Abstract
The prevalence of asthma has steadily increased during the last decade, probably as the result of changes in the environment, including reduced microbial exposure during infancy. Accordingly, experimental studies have shown that deliberate infections with live pathogens prevent the development of allergic airway diseases in mice. Bacterial extracts are currently used in children suffering from repeated upper respiratory tract infections. In the present study, we have investigated whether bacterial extracts, commercially available as Broncho-Vaxom (BV), could prevent allergic airway disease in mice. Oral treatment with BV suppressed airway inflammation through interleukin-10 (IL-10)-dependent and MyD88 (myeloid differentiation primary response gene (88))-dependent mechanisms and induced the conversion of FoxP3 (forkhead box P3)− T cells into FoxP3+ regulatory T cells. Furthermore, CD4+ T cells purified from the trachea of BV-treated mice conferred protection against airway inflammation when adoptively transferred into sensitized mice. Therefore, treatment with BV could possibly be a safe and efficient strategy to prevent the development of allergic diseases in children.
Similar content being viewed by others
Introduction
Allergic asthma is a Th2 lymphocyte-mediated inflammatory airway disease. It is characterized by airway eosinophilia, increased mucus production by goblet cells, and structural remodeling of the airway wall, leading to airway obstruction and airway hyperreactivity (AHR) to nonspecific stimuli. Allergy affects 300 million individuals worldwide and its prevalence has dramatically increased in the recent decades, most probably as a result of changes in environmental factors including reduced exposure to microbial antigens during infancy. Reduced risk of atopy has been linked to increased numbers of older siblings, early day-care attendance, agrarian lifestyle, early-life measles, and enteric infections.1, 2 Accordingly, infection with various parasites prevents the development of allergic airway inflammation in mice.3, 4, 5 These observations support the hygiene hypothesis that states that early exposure to pathogens provides protective effects, possibly through the activation of innate immune responses and/or the development of regulatory T cells (Treg).
Along the same lines, an inverse correlation between exposure to food-borne microbes and respiratory allergy has pointed toward a role for the gut and gut-associated lymphoid tissue in shaping the immune response against environmental antigens.6, 7 Furthermore, the intestinal bacterial flora was demonstrated to alter the mechanisms that are responsible for oral tolerance induction in mice.8 For example, although oral administration of ovalbumin (OVA) abrogates both Th1 and Th2 responses in specific pathogen-free mice, only Th1 responses are reduced in germ-free mice. The reconstitution of intestinal flora of germ-free mice with Bifidobacterium infantis, one of the predominant bacteria in the intestinal flora, restored the susceptibility of these Th2 responses to oral tolerance induction. In addition, oral administration of live Lactobacillus reuteri to specific pathogen-free mice prevented allergic airway inflammation via the induction of Treg.9, 10 This latter finding and potential problems arising upon parenteral administration of live microorganisms have resulted in a renewed interest in the possible modulation of the immune response through the oral delivery of bacterial extracts. This procedure is a common practice in Europe to reduce the clinical symptoms, the incidence, the duration, and the severity of disease in patients suffering from recurrent respiratory infections.11, 12 According to the concept of “common mucosal immune system,” presentation of an antigen at one mucosal site leads to the stimulation of an immune response at a distant mucosal site. Similarly, oral treatment with bacterial extracts modulates both systemic and local immune responses.13 However, the mechanisms underlying the immunostimulatory effect of these bacterial extracts via the gastrointestinal route remain unclear.
Broncho-Vaxom (BV) is an endotoxin-low, lyophilized fractionated alkaline extract of the following eight bacterial strains: Haemophilus influenzae, Diplococcus pneumonia, Klebsiella pneumoniae, Klebsiella ozaenae, Staphylococcus aureus, Streptococcus pyogenes, Streptococcus viridans, and Neisseria catarrhalis. It has been widely used in children and adults suffering from repeated upper respiratory tract infections,14, 15, 16 and it efficiently reduces both the frequency and the duration of the infections.12 In the present report, we have investigated the effect of oral BV administration in the development of allergic airway inflammation. To this aim, BALB/c mice were treated with BV orally, and subsequently sensitized and challenged with the Leishmania homolog of receptors for activated c kinase (LACK) antigen17 or with the commonly used OVA antigen.
