Abstract
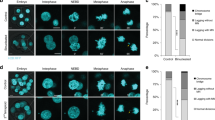
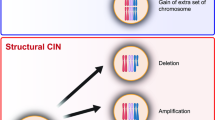
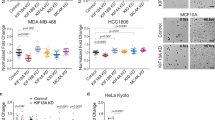
Chromosomal instability (CIN) is a hallmark of many tumours and correlates with the presence of extra centrosomes1,2,3,4. However, a direct mechanistic link between extra centrosomes and CIN has not been established. It has been proposed that extra centrosomes generate CIN by promoting multipolar anaphase, a highly abnormal division that produces three or more aneuploid daughter cells. Here we use long-term live-cell imaging to demonstrate that cells with multiple centrosomes rarely undergo multipolar cell divisions, and the progeny of these divisions are typically inviable. Thus, multipolar divisions cannot explain observed rates of CIN. In contrast, we observe that CIN cells with extra centrosomes routinely undergo bipolar cell divisions, but display a significantly increased frequency of lagging chromosomes during anaphase. To define the mechanism underlying this mitotic defect, we generated cells that differ only in their centrosome number. We demonstrate that extra centrosomes alone are sufficient to promote chromosome missegregation during bipolar cell division. These segregation errors are a consequence of cells passing through a transient ‘multipolar spindle intermediate’ in which merotelic kinetochore–microtubule attachment errors accumulate before centrosome clustering and anaphase. These findings provide a direct mechanistic link between extra centrosomes and CIN, two common characteristics of solid tumours. We propose that this mechanism may be a common underlying cause of CIN in human cancer.
This is a preview of subscription content, access via your institution
Access options




Similar content being viewed by others
References
Lengauer, C., Kinzler, K. W. & Vogelstein, B. Genetic instability in colorectal cancers. Nature 386, 623–627 (1997)
D’Assoro, A. B., Lingle, W. L. & Salisbury, J. L. Centrosome amplification and the development of cancer. Oncogene 21, 6146–6153 (2002)
Nigg, E. A. Centrosome aberrations: cause or consequence of cancer progression? Nature Rev. Cancer 2, 815–825 (2002)
Sluder, G. & Nordberg, J. J. The good, the bad and the ugly: the practical consequences of centrosome amplification. Curr. Opin. Cell Biol. 16, 49–54 (2004)
Rajagopalan, H. & Lengauer, C. Aneuploidy and cancer. Nature 432, 338–341 (2004)
Weaver, B. A., Silk, A. D., Montagna, C., Verdier-Pinard, P. & Cleveland, D. W. Aneuploidy acts both oncogenically and as a tumor suppressor. Cancer Cell 11, 25–36 (2007)
Gao, C. et al. Chromosome instability, chromosome transcriptome, and clonal evolution of tumor cell populations. Proc. Natl Acad. Sci. USA 104, 8995–9000 (2007)
Nowell, P. C. The clonal evolution of tumor cell populations. Science 194, 23–28 (1976)
Ghadimi, B. M. et al. Centrosome amplification and instability occurs exclusively in aneuploid, but not in diploid colorectal cancer cell lines, and correlates with numerical chromosomal aberrations. Genes Chromosom. Cancer 27, 183–190 (2000)
Lingle, W. L. et al. Centrosome amplification drives chromosomal instability in breast tumor development. Proc. Natl Acad. Sci. USA 99, 1978–1983 (2002)
Pihan, G. A. et al. Centrosome defects can account for cellular and genetic changes that characterize prostate cancer progression. Cancer Res. 61, 2212–2219 (2001)
Sato, N. et al. Correlation between centrosome abnormalities and chromosomal instability in human pancreatic cancer cells. Cancer Genet. Cytogenet. 126, 13–19 (2001)
Thompson, S. L. & Compton, D. A. Examining the link between chromosomal instability and aneuploidy in human cells. J. Cell Biol. 180, 665–672 (2008)
Torres, E. M., Williams, B. R. & Amon, A. Aneuploidy: cells losing their balance. Genetics 179, 737–746 (2008)
Brinkley, B. R. Managing the centrosome numbers game: from chaos to stability in cancer cell division. Trends Cell Biol. 11, 18–21 (2001)
Basto, R. et al. Centrosome amplification can initiate tumorigenesis in flies. Cell 133, 1032–1042 (2008)
Kwon, M. et al. Mechanisms to suppress multipolar divisions in cancer cells with extra centrosomes. Genes Dev. 22, 2189–2203 (2008)
Quintyne, N. J., Reing, J. E., Hoffelder, D. R., Gollin, S. M. & Saunders, W. S. Spindle multipolarity is prevented by centrosomal clustering. Science 307, 127–129 (2005)
Yang, Z., Loncarek, J., Khodjakov, A. & Rieder, C. L. Extra centrosomes and/or chromosomes prolong mitosis in human cells. Nature Cell Biol. 10, 748–751 (2008)
Cimini, D. Merotelic kinetochore orientation, aneuploidy, and cancer. Biochim. Biophys. Acta 1786, 32–40 (2008)
Salmon, E. D., Cimini, D., Cameron, L. A. & DeLuca, J. G. Merotelic kinetochores in mammalian tissue cells. Phil. Trans. R. Soc. Lond. B 360, 553–568 (2005)
