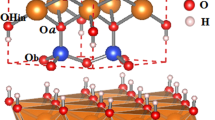
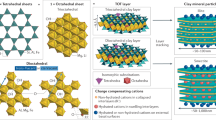
Abstract
Illite–smectite interstratified clay minerals are ubiquitous in sedimentary basins and they have been linked to the maturation, migration and trapping of hydrocarbons1, rock cementation2, evolution of porewater chemistry during diagenesis3 and the development of pore pressure4. But, despite the importance of these clays, their structures are controversial. Two competing models exist, each with profoundly different consequences for the understanding of diagenetic processes: model A views such interstratified clays as a stacking of layers identical to endmember illite and smectite layers, implying discrete and independently formed units (fundamental particles)5, whereas model B views the clays as composed of crystallites with a unique structure that maintains coherency over much greater distances, in line with local charge balance about interlayers6. Here we use first-principles density-functional theory to explore the energetics and structures of these two models for an illite–smectite interstratified clay mineral with a ratio of 1:1 and a Reichweite parameter of 1. We find that the total energy of model B is 2.3 kJ atom-1 mol-1 lower than that of model A, and that this energy difference can be traced to structural distortions in model A due to local charge imbalance. The greater stability of model B requires re-evaluation of the evolution of the smectite-to-illite sequence of clay minerals, including the nature of coexisting species, stability relations, growth mechanisms and the model of fundamental particles.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Weaver, C. E. Possible uses of clay minerals in search for oil. Bull. Am. Assoc. Petrol. Geol. 44, 1505–1518 (1960)
Boles, J. R. & Franks, S. G. Clay diagenesis in Wilcox sandstones of southwest Texas—implications of smectite diagenesis on sandstone cementation. J. Sedim. Petrol. 49, 55–70 (1979)
Brown, K. M., Saffer, D. M. & Bekins, B. A. Smectite diagenesis, pore-water freshening, and fluid flow at the toe of the Nankai wedge. Earth Planet. Sci. Lett. 194, 97–109 (2001)
Bethke, C. M. Inverse hydrologic analysis of the distribution and origin of Gulf Coast-type geopressured zones. J. Geophys. Res. 91, 6535–6545 (1986)
Nadeau, P. H., Wilson, M. J., McHardy, W. J. & Tait, J. M. Interstratified clays as fundamental particles. Science 255, 923–925 (1984)
Altaner, S. P., Weiss, C. A. & Kirkpatrick, R. J. Evidence from 29Si NMR for the structure of mixed layer illite–smectite clay minerals. Nature 331, 699–702 (1988)
Brown, G. Crystal-structures of clay-minerals and related phyllosilicates. Phil. Trans. R. Soc. Lond. A 311, 221–240 (1984)
Kresse, G. & Furthmüller, J. Efficient iterative schemes for ab initio total-energy calculations using a plane-wave basis set. Phys. Rev. B 54, 11169–11186 (1996)
Stixrude, L. Talc under tension and compression: spinodal instability and structure at high pressure. J. Geophys. Res. (in the press)
Berman, R. G. Internally-consistent thermodynamic data for minerals in the system Na2O-K2O-CaO-MgO-FeO-Fe2O3-Al2O3-SiO2-TiO2-H2O-CO2 . J. Petrol. 29, 445–522 (1988)
Robinson, K., Gibbs, G. V. & Ribbe, P. H. Quadratic elongation—quantitative measure of distortion in coordination polyhedra. Science 172, 567–570 (1971)
Smyth, J. R. & Bish, D. L. Crystal Structures and Cation Sites of the Rock-Forming Minerals (Allen and Unwin, Boston, 1988)
Rothbauer, R. Untersuchung eines 2M1-Muskovits mit Neutronenstrahlen. Neues Jb. Mineral. Mh. 4, 143–154 (1971)
Rossman, G. R. Spectroscopy of micas. Rev. Mineral. 13, 145–181 (1984)
Essene, E. J. & Peacor, D. R. Clay mineral thermometry—A critical perspective. Clays Clay Minerals 43, 540–553 (1995)
Dong, H., Peacor, D. R. & Freed, R. L. Phase relations among smectite, R1 illite-smectite, and illite. Am. Mineral. 82, 379–391 (1997)