Results
Oral administration of BV protects mice from allergic airway inflammation
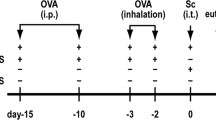
To investigate whether BV could prevent allergic airway inflammation in mice, BALB/c mice were treated daily with BV (25 mg orally) or with the vehicle control for 15 consecutive days. Mice were sensitized with two intraperitoneal (i.p.) injections of the Leishmania major antigen LACK in the Th2 adjuvant aluminum hydroxide and challenged 5 times 10 days later with LACK, or phosphate-buffered saline (PBS) aerosols (Figure 1a). Compared with LACK-sensitized and PBS-challenged mice, LACK-sensitized and -challenged mice treated with the vehicle exhibited the expected increased AHR to the nonspecific stimulus methacholine (Figure 1b), airway eosinophilia (Figure 1c), peribronchial and perivascular cellular infiltration of the lungs (Figure 1d), and mucus hypersecretion (Figure 1d and e). Administration of BV to LACK-sensitized and -challenged mice resulted in reduced AHR (Figure 1b), fourfold less eosinophils (P<0.0001) and twofold less lymphocytes (P=0.017) in the bronchoalveolar lavage fluids (BALFs; Figure 1c). Cellular infiltration of the lungs and mucus hypersecretion were also strongly decreased (Figure 1d, e).
Airway hyperreactivity (AHR), airway inflammation, and mucus secretion are reduced in Broncho-Vaxom (BV)-treated mice. (a) Schematic representation of the experimental procedure. Mice were treated daily with BV or vehicle from day –1 to day 13, sensitized intraperitoneal (i.p.) with LACK/aluminum hydroxide (alum) on days 0 and 7, challenged with daily aerosols of LACK or phosphate-buffered saline (PBS) from days 17 to 21, analyzed for AHR on day 22 and killed on day 23. (b) Mice were analyzed for resistance and compliance by invasive plethysmography in response to increasing doses of methacholine. Data are shown for BV-treated (empty symbols) and vehicle-treated (filled symbols) mice upon challenge with LACK (circles) or PBS (squares). Data are expressed as mean±s.e.m., n=7 mice per group. (c) Bronchoalveolar lavage fluid (BALF) cells were analyzed by fluorescence-activated cell sorting (FACS). Data show the number of eosinophils (E), lymphocytes (L), neutrophils (N), and other cells (O) in BV-treated (empty bars) and vehicle-treated (dark bars) LACK-challenged mice and in vehicle-treated PBS-challenged mice (gray bars). Data are expressed as mean±s.e.m., n=30 mice per group pooled from five experiments. (d) Lung sections were prepared from the indicated mice. Sections were stained with May-Grünwald Giemsa (MGG) or periodic acid Schiff (PAS). Data show representative tenfold magnification microscopic images, n=6 mice per group. (e) Mucus production was quantified in the indicated mice. Data are expressed as mean±s.e.m., n=6 mice per group pooled from two experiments. *P<0.05; **P<0.01; ***P<0.001.
Compared with PBS-challenged animals, vehicle-treated mice challenged with LACK exhibited the expected cardinal features of allergic asthma, including higher numbers of CD4+ cells expressing the Th2-specific marker T1/ST2 in BALF and lungs (Figure 2a), increased levels of interleukin (IL)-4, IL-5, IL-6, IL-10, IL-13, eotaxin (CCL11), and very low to undetectable levels of interferon-γ (IFN-γ) in lungs (Figure 2b), increased serum levels of LACK-specific immunoglobulin (Ig)G1 and IgE (Figure 2c), and similar low levels of LACK-specific IgG2a. In these animals, we found that BV treatment resulted in six- and three-fold less T1/ST2+ CD4+ T cells in BALF and lungs, respectively (Figure 2a), and two- to five-fold less IL-4, IL-5, IL-6, IL-10, IL-13, and eotaxin in lungs (Figure 2b). IFN-γ levels were close to the lower limit of detection and remained similar in the two groups (data not shown). Although BV- and vehicle-treated mice exhibited similar serum levels of LACK-specific IgG1 and IgG2a (Figure 2c), BV-treated mice exhibited reduced serum levels of LACK-specific IgE (Figure 2c). In order to investigate whether BV-induced protection lasted over time, BV-treated mice that have previously undergone an acute episode of asthma were re-challenged with LACK aerosols 3 weeks later. In that case, no protection was observed, demonstrating that BV-induced suppression was short lived (Supplementary Figure S1 online).
