Abstract
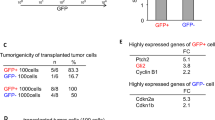
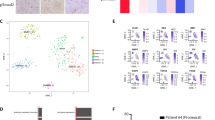
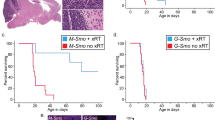
Transformed, oncogenic precursors, possessing both defining neural-stem-cell properties and the ability to initiate intracerebral tumours, have been identified in human brain cancers1. Here we report that bone morphogenetic proteins (BMPs), amongst which BMP4 elicits the strongest effect, trigger a significant reduction in the stem-like, tumour-initiating precursors of human glioblastomas (GBMs). Transient in vitro exposure to BMP4 abolishes the capacity of transplanted GBM cells to establish intracerebral GBMs. Most importantly, in vivo delivery of BMP4 effectively blocks the tumour growth and associated mortality that occur in 100% of mice after intracerebral grafting of human GBM cells. We demonstrate that BMPs activate their cognate receptors (BMPRs) and trigger the Smad signalling cascade in cells isolated from human glioblastomas (GBMs). This is followed by a reduction in proliferation, and increased expression of markers of neural differentiation, with no effect on cell viability. The concomitant reduction in clonogenic ability, in the size of the CD133+ population and in the growth kinetics of GBM cells indicates that BMP4 reduces the tumour-initiating cell pool of GBMs. These findings show that the BMP–BMPR signalling system—which controls the activity of normal brain stem cells2,3—may also act as a key inhibitory regulator of tumour-initiating, stem-like cells from GBMs and the results also identify BMP4 as a novel, non-cytotoxic therapeutic effector, which may be used to prevent growth and recurrence of GBMs in humans.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Vescovi, A. L., Galli, R. & Reynolds, B. A. Brain tumour stem cells. Nature Rev. Cancer 6, 425–436 (2006)
Lim, D. A. et al. Noggin antagonizes BMP signaling to create a niche for adult neurogenesis. Neuron 28, 713–726 (2000)
Panchision, D. M. & McKay, R. D. The control of neural stem cells by morphogenic signals. Curr. Opin. Genet. Dev. 12, 478–487 (2002)
Lapidot, T. et al. A cell initiating human acute myeloid leukaemia after transplantation into SCID mice. Nature 367, 645–648 (1994)
Al-Hajj, M., Wicha, M. S., Benito-Hernandez, A., Morrison, S. J. & Clarke, M. F. Prospective identification of tumorigenic breast cancer cells. Proc. Natl Acad. Sci. USA 100, 3983–3988 (2003)
Ignatova, T. N. et al. Human cortical glial tumors contain neural stem-like cells expressing astroglial and neuronal markers in vitro.. Glia 39, 193–206 (2002)
Hemmati, H. D. et al. Cancerous stem cells can arise from pediatric brain tumors. Proc. Natl Acad. Sci. USA 100, 15178–15183 (2003)
Galli, R. et al. Isolation and characterization of tumorigenic, stem-like neural precursors from human glioblastoma. Cancer Res. 64, 7011–7021 (2004)
Singh, S. K. et al. Identification of human brain tumour initiating cells. Nature 432, 396–401 (2004)
Taylor, M. D. et al. Radial glia cells are candidate stem cells of ependymoma. Cancer Cell 8, 323–335 (2005)
Sell, S. Stem cell origin of cancer and differentiation therapy. Crit. Rev. Oncol. Hematol. 51, 1–28 (2004)
Reya, T., Morrison, S. J., Clarke, M. F. & Weissman, I. L. Stem cells, cancer, and cancer stem cells. Nature 414, 105–111 (2001)
Potten, C. S. & Loeffler, M. Stem cells: attributes, cycles, spirals, pitfalls and uncertainties. Lessons for and from the crypt. Development 110, 1001–1020 (1990)
Weiss, S. et al. Multipotent CNS stem cells are present in the adult mammalian spinal cord and ventricular neuroaxis. J. Neurosci. 16, 7599–7609 (1996)
Shah, N. M., Groves, A. K. & Anderson, D. J. Alternative neural crest cell fates are instructively promoted by TGFβ superfamily members. Cell 85, 331–343 (1996)
Furuta, Y., Piston, D. W. & Hogan, B. L. Bone morphogenetic proteins (BMPs) as regulators of dorsal forebrain development. Development 124, 2203–2212 (1997)
Li, W., Cogswell, C. A. & LoTurco, J. J. Neuronal differentiation of precursors in the neocortical ventricular zone is triggered by BMP. J. Neurosci. 18, 8853–8862 (1998)
Iantosca, M., McPherson, C., Ho, S. & Maxwell, G. Bone morphogenetic proteins-2 and -4 attenuate apoptosis in a cerebellar primitive neuroectodermal tumor cell line. J. Neurosci. Res. 56, 248–258 (1999)
Chen, D., Zhao, M. & Mundy, G. R. Bone morphogenetic proteins. Growth Factors 22, 233–241 (2004)
Hallahan, A. R. et al. BMP-2 mediates retinoid-induced apoptosis in medulloblastoma cells through a paracrine effect. Nature Med. 9, 1033–1038 (2003)
Kjellman, C. et al. Expression of TGF-β isoforms, TGF-β receptors, and SMAD molecules at different stages of human glioma. Int. J. Cancer 89, 251–258 (2000)
Canalis, E., Economides, A. & Gazzerro, E. Bone morphogenetic proteins, their antagonists, and the skeleton. Endocr. Rev. 2, 218–235 (2003)
Golestaneh, N. & Mishra, B. TGF-β, neuronal stem cells and glioblastoma. Oncogene 24, 5722–5730 (2005)
