Abstract
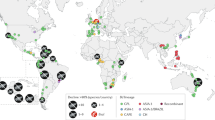
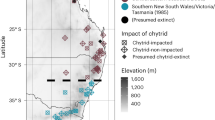
As the Earth warms, many species are likely to disappear, often because of changing disease dynamics. Here we show that a recent mass extinction associated with pathogen outbreaks is tied to global warming. Seventeen years ago, in the mountains of Costa Rica, the Monteverde harlequin frog (Atelopus sp.) vanished along with the golden toad (Bufo periglenes). An estimated 67% of the 110 or so species of Atelopus, which are endemic to the American tropics, have met the same fate, and a pathogenic chytrid fungus (Batrachochytrium dendrobatidis) is implicated. Analysing the timing of losses in relation to changes in sea surface and air temperatures, we conclude with ‘very high confidence’ (> 99%, following the Intergovernmental Panel on Climate Change, IPCC) that large-scale warming is a key factor in the disappearances. We propose that temperatures at many highland localities are shifting towards the growth optimum of Batrachochytrium, thus encouraging outbreaks. With climate change promoting infectious disease and eroding biodiversity, the urgency of reducing greenhouse-gas concentrations is now undeniable.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Houghton, J. T. et al. (eds) Climate Change 2001, The Scientific Basis. Third Assessment Report of the Intergovernmental Panel on Climate Change (Cambridge Univ. Press, Cambridge, 2001)
Santer, B. D. et al. Contributions of anthropogenic and natural forcing to recent tropopause height changes. Science 301, 479–483 (2003)
Stott, P. A. Attribution of regional-scale temperature changes to anthropogenic and natural causes. Geophys. Res. Lett. 30, 1728–1731 (2003)
Barnett, T. P. et al. Penetration of human-induced warming into the world's oceans. Science 309, 284–287 (2005)
Parmesan, C. & Yohe, G. A globally coherent fingerprint of climate change impacts across natural systems. Nature 421, 37–42 (2003)
Root, T. L. et al. Fingerprints of global warming on wild animals and plants. Nature 421, 57–60 (2003)
Root, T. L., MacMynowski, D. P., Mastrandrea, M. D. & Schneider, S. H. Human-modified temperatures induce species changes: Joint attribution. Proc. Natl Acad. Sci. USA 102, 7465–7469 (2005)
Lovejoy, T. & Hannah, L. (eds) Climate Change and Biodiversity (Yale Univ. Press, New Haven, Connecticut, 2005)
Epstein, P. R. Climate change and emerging infectious diseases. Microbes Infect. 3, 747–754 (2001)
Harvell, C. D. et al. Climate warming and disease risks for terrestrial and marine biota. Science 296, 2158–2162 (2002)
Rodó, X., Pascual, M., Fuchs, G. & Faruque, A. S. G. ENSO and cholera: A nonstationary link related to climate change? Proc. Natl Acad. Sci. USA 99, 12901–12906 (2002)
Thomas, C. D. et al. Extinction risk from climate change. Nature 427, 145–148 (2004)
Stuart, S. M. et al. Status and trends of amphibian declines and extinctions worldwide. Science 306, 1783–1786 (2004)
Alford, R. A. & Richards, S. J. Global amphibian declines: a problem in applied ecology. Annu. Rev. Ecol. Syst. 30, 133–165 (1999)
Kiesecker, J. M., Blaustein, A. R. & Belden, L. K. Complex causes of amphibian population declines. Nature 410, 681–684 (2001)
Blaustein, A. R. & Kiesecker, J. M. Complexity in conservation: lessons from the global decline of amphibian populations. Ecol. Lett. 5, 597–608 (2002)
Berger, L. et al. Chytridiomycosis causes amphibian mortality associated with population declines in the rain forests of Australia and Central America. Proc. Natl Acad. Sci. USA 95, 9031–9036 (1998)
Carey, C. & Alexander, M. A. Climate change and amphibian declines: Is there a link? Divers. Distrib. 9, 111–121 (2003)
Daszak, P., Cunningham, A. A. & Hyatt, A. D. Infectious disease and amphibian population declines. Divers. Distrib. 9, 141–150 (2003)