Antibody and T-cell responses in Broncho-Vaxom (BV)-treated mice. Mice were treated daily with BV or vehicle (vcl) from day –1 to day 13, sensitized with LACK/aluminum hydroxide (alum) on days 0 and 7, and challenged with daily aerosols of LACK or phosphate-buffered saline (PBS) from days 17 to 21. Mice were analyzed on day 23. (a) Bronchoalveolar lavage fluid (BALF) and lung cells were prepared from individual mice and analyzed by fluorescence-activated cell sorting (FACS) upon staining with anti-CD4 and anti-T1/ST2 monoclonal antibodies (mAbs). Data show the number of T1/ST2+ CD4+ cells in individual mice and are expressed as mean±s.e.m., n=8–12 mice per group pooled from two experiments. (b) Protein extracts were prepared from the lungs of individual mice and analyzed for interleukin (IL)-4, IL-5, IL-6, IL-10, and IL-13 content by cytometric bead array (CBA), and for eotaxin content by enzyme-linked immunosorbent assay (ELISA). Data are shown for individual mice with bars indicating the mean value, n=8–12 per group pooled from two experiments. (c) Levels of LACK-specific IgE, IgG1, and IgG2a in the serum of the indicated mice were measured by ELISA. Data are expressed as mean±s.e.m., n=30 mice per group pooled from five experiments. ***P<0.001; **P<0.01; *P<0.05; NS, nonsignificant.
In order to demonstrate that the observed effects were not model dependent, we investigated whether BV protected mice from allergic airway inflammation induced by a different antigen. To this aim, BALB/c mice were treated with BV or vehicle as previously described, sensitized with OVA, and challenged with OVA or PBS. As observed in LACK-sensitized and -challenged mice, BV-treated OVA-sensitized and -challenged animals exhibited reduced AHR (Supplementary Figure 2a online), reduced eosinophilia in BALF (Supplementary Figure 2B online), lower number of T1/ST2+ CD4+ T cells in lungs (Supplementary Figure 2c online), and reduced amounts of IL-4, IL-5, and IL-13 in lungs when compared with vehicle-treated animals (Supplementary Figure 2d online). Therefore, the protection from allergic airway disease that was induced by BV was not restricted to LACK-induced allergic asthma. BV-induced protection was also observed in C57BL/6 mice and was dose dependent (data not shown, and Supplementary Figure 3 online).
BV-induced protection requires the adaptor MyD88
BV is composed of bacterial extracts. As bacteria express pathogen-associated molecular patterns that bind Toll-like receptors (TLRs), we next sought to investigate whether the ability of BV to protect against allergic airway inflammation required TLR signaling. To this aim, we first used MyD88 (myeloid differentiation primary response gene (88)) knockout (KO) mice that are deficient in the adaptor protein that is used by nearly all TLRs and IL-1R to activate the transcription factor nuclear factor-κB. These mice have been shown to develop similar Th2 responses upon OVA sensitization in aluminum hydroxide, and airway inflammation upon OVA challenge. MyD88 KO mice were treated with BV or vehicle, and sensitized and challenged with LACK. In contrast to what was observed in wild-type (WT) mice, vehicle- and BV-treated MyD88 KO mice exhibited similar numbers of total cells, eosinophils, and lymphocytes in BALF (Figure 3a) and identical levels of IL-4, IL-5, and IL-13 in lungs (Figure 3b). These results suggested that the ability of BV to inhibit allergic airway inflammation was dependent on MyD88. To determine whether MyD88 was required for IL-1 or TLR signaling, we have treated WT mice with the commercialized IL-1R antagonist Kineret. In agreement with data obtained in IL-1R-deficient mice,18 treatment of WT mice with Kineret did not modify AHR and airway inflammation in vehicle-treated mice. Most importantly, Kineret treatment did not prevent BV-induced protection from asthma (Figure 3c, d). Altogether, our results suggest that BV-induced protection requires TLR signaling.
Number and phenotype of bronchoalveolar lavage fluid (BALF) cells and lung cytokine levels in Broncho-Vaxom (BV)-treated MyD88 (myeloid differentiation primary response gene (88)) knockout (KO) mice and in Kineret-treated wild-type (WT) mice. MyD88 KO (a, b) and WT (a–d) mice were treated daily with BV or vehicle (vcl) from day −1 to day 13. WT mice were treated or not with Kineret intraperitoneal (i.p.) daily along with BV (c, d). All mice were sensitized with LACK/aluminum hydroxide (alum) on days 0 and 7, and challenged with daily aerosols of LACK from days 17 to 21. Mice were analyzed on day 23. (a, c) BALF cells were analyzed by fluorescence-activated cell sorting (FACS). Data show the number of eosinophils (E), lymphocytes (L), neutrophils (N), and other cells (O). Data are expressed as mean±s.e.m., n=8 mice per group. (b, d) Protein extracts were prepared from the lungs of individual mice and analyzed for interleukin (IL)-5 and IL-13 content by cytometric bead array (CBA). Data are shown for individual mice with bars indicating the mean, n=6–8 mice per group. A representative experiment is shown (out of two). ***P<0.001; **P<0.01; *P<0.05; NS, nonsignificant.
