Abstract
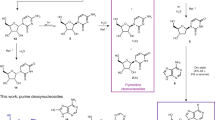
At some stage in the origin of life, an informational polymer must have arisen by purely chemical means. According to one version of the ‘RNA world’ hypothesis1,2,3 this polymer was RNA, but attempts to provide experimental support for this have failed4,5. In particular, although there has been some success demonstrating that ‘activated’ ribonucleotides can polymerize to form RNA6,7, it is far from obvious how such ribonucleotides could have formed from their constituent parts (ribose and nucleobases). Ribose is difficult to form selectively8,9, and the addition of nucleobases to ribose is inefficient in the case of purines10 and does not occur at all in the case of the canonical pyrimidines11. Here we show that activated pyrimidine ribonucleotides can be formed in a short sequence that bypasses free ribose and the nucleobases, and instead proceeds through arabinose amino-oxazoline and anhydronucleoside intermediates. The starting materials for the synthesis—cyanamide, cyanoacetylene, glycolaldehyde, glyceraldehyde and inorganic phosphate—are plausible prebiotic feedstock molecules12,13,14,15, and the conditions of the synthesis are consistent with potential early-Earth geochemical models. Although inorganic phosphate is only incorporated into the nucleotides at a late stage of the sequence, its presence from the start is essential as it controls three reactions in the earlier stages by acting as a general acid/base catalyst, a nucleophilic catalyst, a pH buffer and a chemical buffer. For prebiotic reaction sequences, our results highlight the importance of working with mixed chemical systems in which reactants for a particular reaction step can also control other steps.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Woese, C. The Genetic Code 179–195 (Harper & Row, 1967)
Crick, F. H. C. The origin of the genetic code. J. Mol. Biol. 38, 367–379 (1968)
Orgel, L. E. Evolution of the genetic apparatus. J. Mol. Biol. 38, 381–393 (1968)
Joyce, G. F. The antiquity of RNA-based evolution. Nature 418, 214–221 (2002)
Joyce, G. F. & Orgel, L. E. in The RNA World (eds Gesteland, R. F., Cech, T. R. & Atkins, J. F.) 23–56 (Cold Spring Harbor Laboratory Press, 2006)
Ferris, J. P., Hill, A. R., Liu, R. & Orgel, L. E. Synthesis of long prebiotic oligomers on mineral surfaces. Nature 381, 59–61 (1996)
Verlander, M. S., Lohrmann, R. & Orgel, L. E. Catalysts for the self-polymerization of adenosine cyclic 2′,3′-phosphate. J. Mol. Evol. 2, 303–316 (1973)
Kofoed, J., Reymond, J.-L. & Darbre, T. Prebiotic carbohydrate synthesis: zinc-proline catalyzes direct aqueous aldol reactions of α-hydroxy aldehydes and ketones. Org. Biomol. Chem. 3, 1850–1855 (2005)
Ricardo, A., Carrigan, M. A., Olcott, A. N. & Benner, S. A. Borate minerals stabilize ribose. Science 303, 196 (2004)
Fuller, W. D., Sanchez, R. A. & Orgel, L. E. Studies in prebiotic synthesis VI. Synthesis of purine nucleosides. J. Mol. Biol. 67, 25–33 (1972)
Orgel, L. E. Prebiotic chemistry and the origin of the RNA world. Crit. Rev. Biochem. Mol. Biol. 39, 99–123 (2004)
Thaddeus, P. The prebiotic molecules observed in the interstellar gas. Phil. Trans. R. Soc. B 361, 1681–1687 (2006)
Sanchez, R. A., Ferris, J. P. & Orgel, L. E. Cyanoacetylene in prebiotic synthesis. Science 154, 784–785 (1966)
Pasek, M. A. & Lauretta, D. S. Aqueous corrosion of phosphide minerals from iron meteorites: a highly reactive source of prebiotic phosphorus on the surface of the early Earth. Astrobiology 5, 515–535 (2005)
Bryant, D. E. & Kee, T. P. Direct evidence for the availability of reactive, water soluble phosphorus on the early Earth. H-phosphinic acid from the Nantan meteorite. Chem. Commun. 2344–2346 (2006)
