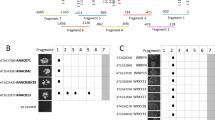
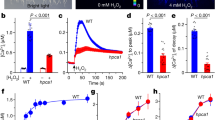
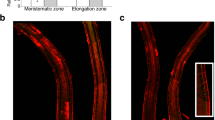
Abstract
Active oxygen species (AOS) generated in response to stimuli and during development can function as signalling molecules in eukaryotes, leading to specific downstream responses1,2. In plants these include such diverse processes as coping with stress (for example pathogen attack3, wounding4 and oxygen deprivation5), abscisic-acid-induced guard-cell closure6, and cellular development (for example root hair growth7). Despite the importance of signalling via AOS in eukaryotes, little is known about the protein components operating downstream of AOS that mediate any of these processes. Here we show that expression of an Arabidopsis thaliana gene (OXI1) encoding a serine/threonine kinase is induced in response to a wide range of H2O2-generating stimuli. OXI1 kinase activity is itself also induced by H2O2 in vivo. OXI1 is required for full activation of the mitogen-activated protein kinases (MAPKs) MPK3 and MPK6 after treatment with AOS or elicitor and is necessary for at least two very different AOS-mediated processes: basal resistance to Peronospora parasitica infection, and root hair growth. Thus, OXI1 is an essential part of the signal transduction pathway linking oxidative burst signals to diverse downstream responses.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Finkel, T. Oxygen radicals and signaling. Curr. Opin. Cell Biol. 10, 248–253 (1998)
Hancock, J. T., Desikan, R. & Neill, S. J. Role of reactive oxygen species in cell signalling pathways. Biochem. Soc. Trans. 29, 345–350 (2001)
Levine, A., Tenhaken, R., Dixon, R. & Lamb, C. H2O2 from the oxidative burst orchestrates the plant hypersensitive disease resistance response. Cell 79, 583–593 (1994)
Orozco-Cárdenas, M. L., Narvaez-Vasquez, J. & Ryan, C. A. Hydrogen peroxide acts as a second messenger for the induction of defense genes in tomato plants in response to wounding, systemin, and methyl jasmonate. Plant Cell 13, 179–191 (2001)
Baxter-Burrell, A., Chang, R., Springer, P. & Bailey-Serres, J. Gene and enhancer trap transposable elements reveal oxygen deprivation-regulated genes and their complex patterns of expression in Arabidopsis. Ann. Bot. 91 (special number), 129–141 (2003)
Pei, Z.-M. et al. Calcium channels activated by hydrogen peroxide mediate abscisic acid signalling in guard cells. Nature 406, 731–734 (2000)
Foreman, J. et al. Reactive oxygen species produced by NADPH oxidase regulate plant cell growth. Nature 422, 442–446 (2003)
Knight, H., Brandt, S. & Knight, M. R. A history of stress alters drought calcium signalling pathways in Arabidopsis. Plant J. 16, 681–687 (1998)
Orozco-Cárdenas, M. & Ryan, C. A. Hydrogen peroxide is generated systemically in plant leaves by wounding and systemin via the octadecanoid pathway. Proc. Natl Acad. Sci. USA 96, 6553–6557 (1999)
Hückelhoven, R., Fodor, J., Preis, C. & Kogel, K.-H. Hypersensitive cell death and papilla formation in barley attacked by the powdery mildew fungus are associated with hydrogen peroxide but not with salicylic acid accumulation. Plant Physiol. 119, 1251–1260 (1999)
Torres, M. A., Dangl, J. L. & Jones, J. D. Arabidopsis gp91phox homologues AtrbohD and AtrbohF are required for accumulation of reactive oxygen intermediates in the plant defense response. Proc. Natl Acad. Sci. USA 99, 517–522 (2002)
Borden, S. & Higgins, V. J. Hydrogen peroxide plays a critical role in the defence response of tomato to Cladosporium fulvum. Physiol. Mol. Plant Pathol. 61, 227–236 (2002)
Krysan, P. J., Young, J. C. & Sussmann, M. R. T-DNA as an insertional mutagen in Arabidopsis. Plant Cell 11, 2283–2290 (1999)
Parker, J. S., Cavell, A. C., Dolan, L., Roberts, K. & Grierson, C. S. Genetic interactions during root hair morphogenesis in Arabidopsis. Plant Cell 12, 1961–1974 (2000)
Kovtun, Y., Chiu, W.-L., Tena, G. & Sheen, J. Functional analysis of oxidative stress-activated mitogen-activated protein kinase cascade in plants. Proc. Natl Acad. Sci. USA 97, 2940–2945 (2000)
Asai, T. et al. MAP kinase signalling cascade in Arabidopsis innate immunity. Nature 415, 977–983 (2002)