BV induces an increase in the number of Treg in the trachea
CD4+ Treg prevent the development of allergic asthma in both humans and mice. To monitor any Treg changes upon BV treatment, we used DEREG mice that express a diphtheria toxin receptor-enhanced green fluorescent protein (GFP) fusion protein under the control of the foxp3 gene locus, as reporter mice.19 Naive DEREG mice were treated for 15 days with either BV or vehicle and the presence of GFP+ CD4+ cells was monitored in various lymphoid and nonlymphoid organs. The cellularity in all the analyzed organs remained similar in both BV- and vehicle-treated mice (data not shown). However, although the frequency of GFP+ CD4+ T cells was similar in BV- and vehicle-treated mice in all tested secondary lymphoid organs, including the mesenteric lymph node (LN) and the spleen, BV-treated mice exhibited a twofold increased frequency of GFP+ CD4+ T cells in the trachea when compared with vehicle-treated animals (Figure 4a). A more modest but significant increase in the frequency of GFP+ CD4+ T cells was observed in the vagina and the intestinal lamina propria (LP) upon treatment with BV (Figure 4a). Most of GFP+ CD4+ cells were stained with an antibody reacting to the T cell receptor (TCR) β chain, but not with a αGalCer/CD1d tetramer or an antibody reacting to the TCR γδ receptor, suggesting that they were bona fide CD4+ Treg (Figure 4b). In addition, in both BV- and vehicle-treated mice, all GFP+ CD4+ cells expressed GITR (glucocorticoid-induced tumor necrosis factor receptor), and 30% expressed high levels of CD25 (Figure 4b). The frequency of GFP+ CD4+ cells that expressed CTLA-4 (cytotoxic T-lymphocyte antigen 4) was higher in BV-treated mice when compared with vehicle-treated animals (21 vs. 8%; Figure 4b). In addition, GFP+ Treg from BV-treated mice and vehicle-treated mice were comparably able to inhibit T-cell proliferation (Figure 4c). In agreement with the data obtained in DEREG mice, the administration of BV to WT BALB/c mice resulted in a twofold increase in the frequency of forkhead box P3 (Foxp3)+ CD4+ T cells in the airway mucosa and in a slight but significant increase in the frequency of these cells in the vagina and the LP (Figure 4d). Interestingly, although only 20% of trachea FoxP3+ CD4+ T cells from vehicle-treated mice expressed the chemokine receptor CCR9 (chemokine (C-C motif) receptor 9), this frequency increased to 50–60% in BV-treated animals (Figure 4e). In contrast, BV did not alter the frequency of trachea FoxP3+ CD4+ T cells that expressed the integrins α4β7, αEβ7 (CD103), or the mucosal chemokine receptor CCR10 (Figure 4e). Further analysis showed that 10–15 and 65–70% of CCR9+ Treg were CD25+ and CTLA-4+, respectively (Figure 4f) and that <1.5% of Treg produced IL-5, IFN-γ, and IL-17 (Figure 4g) in both vehicle- and BV-treated mice. In contrast, the frequency of IL-10-secreting cells increased from 13.9±3.6% in vehicle-treated mice to 26.3±3.3% in BV-treated animals (P=0.003). Therefore, when compared with vehicle-treated mice, the trachea and other mucosal tissues of BV-treated animals contained an increased number of IL-10-secreting Treg with suppressive activity.
