Abstract
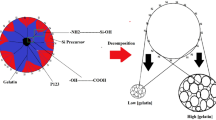
Since the discovery1 of MCM-41 more than ten years ago, many investigations have explored the suitability of hexagonal mesoporous silicas for potential practical applications2,3,4. These range from catalysis5,6 and optically active materials7,8 to polymerization science9,10,11,12, separation technology3,13,14 and drug delivery15,16,17,18, with recent successes in the fabrication of hybrid mesoporous organosilicas19,20,21 expected to open up further application possibilities. Because the pore voids of this class of materials exhibit relatively narrow pore size distributions in the range of 2–4 nm in diameter, mesoporous silicas can selectively include organic compounds and release them continuously at a later stage. The functionalization of MCM-41 pore voids with photoactive derivatives22,23,24,25 provides influence over the material's absorption behaviour, but full control over the release process remains difficult. Here we show that the uptake, storage and release of organic molecules in MCM-41 can be regulated through the photocontrolled and reversible intermolecular dimerization26,27 of coumarin derivatives attached to the pore outlets. Successful functionalization requires uncalcined MCM-41 still filled with the template molecules that directed the formation of its pores, to ensure that coumarin derivatives attach preferentially to the pore outlets, rather than their inside walls. We find that this feature and the one-dimensional, isolated nature of the individual pores allow for efficient and reversible photocontrol over guest access to the material's interior.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Kresge, C. T., Leonowicz, M. E., Roth, W. J., Vartuli, J. C. & Beck, J. S. Ordered mesoporous molecular sieves synthesized by a liquid-crystal template mechanism. Nature 359, 710–712 (1992)
Ying, J. Y., Mehnert, C. P. & Wong, M. S. Synthesis and applications of supramolecular-templated mesoporous materials. Angew. Chem. Int. Edn Engl. 38, 56–77 (1999)
Stein, A., Melde, B. J. & Schroden, R. C. Hybrid inorganic-organic mesoporous silicates—Nanoscopic reactors coming of age. Adv. Mater. 12, 1403–1419 (2000)
Davis, M. E. Ordered porous materials for emerging applications. Nature 417, 813–821 (2002)
Maschmeyer, T., Rey, F., Sanker, G. & Thomas, J. M. Heterogeneous catalysts obtained by grafting metallocene complexes onto mesoporous silica. Nature 378, 159–162 (1995)
Corma, A. From microporous to mesoporous molecular sieve materials and their use in catalysis. Chem. Rev. 97, 2373–2419 (1997)
Nguyen, T. Q., Wu, J. J., Doan, V., Schwartz, B. J. & Tolbert, S. H. Control of energy transfer in oriented conjugated polymer-mesoporous silica composites. Science 288, 652–656 (2000)
Ganschow, M., Wark, M., Wohrle, D. & Schulz-Ekloff, G. Anchoring of functional dye molecules in MCM-41 by microwave-assisted hydrothermal co-condensation. Angew. Chem. Int. Edn Engl. 39, 161–163 (2000)
Wu, C. G. & Bein, T. Conducting polyaniline filaments in a mesoporous channel host. Science 264, 1757–1759 (1994)
Zhou, W. Z. et al. Ordering of ruthenium cluster carbonyls in mesoporous silica. Science 280, 705–708 (1998)
Kageyama, K., Tamazawa, J. & Aida, T. Extrusion polymerization: Catalyzed synthesis of crystalline linear polyethylene nanofibers within a mesoporous silica. Science 285, 2113–2115 (1999)
Spange, S. et al. Cationic host-guest polymerization of N-vinylcarbazole and vinyl ethers in MCM-41, MCM-48, and nanoporous glasses. Chem. Eur. J. A 7, 3722–3728 (2001)
Feng, X. et al. Functionalized monolayers on ordered mesoporous supports. Science 276, 923–926 (1997)
Mercier, L. & Pinnavaia, T. J. Access in mesoporous materials: Advantages of a uniform pore structure in the design of a heavy metal ion adsorbent for environmental remediation. Adv. Mater. 9, 500–503 (1997)
Ahola, M., Kortesuo, P., Kangasniemi, I., Kiesvaara, J. & Antti Yli-Urpo, A. Silica xerogel carrier material for controlled release of toremifene citrate. Int. J. Pharm. 195, 219–227 (2000)