Robertson, M. P. & Miller, S. L. An efficient prebiotic synthesis of cytosine and uracil. Nature 373, 772–774 (1995)
Ferris, J. P., Goldstein, G. & Beaulieu, D. J. Chemical evolution, IV. An evaluation of cyanovinyl phosphate as a prebiotic phosphorylating agent. J. Am. Chem. Soc. 92, 6598–6603 (1970)
Lohrmann, R. & Orgel, L. E. Prebiotic synthesis: phosphorylation in aqueous solution. Science 161, 64–66 (1968)
Anastasi, C. et al. RNA: prebiotic product, or biotic invention? Chem. Biodivers. 4, 721–739 (2007)
Anastasi, C., Crowe, M. A., Powner, M. W. & Sutherland, J. D. Direct assembly of nucleoside precursors from two- and three-carbon units. Angew. Chem. Int. Edn Engl. 45, 6176–6179 (2006)
Cockerill, A. F. et al. An improved synthesis of 2-amino-1,3-oxazoles under basic catalysis. Synthesis 591–593 (1976)
Springsteen, G. & Joyce, G. F. Selective derivatization and sequestration of ribose from a prebiotic mix. J. Am. Chem. Soc. 126, 9578–9583 (2004)
Sanchez, R. A. & Orgel, L. E. Studies in prebiotic synthesis V. Synthesis and photoanomerization of pyrimidine nucleosides. J. Mol. Biol. 47, 531–543 (1970)
Tapiero, C. M. & Nagyvary, J. Prebiotic formation of cytidine nucleotides. Nature 231, 42–43 (1971)
Shannahoff, D. H. & Sanchez, R. A. 2,2′-Anhydronucleosides. Novel syntheses and reactions. J. Org. Chem. 38, 593–598 (1973)
Lohrmann, R. &. Orgel, L. E. Urea-inorganic phosphate mixtures as prebiotic phosphorylating agents. Science 171, 490–494 (1971)
Schoffstall, A. M. Prebiotic phosphorylation of nucleosides in formamide. Orig. Life 7, 399–412 (1976)
Miller, N. & Cerutti, P. Structure of the photohydration products of cytidine and uridine. Proc. Natl Acad. Sci. USA 59, 34–38 (1968)
Powner, M. W. et al. On the prebiotic synthesis of ribonucleotides: photoanomerisation of cytosine nucleosides and nucleotides revisited. ChemBioChem 8, 1170–1179 (2007)
Eschenmoser, A. & Loewenthal, E. Chemistry of potentially prebiological natural products. Chem. Soc. Rev. 21, 1–16 (1992)
Acknowledgements
This work has been funded by the UK Engineering and Physical Sciences Research Council through the provision of a postdoctoral fellowship (B.G.) and a PhD studentship (M.W.P.). We thank J. Raftery and M. Helliwell for X-ray crystallography.
Author information
Authors and Affiliations
Corresponding author
Additional information
X-ray crystallographic data (excluding structure factors) for 13 have been deposited at the Cambridge Crystallographic Data Centre, UK, under deposition number CCDC 701055. These data can be obtained free of charge from the Cambridge Crystallographic Data Centre (http://www.ccdc.cam.ac.uk/data_request/cif).
Supplementary information
Supplementary Information
This file contains Supplementary Methods and Data with Supplementary Figures S1-S13 and Supplementary References. (PDF 2705 kb)
Rights and permissions
About this article
Cite this article
Powner, M., Gerland, B. & Sutherland, J. Synthesis of activated pyrimidine ribonucleotides in prebiotically plausible conditions. Nature 459, 239–242 (2009). https://doi.org/10.1038/nature08013
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nature08013
This article is cited by
-
Biogeochemical explanations for the world’s most phosphate-rich lake, an origin-of-life analog
Communications Earth & Environment (2024)
-
Symmetry breaking and chiral amplification in prebiotic ligation reactions
Nature (2024)
-
Cosmic dust fertilization of glacial prebiotic chemistry on early Earth
Nature Astronomy (2024)
-
Physical non-equilibria for prebiotic nucleic acid chemistry
Nature Reviews Physics (2023)
-
Regioselective ribonucleoside synthesis through Ti-catalysed ribosylation of nucleobases
Nature Synthesis (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.