Frequency and surface phenotype of Treg in Broncho-Vaxom (BV)-treated mice. DEREG (a–c) and wild-type (WT) BALB/c (d, e) mice were treated with BV or vehicle for 15 days. Cells from the indicated organs were analyzed by fluorescence-activated cell sorting (FACS). (a) Frequency of FoxP3+ CD4+ T cells. Data show the frequency of green fluorescent protein (GFP)+ cells among CD4+ lymphocytes in the indicated organs of BV-treated (empty bars) and vehicle-treated (filled bars) mice. (b) Cells were prepared from the trachea of BV- and vehicle- treated mice and analyzed by FACS after staining with antibodies to TCR Vβ, TCRγδ, glucocorticoid-induced tumor necrosis factor receptor (GITR), CTLA-4, and CD25, or with αGalCer/CD1d tetramer. Data show the frequency of GFP+ CD4+ T cells that stained positive for the indicated marker in BV-treated (empty bars) and vehicle-treated (filled bars) mice. (c) GFP+ CD4+ T cells (Treg) were prepared from the trachea of BV-treated (empty circle) and untreated (filled circle) mice and incubated at different ratios with carboxyfluorescein succinimidyl ester (CFSE)-labeled GFP− CD4+ cells (Teff) in the presence of anti-CD3 and anti-CD28 monoclonal antibodies (mAbs). Data show the mean frequency±s.e.m. of GFP− cells that have divided as demonstrated by decreased CFSE fluorescence. (d) Cells were purified from the indicated organs of BV- and vehicle- treated mice and stained with anti-CD4 and anti-FoxP3 mAbs. Data show the frequency of FoxP3+ cells after gating on CD4+ in individual mice. (e) Surface phenotype of FoxP3+ CD4+ T cells. (F) Surface phenotype of CCR9+ FoxP3+ CD4+ T cells. Cells were prepared from the trachea of BV- (empty bars) and vehicle (filled bars)-treated mice and stained with antibodies to CD4, FoxP3, CCR9, α4β7, CD103, CCR10, CD25, and CLTA-4. Data show the frequency of cells that were positive for the indicated marker after gating on total (e) or CCR9+ (f) FoxP3+ CD4+ T cells. (g) Trachea cells from BV- and vehicle-treated mice were stimulated with phorbol myristate acetate (PMA)/ionomycin in the presence of Golgiplug for 4 h and further stained with antibodies to CD4, FoxP3, IL-5, IL-10, IL-17, and interferon-γ (IFN-γ). Data show representative FACS profiles after gating on CD4+ FoxP3+ T cells. Numbers indicate mean±s.e.m. of n=10 mice pooled from two experiments. **P<0.01; *P<0.05; NS, nonsignificant.
BV induces the conversion of Foxp3− into Foxp3+ CD4+ lymphocytes
Although “natural” Foxp3+ Treg arise and mature in the thymus, peripheral conversion of Foxp3− CD4+ T cells into Foxp3+ Treg has been observed in the intestinal LP and gut-associated lymphoid tissues after oral exposure to antigen.20 These inducible Treg are believed to mediate peripheral T-cell tolerance to antigens derived from the diet and the commensal flora. To investigate whether BV promoted the conversion of FoxP3− into FoxP3+ CD4+ cells, GFP− CD4+ T cells were purified from the spleen of naive DEREG mice and injected into recipient mice that were then orally treated with either BV or vehicle. Although GFP+ cells were present at very low frequency or were undetectable in the spleen, peripheral LN, and mesenteric LN, they were readily detected in the trachea of BV-treated but not of vehicle-treated animals (Figure 5a). Therefore, the increased number of FoxP3+ CD4+ cells that were present in the airway mucosa of BV-treated animals resulted, at least in part, from the BV-induced conversion of FoxP3− CD4+ cells into FoxP3+ CD4+ cells.
Conversion and recruitment of FoxP3− CD4+ lymphocytes upon transfer in recipient mice. (a) CD4+ GFP− lymphocytes were purified from naive DEREG mice and injected into syngenic recipients. Mice were then treated daily with either Broncho-Vaxom (BV) or vehicle for 15 days. Cells from the indicated organs were stained with anti-CD4 monoclonal antibodies (mAbs) and analyzed by fluorescence-activated cell sorting (FACS). Data show the frequency of GFP+ cells in BV-treated (empty bars) and vehicle-treated (filled bars) mice after gating on CD4+ T cells. (b) Mice were treated daily with either BV or vehicle for 15 days and injected with FTY720 or phosphate-buffered saline (PBS) intraperitoneal (i.p.) every 3 days. Cells were purified from the indicated organs and analyzed by FACS after staining with anti-CD4 mAbs. Data show the frequency of GFP+ cells after gating on CD4+ T cells in mice treated with FTY720 (empty bars) or PBS (filled bars).