Ichimura, K., Mizoguchi, T., Yoshida, R., Yuasa, T. & Shinozaki, K. Various abiotic stresses rapidly activate Arabidopsis MAP kinases ATMPK4 and ATMPK6. Plant J. 24, 655–665 (2000)
Samaj, J. et al. Involvement of the mitogen-activated protein kinase SIMK in regulation of root hair tip growth. EMBO J. 21, 3296–3306 (2002)
Allan, A. C. & Fluhr, R. Two distinct sources of elicited reactive oxygen species in tobacco epidermal cells. Plant Cell 9, 1559–1572 (1997)
Oyama, T., Shimura, Y. & Okada, K. The IRE gene encodes a protein kinase homologue and modulates root hair growth in Arabidopsis. Plant J. 30, 289–299 (2002)
Pietrzak, M., Shillito, R. D., Hohn, T. & Potrykus, I. Expression in plants of two bacterial antibiotic-resistance genes after protoplast transformation with a new plant expression vector. Nucleic Acids Res. 14, 5857–5868 (1986)
Bevan, M. W. Binary Agrobacterium vectors for plant transformation. Nucleic Acids Res. 12, 8711–8721 (1984)
Deblaere, R. et al. Efficient octopine Ti plasmid-derived vectors for Agrobacterium-mediated gene-transfer to plants. Nucleic Acids Res. 13, 4777–4788 (1985)
Clough, S. J. & Bent, A. F. Floral dip: a simplified method for Agrobacterium-mediated transformation of Arabidopsis thaliana. Plant J. 16, 735–743 (1998)
Martin, T., Schmidt, R., Altmann, T. & Frommer, W. Non-destructive assay systems for detection of β-glucuronidase activity in higher plants. Plant Mol. Biol. Reptr 10, 37–46 (1992)
Weigel, D. & Glazebrook, J. Arabidopsis – a Laboratory Manual (Cold Spring Harbor Laboratory Press, Cold Spring Harbor, NY, 2001)
Wymer, C. L., Bibikova, T. N. & Gilroy, S. Cytoplasmic free calcium distributions during the development of root hairs of Arabidopsis thaliana. Plant J. 12, 427–439 (1997)
Cardinale, F., Meskiene, I., Ouaked, F. & Hirt, H. Protein kinase signaling pathways at the level of two distinct mitogen-activated protein kinase kinases. Plant Cell 14, 703–711 (2002)
Acknowledgements
We thank A. Rehmany for the P. parasitica isolates; A. Hailey for statistical analysis; B. Ellis and G. Miles for discussions; and N. Evans, S. Gurr and R. Capper and K. Denby for practical and moral support. This work was funded by the BBSRC, the Gatsby Foundation, the EU TMR program, the Cannon Collins Educational Trust for Southern Africa (CCETSA), the South African National Research Foundation (NRF), and the Austrian Science Foundation.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare that they have no competing financial interests.
Supplementary information
Supplementary Figure 1
Plants containing an OXI1::GUS gene construct were wounded with tweezers and stained for (a) H2O2 production with diaminobenzidine (DAB) or (b) expression of the GUS protein24. (PDF 236 kb)
Supplementary Figure 2
Low magnification (top) and higher magnification (bottom) of roots grown on (a) ordinary agar and (b) phytagel. (PDF 132 kb)
Supplementary Figure 3
Distribution of mature root hair lengths of (a) oxi1 and Ws-2 seedlings and (b) oxi1 and complemented oxi1 seedlings (oxi1 + OXI1) with roots growing through air. (PDF 45 kb)
Rights and permissions
About this article
Cite this article
Rentel, M., Lecourieux, D., Ouaked, F. et al. OXI1 kinase is necessary for oxidative burst-mediated signalling in Arabidopsis. Nature 427, 858–861 (2004). https://doi.org/10.1038/nature02353
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nature02353
This article is cited by
-
Cadmium toxicity: its’ uptake and retaliation by plant defence system and ja signaling
BioMetals (2024)
-
Comprehensive meta-QTL analysis for dissecting the genetic architecture of stripe rust resistance in bread wheat
BMC Genomics (2023)
-
Multiple pathways mediate chloroplast singlet oxygen stress signaling
Plant Molecular Biology (2023)
-
Phenylalanine suppresses cell death caused by loss of fumarylacetoacetate hydrolase in Arabidopsis
Scientific Reports (2022)
-
Heavy Metal Stress–Induced Activation of Mitogen-Activated Protein Kinase Signalling Cascade in Plants
Plant Molecular Biology Reporter (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.