Schroeder, P. A. & Irby, R. Detailed X-ray diffraction characterization of illite-smectite from an Ordovician K-bentonite, Walker County, Georgia, USA. Clay Minerals 33, 671–674 (1998)
Shau, Y. H., Peacor, D. R. & Essene, E. J. Corrensite and mixed-layer chlorite/corrensite in metabasalt from northern Taiwan: TEM/AEM, EMPA, XRD, and optical studies. Contrib. Mineral. Petrol. 105, 123–142 (1990)
Srodon, J., Morgan, D. J., Eslinger, E. V., Eberl, D. D. & Karlinger, M. A. Chemistry of illite/smectite and end-member illite. Clays Clay Minerals 34, 368–378 (1986)
Srodon, J. Nature of mixed-layer clays and mechanisms of their formation and alteration. Annu. Rev. Earth Planet. Sci. 27, 19–53 (1999)
Nadeau, P. H., Wilson, M. J., McHardy, W. J. & Tait, J. M. Interparticle diffraction: a new concept for interstratified clays. Clay Minerals 19, 757–769 (1984)
Kasama, T., Murakami, T., Kohyama, N. & Watanabe, T. Experimental mixtures of smectite and rectorite: Re-investigation of “fundamental particles” and “interparticle diffraction”. Am. Mineral. 86, 105–114 (2001)
Jakobsen, H. J., Nielsen, N. C. & Lindgreen, H. Sequences of charged sheets in rectorite. Am. Mineral. 80, 247–252 (1995)
Altaner, S. P. & Ylagan, R. F. Comparison of structural models of mixed-layer illite-smectite and reaction mechanisms of smectite illitization. Clays Clay Minerals 45, 517–533 (1997)
Ahn, J. H. & Peacor, D. R. A transmission and analytical electron microscopic study of the smectite to illite transition. Clays Clay Minerals 34, 165–179 (1986)
Nadeau, P. H., Wilson, M. J., McHardy, W. J. & Tait, J. M. The conversion of smectite to illite during diagenesis: evidence from some illitic clays from bentonites and sandstones. Mineral. Mag. 49, 393–400 (1985)
Hower, J., Eslinger, E. V., Hower, M. E. & Perry, E. A. Mechanism of burial metamorphism of argillaceous sediments: Mineralogical and chemical evidence. Geol. Soc. Am. Bull. 87, 725–737 (1976)
Eberl, D. D., Drits, V. A. & Srodon, J. Deducing growth mechanisms for minerals from the shapes of crystal size distributions. Am. J. Sci. 298, 499–533 (1998)
Monkhorst, H. J. & Pack, J. D. Special points for Brillouin-zone integrations. Phys. Rev. B 13, 5188–5192 (1976)
Benincasa, E., Brigatti, M. F., Medici, L. & Poppi, L. K-rich rectorite from kaolinized micaschist of the Sesia-Lanzo Zone, Italy. Clay Minerals 36, 421–433 (2001)
Acknowledgements
This work was supported by the US National Science Foundation.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare that they have no competing financial interests.
Rights and permissions
About this article
Cite this article
Stixrude, L., Peacor, D. First-principles study of illite–smectite and implications for clay mineral systems. Nature 420, 165–168 (2002). https://doi.org/10.1038/nature01155
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nature01155
This article is cited by
-
Adsorption forms of cesium in specific soils based on EXAFS spectroscopic investigations and sequential extraction experiments
Journal of Radioanalytical and Nuclear Chemistry (2023)
-
Molecular dynamics and density functional theory simulations of cesium and strontium adsorption on illite/ smectite
Journal of Radioanalytical and Nuclear Chemistry (2022)
-
Conglomerate Reservoir Pore Evolution Characteristics and Favorable Area Prediction: A Case Study of the Lower Triassic Baikouquan Formation in the Northwest Margin of the Junggar Basin, China
Journal of Earth Science (2021)
-
Formation Mechanism and Numerical Model of Quartz in Fine-Grained Organic-Rich Shales: A Case Study of Wufeng and Longmaxi Formations in Western Hubei Province, South China
Journal of Earth Science (2020)
-
Diagentic features of illite in Upper Triassic Chang-7 tight oil sandstones, Ordos Basin
Geosciences Journal (2019)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.