To investigate whether the Treg that accumulated in the airway mucosa of BV-treated mice originated from LN, DEREG mice were injected i.p. every 3 days with the drug FTY720 that blocks the egress into the medullary sinus of LNs and efferent lymphatics,21, 22, 23 and were then treated or not with BV for 15 days. As previously observed, BV induced a 2–3-fold increase in the frequency of GFP+ CD4+ cells in the trachea, but not in the spleen or in the mesenteric LN (Figure 5b). However, this increase was abolished when mice were treated with FTY720, further suggesting that the GFP+ CD4+ cells that accumulated in the trachea in BV-treated mice had migrated from the LN. Furthermore, a slight but significant increase in the frequency of GFP+ CD4+ cells was observed in the mesenteric LN of BV-treated mice upon FTY720 treatment, suggesting that the Treg that were present in the trachea of BV-treated mice derive from the mesenteric LN.
Mesenteric LN of BV-treated mice exhibit an increased number of CD103+ DCs that drive Foxp3+ T-cell differentiation
In both mice and humans, CD103+ dendritic cells (DCs) that originate from the intestinal LP have been shown to imprint CD4+ T cells to specifically express the gut homing receptor CCR9, and to enhance Treg differentiation.24, 25, 26, 27 Therefore, we hypothesized that Treg that accumulated in the trachea as the result of BV treatment might have been generated by the CD103+ MesLN DCs. To test this hypothesis, we measured the number and analyzed the phenotype of CD103+ DCs in the MesLN of BV- and vehicle-treated mice. Although the frequency of CD103− DCs was similar in both groups, the frequency and number of CD103+ DCs were increased in BV-treated mice when compared with vehicle-treated animals (Figure 6a, b). CD103+ DCs from vehicle- and BV-treated mice expressed similar levels of CD40, CD80, CD86, and major histocompatibility complex class II molecules (Figure 6c). To investigate whether CD103+ DCs from BV-treated were capable of promoting the development of Treg, CD103− and CD103+ DCs were purified from the MesLN of BV- and vehicle-treated mice and incubated with LACK peptide and naive CD4+ T cells from LACK-specific TCR transgenic mice. As previously demonstrated with OVA-specific TCR transgenic CD4+ T cells,26 CD103+ DCs but not CD103− DCs from vehicle-treated mice induced the development of FoxP3+ CD4+ T cells (Figure 6d). Similarly, CD103+ DCs but not CD103− DCs from BV-treated mice induced the development of FoxP3+ CD4+ T cells (Figure 6d). Therefore, BV-treated mice exhibited an increased number of CD103+ DCs that have the ability to promote the differentiation of naive T cells into Treg.
Number and phenotype of dendritic cells (DCs) in the mesenteric lymph node (LN) of Broncho-Vaxom (BV)-treated mice. BALB/c mice were treated daily with BV or vehicle for 15 days and mesenteric LN cells were prepared. (a, b) Cells were analyzed by fluorescence-activated cell sorting (FACS) after staining with anti-CD11c and anti-CD103 monoclonal antibodies (mAbs), n=20 mice per group pooled from four experiments. (a) Data show representative FACS profiles after gating on CD11c+ cells. Numbers indicate mean frequencies±s.e.m. (b) Data show mean frequencies±s.e.m. and absolute numbers of CD11c+ CD103+ DCs. (c) Cells were stained with the indicated antibodies and analyzed by FACS. Data show representative FACS profiles after gating on the indicated DC populations. Numbers indicate mean fluorescence intensity (MFI) from one representative experiment out of four. (d) CD103+ DCs were purified by FACS and 1 × 105 cells were incubated with 2 × 105 carboxyfluorescein succinimidyl ester (CFSE)-labeled T cell receptor (TCR) transgenic T cells and 0.2 ng ml–1 of LACK peptide for 4 days. Cells were then analyzed by FACS 3 days later after staining with anti-CD4 and anti-FoxP3 mAbs. Data show representative FACS profiles after gating on CD4+ T cells. Numbers indicate mean of triplicates±s.e.m., n=30 mice per group pooled from three experiments.
BV-induced protection is mediated by a subset of CD4+ T cells expressing CCR9
We next investigated whether the inhibition of allergic asthma induced by BV was mediated by Treg. CD4+ T cells were purified from the trachea of BV- or vehicle-treated mice and injected into LACK-sensitized mice that were then challenged with LACK aerosols. Compared with recipient mice injected with CD4+ T cells from vehicle-treated mice, animals injected with CD4+ T cells from BV-treated mice exhibited decreased AHR (Figure 7a), reduced numbers of eosinophils and lymphocytes in BALF (Figure 7b), reduced numbers of T1/ST2+ CD4+ T cells in BALF and lungs (Figure 7c), and a 50% reduction in the IL-5 and IL-13 levels in lungs (Figure 7d). Therefore, CD4+ T cells from BV-treated mice, but not those from vehicle-treated animals, could protect recipient mice from allergic airway inflammation upon adoptive transfer.