Lips, K. R., Green, D. E. & Pappendick, R. Chytridiomycosis in wild frogs from southern Costa Rica. J. Herpetol. 37, 215–218 (2003)
Ron, S. R., Duellman, W. E., Coloma, L. A. & Bustamante, M. Population decline of the Jambato toad Atelopus ignescens (Anura: Bufonidae) in the Andes of Ecuador. J. Herpetol. 37, 116–126 (2003)
Berger, L. et al. Effect of season and temperature on mortality in amphibians due to chytridiomycosis. Aust. Vet. J. 82, 434–439 (2004)
Burrowes, P. A., Joglar, R. L. & Green, D. E. Potential causes for amphibian declines in Puerto Rico. Herpetologica 60, 141–154 (2004)
Piotrowski, J. S., Annis, S. L. & Longcore, J. E. Physiology of Batrachochytrium dendrobatidis, a chytrid pathogen of amphibians. Mycologia 96, 9–15 (2004)
Retallick, R. W. R., McCallum, H. & Speare, R. Endemic infection of the amphibian chytrid fungus in a frog community post-decline. PLoS Biol. 2, 1966–1971 (2004)
La Marca, E. et al. Catastrophic population declines and extinctions in Neotropical harlequin frogs (Bufonidae: Atelopus). Biotropica 37, 190–201 (2005)
Ron, S. R. Predicting the distribution of the amphibian pathogen Batrachochytrium dendrobatidis in the New World. Biotropica 37, 209–221 (2005)
Merino-Viteri, A., Coloma, L. A. & Almendáriz, A. in Studies on the Andean Frogs of the Genera Telmatobius and Bratrachophrynus (eds Lavilla, E. O. & De la Riva, I.) 9–37 (Asociación Herpetológica Española, Valencia, 2005)
Pounds, J. A. & Crump, M. L. Amphibian declines and climate disturbance: the case of the golden toad and the harlequin frog. Conserv. Biol. 8, 72–85 (1994)
Pounds, J. A., Fogden, M. P. L., Savage, J. M. & Gorman, G. C. Tests of null models for amphibian declines on a tropical mountain. Conserv. Biol. 11, 1307–1322 (1997)
Pounds, J. A., Fogden, M. P. L. & Campbell, J. H. Biological response to climate change on a tropical mountain. Nature 398, 611–615 (1999)
Pounds, J. A. in Monteverde: Ecology and Conservation of a Tropical Cloud Forest (eds Nadkarni, N. M. & Wheelwright, N. T.) 149–177 (Oxford Univ. Press, Oxford, 2000)
Pounds, J. A. Climate and amphibian declines. Nature 410, 639–640 (2001)
Pounds, J. A. & Puschendorf, R. Ecology: Clouded futures. Nature 427, 107–109 (2004)
Root, T. L. & Schneider, S. H. Ecology and climate: Research strategies and implications. Science 269, 334–341 (1995)
Lawton, R. O., Nair, U. S., Pielke, R. A. Sr & Welch, R. M. Climatic impact of tropical lowland deforestation on nearby montane cloud forests. Science 294, 584–587 (2001)
Vuille, M., Bradley, R. S., Werner, M. & Keimig, F. 20th century climate change in the tropical Andes: observations and model results. Clim. Change 59, 75–99 (2003)
Malhi, Y. & Wright, J. Spatial patterns and recent trends in the climate of tropical forest regions. Proc. R. Soc. Lond. B 359, 311–329 (2004)
Thompson, L. G. et al. Tropical glacier and ice core evidence of climate change on annual to millennial time scales. Clim. Change 59, 137–155 (2003)
Bush, M. B., Silman, M. R. & Urrego, D. H. 48,000 years of climate and forest change in a biodiversity hot spot. Science 303, 827–829 (2004)
Still, C. J., Foster, P. N. & Schneider, S. H. Simulating the effects of climate change on tropical montane cloud forests. Nature 398, 608–610 (1999)
Wentz, F. J. & Schabel, M. Precise climate monitoring using complementary satellite data sets. Nature 403, 414–416 (2000)
Ross, R. J. & Elliott, W. P. Radiosonde-based Northern Hemisphere tropospheric water vapor trends. J. Clim. 14, 1602–1612 (2001)
Penner, J. E. Climate change: The cloud conundrum. Nature 432, 962–963 (2004)
Dai, A., Trenberth, K. E. & Karl, T. R. Effects of clouds, soil moisture, precipitation, and water vapor on diurnal temperature range. J. Clim. 12, 2451–2473 (1999)