Cimini, D. et al. Merotelic kinetochore orientation is a major mechanism of aneuploidy in mitotic mammalian tissue cells. J. Cell Biol. 153, 517–527 (2001)
Cimini, D., Moree, B., Canman, J. C. & Salmon, E. D. Merotelic kinetochore orientation occurs frequently during early mitosis in mammalian tissue cells and error correction is achieved by two different mechanisms. J. Cell Sci. 116, 4213–4225 (2003)
Heneen, W. K. Kinetochores and microtubules in multipolar mitosis and chromosome orientation. Exp. Cell Res. 91, 57–62 (1975)
Sluder, G., Thompson, E. A., Miller, F. J., Hayes, J. & Rieder, C. L. The checkpoint control for anaphase onset does not monitor excess numbers of spindle poles or bipolar spindle symmetry. J. Cell Sci. 110, 421–429 (1997)
Gascoigne, K. E. & Taylor, S. S. Cancer cells display profound intra- and interline variation following prolonged exposure to antimitotic drugs. Cancer Cell 14, 111–122 (2008)
Kleylein-Sohn, J. et al. Plk4-induced centriole biogenesis in human cells. Dev. Cell 13, 190–202 (2007)
Tsou, M. F. & Stearns, T. Mechanism limiting centrosome duplication to once per cell cycle. Nature 442, 947–951 (2006)
Bakhoum, S. F., Thompson, S. L., Manning, A. L. & Compton, D. A. Genome stability is ensured by temporal control of kinetochore-microtubule dynamics. Nature Cell Biol. 11, 27–35 (2008)
Chandhok, N. S. & Pellman, D. A little CIN may cost a lot: revisiting aneuploidy and cancer. Curr. Opin. Genet. Dev. 19, 74–81 (2009)
Acknowledgements
We would like to thank D. Compton, R. King, D. Livingston, A. Manning, T. Rapoport, J. Shah, T. Stukenberg and members of the Pellman laboratory for comments and discussion of the manuscript; L. Cameron for setting up the confocal microscope; P. Schow for help with FACS; D. Neuberg for help with statistical analysis; C. King for help with the model figure; J. Iwasa for creating the animations; and L. Moreau and S. Thompson for technical advice on chromosome spreads and FISH analysis. Reagents were provided by D. Compton, S. Gollin, R. King, A. Khodjakov, E. Nigg and G.Wahl. N.J.G. is a fellow of the Leukemia and Lymphoma Society and D.P. is an HHMI Investigator. This work was supported by NIH grant GM083299.
Author Contributions N.J.G. and D.P. designed the experiments and wrote the manuscript. N.J.G. performed most of the experiments and data analysis. S.A.G. conceived and conducted experiments and performed data analysis, particularly for Fig. 4. D.P. supervised all aspects of the project. All authors discussed results and edited the manuscript.
Author information
Authors and Affiliations
Corresponding author
Supplementary information
Supplementary Figures
This file contains Supplementary Figures 1-8 with Legends. (PDF 748 kb)
Supplementary Movie 1
This movie shows a representative SCC114 cell expressing H2B-GFP that undergoes a multipolar cell division to produce three aneuploid daughter cells. All three daughter cells die in the subsequent cell cycle. Images captured every 10 minutes. (MOV 463 kb)
Supplementary Movie 2
This movie shows a tetraploid RPE-1 cell stably expressing GFP-centrin and mRFP-H2B that undergoes a multipolar cell division. Numerous lagging chromosomes can be observed at anaphase. Images captured every 2 minutes. (MOV 3694 kb)
Supplementary Movie 3
This movie shows tetraploid RPE-1 cell stably expressing GFP-centrin and mRFP-H2B during mitosis. While the majority of chromosomes are aligned at the metaphase plate, a single polar chromosome can be observed bi-orienting between two semi-clustered centrosomes. Anaphase ensues before this chromosome aligns, resulting in a single chromosomes missegregation event. Images captured every 2 minutes. (MOV 3462 kb)
Supplementary Movie 4
This file contains a cartoon animation demonstrating that both syntelic and merotelic attachments errors accumulate when extra-centrosomal cells pass through a transient multipolar intermediate prior to centrosome clustering and anaphase. (MOV 5028 kb)
Rights and permissions
About this article
Cite this article
Ganem, N., Godinho, S. & Pellman, D. A mechanism linking extra centrosomes to chromosomal instability. Nature 460, 278–282 (2009). https://doi.org/10.1038/nature08136
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature08136
This article is cited by
-
scAbsolute: measuring single-cell ploidy and replication status
Genome Biology (2024)
-
Prolonged overexpression of PLK4 leads to formation of centriole rosette clusters that are connected via canonical centrosome linker proteins
Scientific Reports (2024)
-
The reckoning of chromosomal instability: past, present, future
Chromosome Research (2024)
-
Mechanisms of ovarian aging in women: a review
Journal of Ovarian Research (2023)
-
A DNA-PK phosphorylation site on MET regulates its signaling interface with the DNA damage response
Oncogene (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.