Airway hyperreactivity (AHR), airway inflammation, and cytokine levels in mice injected with CD4+ T cells. BALB/c mice were treated daily with Broncho-Vaxom (BV) or vehicle for 15 days. Cells from the trachea were prepared and depleted or not of CCR9+ cells. Recipient mice were sensitized with LACK and injected or not with the indicated cell population. Mice were challenged 5 days later with LACK aerosols. (a) AHR was measured by noninvasive plethysmography 1 day after the last aerosol. Data show the mean±s.e.m. of 12 mice per group pooled from two representative experiments (out of four) in response to 50 mg ml–1 of methacholine. (b) Bronchoalveolar lavage fluid (BALF0 cells were analyzed 2 days after the last aerosol. Data show the mean number±s.e.m. of eosinophils (E), lymphocytes (L), neutrophils (N), and other cells (O) in the indicated mice, n=8–12 mice per group pooled from two experiments. (c) BALF and lung cells were prepared from individual mice and analyzed by fluorescence-activated cell sorting (FACS) upon staining with anti-CD4 and anti-T1/ST2 monoclonal antibodies (mAbs). Data show the mean number±s.e.m. of T1/ST2+ cells after gating on CD4+ T cells, n=8–12 mice per group pooled from two representative experiments out of four. (d) Lungs from the indicated mice were analyzed for interleukin (IL)-5 and IL-13 content by cytometric bead array (CBA) array. Data are shown for individual mice with bars indicating the mean. ***P<0.001; **P<0.01; *P<0.05; NS, nonsignificant.
We have found that BV treatment induced a selective accumulation of CCR9-expressing Treg in the airway mucosa (Figure 4e). In an attempt to identify the subset of Treg that accounted for the ability of BV to protect from allergic airway disease, CD4+ T cells from the trachea of BV- or vehicle-treated mice were depleted of CCR9-positive cells before injection into LACK-sensitized recipients. Mice were then challenged with LACK aerosols and analyzed for the cardinal features of asthma. In contrast to what was observed with nondepleted CD4+ population, mice injected with CCR9-depleted CD4+ T cells from BV-treated mice and those injected with CCR9-depleted CD4+ T cells from vehicle-treated mice exhibited similar AHR (Figure 7a),similar number of eosinophils in BALF (Figure 7b), as well as similar numbers of T1/ST2+ CD4+ T cells in BALF and lungs (Figure 7c) and comparable levels of IL-5 and IL-13 cytokines in lungs (Figure 7d). These results suggested that the ability of BV to induce protection was mediated by a subset of Treg expressing CCR9.
IL-10 is critical to control airway inflammation but not AHR
As 20–25% of BV-induced Treg secreted IL-10 (Figure 4g), we investigated whether BV-induced protection was dependent on IL-10. To this aim, vehicle- and BV-treated LACK-sensitized mice were treated or not with anti-IL-10R monoclonal antibody (mAb) during aerosol challenges. AHR was not affected by antibody treatment in both groups (Figure 8a). In sharp contrast, although anti-IL-10R mAb treatment did not affect the airway inflammation in vehicle-treated mice, BV-treated mice were not protected and exhibited similar numbers of eosinophils and lymphocytes in BALF (Figure 8b). In addition, upon treatment with anti-IL-10R mAb, the number of Th2 cells in both BALF and lungs and the amounts of cytokines in lungs were similar in both vehicle- and BV-treated mice (Figure 8c, d). Therefore, although AHR was unaltered by IL-10R blockade, the protection conferred by BV against airway inflammation was dependent on IL-10.