Gómez, I. E. & Fernández, W. Variación interanual de la temperatura en Costa Rica. Top. Meteor. Oceanogr. 3, 27–44 (1996)
Quintana-Gómez, R. A. Trends of maximum and minimum temperatures in northern South America. J. Clim. 12, 2104–2112 (1999)
Cáceres, L., Mejia, R. & Otaneda, G. in Consecuencias Climáticas e Hidrológicas del Evento El Niño a Escala Regional y Local. Incidencia en America del Sur (eds Cadier, E., Gómez, G., Galarraga, R. & Fernández-Jáuregui, C.) 〈http://www.unesco.org.uy/phi/libros/enso/caceres.html〉 (UNESCO-PHI, 1998)
Sanchez-Azofeifa, G. A., Harriss, R. C. & Skole, D. L. Deforestation in Costa Rica: a quantitative analysis using remote sensing imagery. Biotropica 33, 378–384 (2001)
Pielke, R. A. et al. A comprehensive meteorological modeling system—RAMS. Meteorol. Atmos. Phys 49, 69–91 (1992)
Acknowledgements
We thank T. P. Mitchell at JISAO and S. J. Worley at NCAR for help in obtaining climate data, R. A. Alford, F. Bolaños, J. P. Collins, R. O. Lawton, K. R. Lips, M. D. Mastrandrea, K. G. Murray, P. Ramírez and B. D. Santer for discussion, and the many contributors to the Atelopus database. S. H. Schneider, A. R. Blaustein and C. Parmesan commented on earlier drafts of the manuscript. The Declining Amphibian Populations Task Force and Conservation International's Critically Endangered Neotropical Species Fund provided partial funding to J.A.P. The Canada Foundation for Innovation and the Tinker Foundation helped produce the remote-sensing databases. The Research and Analysis Network for Neotropical Amphibians and the US National Science Foundation sponsored meetings that facilitated portions of the study. Author Contributions All authors after the first are listed alphabetically. J.A.P. conceived, designed and orchestrated the study, conducted most of the analyses (principally with J.A.C. and K.L.M.), formulated the chytrid-thermal-optimum hypothesis (with R.P.), and wrote the paper (with editing by J.A.C. and K.L.M.). M.R.B., L.A.C., M.P.L.F., E.L.M., A.M.-V. and S.R.R. provided key data and background information. E.L.M. compiled the Atelopus database (with B.E.Y.). P.N.F. conducted the climate simulations. G.A.S.-A. analysed the remote-sensing data. C.J.S. helped with the climate analyses and their interpretation. B.E.Y. obtained funding for meetings, provided logistics, and analysed GAA data for New World amphibians.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
Reprints and permissions information is available at npg.nature.com/reprintsandpermissions. The authors declare no competing financial interests.
Supplementary information
Supplementary Notes
This file contains Supplementary Notes, Supplementary Figures 1–5, Supplementary Tables 1 and 2, Supplementary References and Supplementary Appendix A. (PDF 360 kb)
Rights and permissions
About this article
Cite this article
Alan Pounds, J., Bustamante, M., Coloma, L. et al. Widespread amphibian extinctions from epidemic disease driven by global warming. Nature 439, 161–167 (2006). https://doi.org/10.1038/nature04246
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nature04246
This article is cited by
-
Irrigation dams threaten Brazilian biodiversity
Environmental Management (2024)
-
A framework to support the identification of critical habitat for wide-ranging species at risk under climate change
Biodiversity and Conservation (2024)
-
Poison frog social behaviour under global change: potential impacts and future challenges
acta ethologica (2023)
-
May future climate change promote the invasion of the marsh frog? An integrative thermo-physiological study
Oecologia (2023)
-
Simulating the response of a threatened amphibian to climate-induced reductions in breeding habitat
Landscape Ecology (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.