, 1984)
Vittinghoff, E., Glidden, D. V., Shiboski, S. C. & McCulloch, C. E. Regression Methods in Biostatistics: Linear, Logistic, Survival, and Repeated Measures Models (Springer Science+Business Media, 2005)
Laemmli, U. K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227, 680—685 (1970)
Safar, J. G. et al. Measuring prions causing bovine spongiform encephalopathy or chronic wasting disease by immunoassays and transgenic mice. Nature Biotechnol. 20, 1147—1150 (2002)
Muramoto, T. et al. Heritable disorder resembling neuronal storage disease in mice expressing prion protein with deletion of an α-helix. Nature Med. 3, 750—755 (1997)
Acknowledgements
G.T. was supported by a fellowship from the Larry L. Hillblom Foundation. This work was supported by the Colorado Division of Wildlife and grants from the US Department of Defense National Prion Research Program (NP020152), the National Institutes of Health (AG02132) and the Hillblom Foundation. We thank P. Lessard and the staff of the Hunters Point animal facility for support with the transgenic animal experiments, K. Giles for screening transgenic animals, A. Serban for antibodies, K. Pomeroy for technical assistance, J. Safar for discussions, H. Nguyen for editorial assistance and B. Williams for insights on CWD transmission.
Author Contributions G.T., M.W.M. and S.B.P. designed the transgenic mouse studies; G.T., M.W.M., L.L.W., T.M.S., C.P., A.L. and S.J.D. performed various aspects of the research on mule deer or transgenic mice; G.T., M.W.M, D.V.G., S.J.D. and S.B.P. analysed the data; G.T., M.W.M., S.J.D. and S.B.P. wrote the paper. All authors discussed the results and commented on the manuscript.
Author information
Authors and Affiliations
Corresponding author
Supplementary information
Supplementary Information
This file contains Supplementary Figures 1-3 with Legends, Supplementary Tables 1-3 and Supplementary Notes. (PDF 781 kb)
Rights and permissions
About this article
Cite this article
Tamgüney, G., Miller, M., Wolfe, L. et al. Asymptomatic deer excrete infectious prions in faeces. Nature 461, 529–532 (2009). https://doi.org/10.1038/nature08289
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature08289
This article is cited by
-
Patients with isolated REM-sleep behavior disorder have elevated levels of alpha-synuclein aggregates in stool
npj Parkinson's Disease (2023)
-
Dynamics of CWD prion detection in feces and blood from naturally infected white-tailed deer
Scientific Reports (2023)
-
Propagation of PrPSc in mice reveals impact of aggregate composition on prion disease pathogenesis
Communications Biology (2023)
-
Vaccines for prion diseases: a realistic goal?
Cell and Tissue Research (2023)
-
Effect of legal regulation of supplemental feeding on space use of red deer in an area with chronic wasting disease
European Journal of Wildlife Research (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.