Arcos, D., Ragel, C. V. & Vallet-Regí, M. Bioactivity in glass/PMMA composites used as drug delivery system. Biomaterials 22, 701–708 (2001)
Czuryszkiewicz, T. et al. Drug release from biodegradable silica fibers. J. Non-Cryst. Solids 306, 1–10 (2002)
Vallet-Regi, M., Rámila, A., del Real, R. P. & Pérez-Pariente, J. A new property of MCM-41: Drug delivery system. Chem. Mater. 13, 308–311 (2001)
Asefa, T., MacLachan, M. J., Coombs, N. & Ozin, G. A. Periodic mesoporous organosilicas with organic groups inside the channel walls. Nature 402, 867–871 (1999)
Fujiwara, M. et al. Preparation of MCM-41/nafion composite material; a selective catalyst of α-methylstyrene dimerization. J. Chem. Soc. Chem. Commun. 1523–1524 (2000)
Inagaki, S., Guan, S., Ohsuna, T. & Terasaki, O. An ordered mesoporous organosilica hybrid material with a crystal-like wall structure. Nature 416, 304–307 (2002)
Alvaro, M., Ferrer, B., García, H. & Rey, F. Photochemical modification of the surface area and tortuosity of a trans-1,2-bis(4-pyridyl)ethylene periodic mesoporous MCM organosilica. J. Chem. Soc. Chem. Commun. 2012–2013 (2002)
Hoffmann, K., Resch-Genger, U. & Marlow, F. Photoinduced switching of nanocomposites consisting of azobenzene and molecular sieves: Investigation of the switching states. Micropor. Mesopor. Mater. 41, 99–106 (2000)
Wirnsberger, G., Scott, B. J., Chmelka, B. F. & Stucky, G. D. Fast response photochromic mesostructures. Adv. Mater. 12, 1450–1454 (2000)
Schomburg, C., Wark, M., Rohlfing, Y., Schulz-Ekloff, G. & Wöhrle, D. Photochromism of spiropyran in molecular sieve voids: Effects of host-guest interaction on isomer status, switching stability and reversibility. J. Mater. Chem. 11, 2014–2021 (2001)
Chujo, Y., Sada, K. & Saegusa, T. Polyoxazoline having a coumarin moiety as a pendant group. Synthesis and photogelation. Macromolecules 23, 2693–2697 (1990)
Li, W. J., Lynch, V., Thompson, H. & Fox, M. A. Self-assembled monolayers of 7-(10-thiodecoxy)coumarin on gold: Synthesis, characterization, and photodimerization. J. Am. Chem. Soc. 119, 7211–7217 (1997)
Berezin, M. Y., Dzenitis, J. M., Hughes, B. M. & Ho, S. V. Separation of sterols using zeolites. Phys. Chem. Chem. Phys. 3, 2184–2189 (2001)
Kruk, M., Antochshuk, V., Matos, J. R., Mercuri, L. P. & Jaroniec, M. Determination and tailoring the pore entrance size in ordered silicas with cage-like mesoporous structures. J. Am. Chem. Soc. 124, 768–769 (2002)
Zhao, X. S., Lu, G. Q. M. & Hu, X. A novel method for tailoring the pore-opening size of MCM-41 materials. J. Chem. Soc. Chem. Commun. 1391–1392 (1999)
Lim, M. H. & Stein, A. Comparative studies of grafting and direct syntheses of inorganic-organic hybrid mesoporous materials. Chem. Mater. 11, 3285–3295 (1999)
Acknowledgements
N.K.M. was supported by an STA fellowship.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare that they have no competing financial interests.
Rights and permissions
About this article
Cite this article
Mal, N., Fujiwara, M. & Tanaka, Y. Photocontrolled reversible release of guest molecules from coumarin-modified mesoporous silica. Nature 421, 350–353 (2003). https://doi.org/10.1038/nature01362
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nature01362
This article is cited by
-
The calcination temperature effect in the synthesis of nanohydroxyapatite: characterization and its application as a nanocarrier
Applied Nanoscience (2023)
-
Nanoarchitectonics: a New Horizon for Drug Targeting
Journal of Pharmaceutical Innovation (2022)
-
A shape memory porous sponge with tunability in both surface wettability and pore size for smart molecule release
Science China Materials (2021)
-
Oxidative formation of bis-N-methylquinolinone from anti-head-to-head N-methylquinolinone cyclodimer
Photochemical & Photobiological Sciences (2021)
-
Redox-triggered switching in three-dimensional covalent organic frameworks
Nature Communications (2020)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.