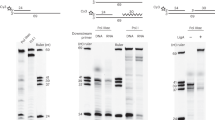
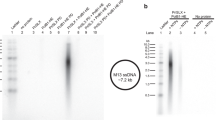
Abstract
RNA primers for DNA replication are usually synthesized by specialized enzymes, the primases1. However, some replication systems have evolved to use cellular DNA-dependent RNA polymerase for primer synthesis1,2. The main requirement for the replication primer, an exposed RNA 3′ end annealed to the DNA template, is not compatible with known conformations of the transcription elongation complex3, raising a question of how the priming is achieved. Here we show that a previously unrecognized kind of transcription complex is formed during RNA polymerase-catalysed synthesis of the M13 bacteriophage replication primer. The complex contains an overextended RNA–DNA hybrid bound in the RNA-polymerase trough that is normally occupied by downstream double-stranded DNA, thus leaving the 3′ end of the RNA available for interaction with DNA polymerase. Transcription complexes with similar topology may prime the replication of other bacterial mobile elements and may regulate transcription elongation under conditions that favour the formation of an extended RNA–DNA hybrid.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Kornberg, A. & Baker, T. A. DNA Replication 2nd edn (Freeman, New York, 1992)
Geider, K. & Kornberg, A. Conversion of the M13 viral single strand to the double-stranded replicative forms by purified proteins. J. Biol. Chem. 249, 3999–4005 (1974)
Nudler, E. Transcription elongation: structural basis and mechanisms. J. Mol. Biol. 288, 1–12 (1999)
Zenkin, N. & Severinov, K. The role of RNA polymerase sigma subunit in promoter-independent initiation of transcription. Proc. Natl Acad. Sci. USA 101, 4396–4400 (2004)
Horiuchi, K. Initiation mechanisms in replication of filamentous phage DNA. Genes Cells 2, 425–432 (1997)
Komissarova, N. & Kashlev, M. Transcriptional arrest: Escherichia coli RNA polymerase translocates backward, leaving the 3′ end of the RNA intact and extruded. Proc. Natl Acad. Sci. USA 94, 1755–1760 (1997)
Korzheva, N. et al. A structural model of transcription elongation. Science 289, 619–625 (2000)
Nechaev, S. & Severinov, K. Inhibition of Escherichia coli RNA polymerase by bacteriophage T7 gene 2 protein. J. Mol. Biol. 289, 815–826 (1999)
Kramer, M. G., Espinosa, M., Misra, T. K. & Khan, S. A. Characterization of a single-strand origin, ssoU, required for broad host range replication of rolling-circle plasmids. Mol. Microbiol. 33, 466–475 (1999)
Kramer, M. G., Khan, S. A. & Espinosa, M. Plasmid rolling circle replication: identification of the RNA polymerase-directed primer RNA and requirement for DNA polymerase I for lagging strand synthesis. EMBO J. 16, 5784–5795 (1997)
Masai, H. & Arai, K. Frpo: a novel single-stranded DNA promoter for transcription and for primer RNA synthesis of DNA replication. Cell 89, 897–907 (1997)
Masukata, H. & Tomizawa, J. A mechanism of formation of a persistent hybrid between elongating RNA and template DNA. Cell 62, 331–338 (1990)
Kashlev, M. et al. Histidine-tagged RNA polymerase of Escherichia coli and transcription in solid phase. Methods Enzymol. 274, 326–334 (1996)
Kuznedelov, K., Korzheva, N., Mustaev, A. & Severinov, K. Structure-based analysis of RNA polymerase function: the largest subunit's rudder contributes critically to elongation complex stability and is not involved in the maintenance of RNA–DNA hybrid length. EMBO J. 21, 1369–1378 (2002)
Mustaev, A. et al. Strategies and methods of cross-linking of RNA polymerase active center. Methods Enzymol. 371, 191–206 (2003)
Acknowledgements
This work is dedicated to Dmitry Salonin who inspired it. We thank A. Mustaev for crosslinking reagents. This work was supported by an NIH R01 grant and a Borroughs Wellcome Career Award to K.S.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
Reprints and permissions information is available at npg.nature.com/reprintsandpermissions. The authors declare no competing financial interests.
Supplementary information
Supplementary Figure 1
This figure shows a schematic view of formation of the priming complex on bacteriophage M13 origin of replication during primer synthesis by DNA-dependent RNA polymerase. The unusual topology of the origin leads to conformational rearrangement during RNA polymerase transcription that extrudes the 3' end of the primer RNA, base-paired to template DNA, from the RNAP catalytic centre and makes it available for interaction with DNA polymerase. (DOC 78 kb)
Rights and permissions
About this article
Cite this article
Zenkin, N., Naryshkina, T., Kuznedelov, K. et al. The mechanism of DNA replication primer synthesis by RNA polymerase. Nature 439, 617–620 (2006). https://doi.org/10.1038/nature04337
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nature04337
This article is cited by
-
Single-peptide DNA-dependent RNA polymerase homologous to multi-subunit RNA polymerase
Nature Communications (2017)
-
Hypothesis: Emergence of Translation as a Result of RNA Helicase Evolution
Journal of Molecular Evolution (2012)
-
A new basal promoter element recognized by RNA polymerase core enzyme
The EMBO Journal (2011)
-
Prime-time progress
Nature (2006)
-
The double helix unzipped
Nature Reviews Molecular Cell Biology (2006)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.