Reynolds, B. A. & Weiss, S. Clonal and population analyses demonstrate that an EGF-responsive mammalian embryonic CNS precursor is a stem cell. Dev. Biol. 175, 1–13 (1996)
Reynolds, B. A. & Rietze, R. L. Neural stem cells and neurospheres–re-evaluating the relationship. Nature Methods 2, 333–336 (2005)
Barker, F. G. et al. Survival and functional status after resection of recurrent glioblastoma multiforme. Neurosurgery 42, 709–720; discussion 720–3. (1998)
Dean, M., Fojo, T. & Bates, S. Tumour stem cells and drug resistance. Nature Rev. Cancer 5, 275–284 (2005)
Pardal, R., Clarke, M. F. & Morrison, S. J. Applying the principles of stem-cell biology to cancer. Nature Rev. Cancer 3, 895–902 (2003)
Rajan, P., Panchision, D. M., Newell, L. F. & McKay, R. D. BMPs signal alternately through a SMAD or FRAP–STAT pathway to regulate fate choice in CNS stem cells. J. Cell Biol. 161, 911–921 (2003)
Vescovi, A. L. et al. Isolation and cloning of multipotential stem cells from the embryonic human CNS and establishment of transplantable human neural stem cell lines by epigenetic stimulation. Exp. Neurol. 156, 71–83 (1999)
Acknowledgements
This work was supported by the Associazione Italiana per la Ricerca sul Cancro, the Plurigenes EEC grant, funding from the Brain Tumours Funders’ Collaborative, from Neurothon Onlus, and by a donation from BMW Italia. We are grateful to R. Galli, J. Laterra, C. L. Solero and F. McBlane for helpful comments and suggestions.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
Reprints and permissions information is available at www.nature.com/reprints. The authors declare no competing financial interests.
Supplementary information
Supplementary Methods
Detailed description of methods used in this study. (DOC 42 kb)
Supplementary Tables
Supplementary Tables 1–9 containing comprehensive data on all cell preparations used. (DOC 183 kb)
Supplementary Legends
This file contains legends for Supplementary Tables and Supplementary Figures. (DOC 63 kb)
Supplementary Figure 1
A quantitative analysis (Real Time PCR (RT-PCR)) of the expression of the BMPR type 1A, 1B, and 2 genes in the different GBM cell preparations investigated in this study – acutely dissociated or briefly cultured (GBSC) cells. (JPG 749 kb)
Supplementary Figure 2
a, Dose response curves showing the ability of BMP4 to inhibit the proliferation of two representative GBM cell preparations in a dose-dependent fashion. b. Cytofluorimetric analysis showing the phosphorylation and nuclear translocation of phospho Smad 1,5,8 as elicited 1.5 hours post BMP4 treatment in cultured GBM cells. (JPG 475 kb)
Supplementary Figure 3
Effects of various BMPs (all at 100 ng/ml) on the growth of GBM cells. (JPG 441 kb)
Supplementary Figure 4
The figure shows the effect of BMP4 on the expansion rate of briefly cultured GBM cells expanding in the presence of mitogens. (JPG 996 kb)
Supplementary Figure 5
The figure shows one example of the dot plot graph concerning the quantitative analysis of the expression of the CD133 antigen. (JPG 632 kb)
Supplementary Figure 6
Untreated GBM cells constitutively express neuronal markers such as MAP2. (JPG 577 kb)
Supplementary Figure 7
BMP4 inhibits the tumorigenicity of GBM cells. (JPG 1564 kb)
Supplementary Figure 8
Ex vivo pre-treatment of GBM cells with BMP4 inhibits ventricular invasion following orthotopic injection. (JPG 486 kb)
Supplementary Figure 9
Phenotype analysis confirmed that, as shown previously, tumours induced by transplanting briefly cultured GBM cells into the brain of scid/bg animals generated astroglial-like cells. (JPG 1573 kb)
Supplementary Figure 10
a. Quantitative RT–PCR analysis of the expression of 13 different BMPs genes in briefly cultured GBM cells. b. When GBM cells, cultured with mitogens, were exposed to a neutralizing antibody that specifically blocks BMP4, the cell growth rate increased significantly as compared to control conditions. (JPG 1073 kb)
Rights and permissions
About this article
Cite this article
Piccirillo, S., Reynolds, B., Zanetti, N. et al. Bone morphogenetic proteins inhibit the tumorigenic potential of human brain tumour-initiating cells. Nature 444, 761–765 (2006). https://doi.org/10.1038/nature05349
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nature05349
This article is cited by
-
Identification of genes with oscillatory expression in glioblastoma: the paradigm of SOX2
Scientific Reports (2024)
-
BMP signaling in cancer stemness and differentiation
Cell Regeneration (2023)
-
Local delivery of hrBMP4 as an anticancer therapy in patients with recurrent glioblastoma: a first-in-human phase 1 dose escalation trial
Molecular Cancer (2023)
-
Systems Medicine for Precise Targeting of Glioblastoma
Molecular Biotechnology (2023)
-
Growth factor independence underpins a paroxysmal, aggressive Wnt5aHigh/EphA2Low phenotype in glioblastoma stem cells, conducive to experimental combinatorial therapy
Journal of Experimental & Clinical Cancer Research (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.