Airway hyperreactivity (AHR) and airway inflammation in mice treated with anti-interleukin (IL)-10R monoclonal antibody (mAb). Mice were treated daily with Broncho-Vaxom (BV) or vehicle (vcl) from day −1 to day 13, sensitized with LACK/aluminum hydroxide (alum) on days 0 and 7, challenged with daily aerosols of LACK or phosphate-buffered saline (PBS) from days 17 to 21, and treated or not with anti-IL-10R mAb (0.5 mg per mouse) intraperitoneal (i.p.) every other day during challenges. (a) AHR was measured by noninvasive plethysmography on day 22. Data show the mean±s.e.m. of n=8 mice per group from one representative experiment (out of two). Statistical analyses were performed by comparing vehicle- and BV- treated LACK-challenged mice (**P<0.01; ***P<0.001), or vehicle- and BV- treated LACK-challenged mice injected with anti-IL-10R mAb (#P<0.05; ##P<0.01). (b) Bronchoalveolar lavage fluid (BALF) cells were analyzed 2 days after the last aerosol. Data show the mean number±s.e.m. of eosinophils (E), lymphocytes (L), neutrophils (N), and other cells (O) in the indicated mice, n=8 mice per group from one representative experiment (out of two). (c) BALF and lung cells were prepared from individual mice and analyzed by fluorescence-activated cell sorting (FACS) upon staining with anti-CD4 and anti-T1/ST2 mAbs. Data show the number of T1/ST2+ CD4+ cells in individual mice and are expressed as mean±s.e.m., n=8 mice per group from one representative experiment (out of two). (d) Protein extracts were prepared from the lungs of individual mice and analyzed for IL-5, and IL-13 content by cytometric bead array (CBA). Data are shown for individual mice with bars indicating the mean value, n=8 mice per group from one representative experiment (out of two). **P<0.01; *P<0.05; NS, nonsignificant.
Discussion
In this study we have demonstrated that oral administration of the bacterial lysate BV inhibited AHR, lung inflammation, mucus hypersecretion, airway eosinophilia, and production of antigen-specific IgE in antigen-sensitized and -challenged mice. The number of Th2 cells in BALF and lungs of BV-treated mice was strongly decreased, and lung contents in IL-4, IL-5, IL-6, IL-10, IL-13, and eotaxin were dramatically reduced. In apparent contrast to published results in which BV increased systemic Th1 responses in OVA-immunized mice,28 we found that IFN-γ levels in lungs remained very low and similar in BV-treated and vehicle-treated mice.
BV is an extract made of several strains of G+ and G− pneumotropic bacteria that consists mainly of acidic proteins, peptides, and amino acids, and also traces of minor components such as sugar fatty acids, and detoxified lipopolysaccharides and lipoteichoic acids. Although we have shown that MyD88 but not IL-1 signaling was required in BV-induced protection, our preliminary data using TLR2/4-double KO mice suggest that neither TLR2 nor TLR4 were necessary for BV-induced protection (data not shown). It remains to be determined which TLR is necessary for BV-induced protection against asthma.
Oral administration of BV induced an increase in the frequency and number of FoxP3+ Treg in the intestinal LP, the airway mucosa, and to a smaller extent in the vagina, but not in secondary lymphoid organs. Our data are in agreement with the concept of the common mucosal immune system, in which the presentation of an antigen at one mucosal site may lead to stimulation of an immune response at a distant mucosal tissue. Transcutaneous, oral, and nasal immunizations can induce immune responses at the gastrointestinal, reproductive, and respiratory surfaces.29, 30, 31, 32 This could be explained by the selective expression of chemokine receptors or integrins by effector cells. For example, nasal immunization induces the expression of high levels of CCR10 and α4β1 integrin by IgA-producing plasma cells, allowing them to migrate to the respiratory and genitourinary tracts that express their corresponding ligands, CCL28 (chemokine (C-C motif) ligand 28) and VCAM1 (vascular cell adhesion molecule-1).33, 34 In contrast, orally induced IgA-producing B cells express CCR9 and CCR10, as well as α4β7 and α4β1 integrins, and hence they migrate to sites such as the small intestine and the airways, which express CCL25 and CCL28 together with MADCAM1 (mucosal vascular addressin cell adhesion molecule 1) and/or VCAM1.34
One important result in this study is the demonstration that a bacterial extract given orally leads to a specific increase in Treg in the respiratory tract. Interestingly, Strickland et al.35 showed that prevention of T-cell activation and AHR during chronic allergen exposure of sensitized rats was mediated by Treg localized in the airway mucosa. The location of these Treg is critical to inhibit memory T helper cell-mediated upregulation of airway mucosal DCs, and their subsequent migration to the lung-draining LN.
As Treg in the trachea of BV-treated mice expressed high levels of CCR9, they were likely to be of intestinal origin, as only DCs from mesenteric LN and Peyer's patches have been shown so far to imprint a CCR9 phenotype on lymphocytes. Initial studies have demonstrated that adoptively transferred TCR transgenic CD4+ and CD8+ T cells primed in the mesenteric, but not cutaneous LNs or spleen, expressed high levels of the α4β7 integrin and CCR9.36 Subsequently, it was shown that T cells primed in vitro with antigen-pulsed mesenteric LN or Peyer's patch DCs, but not with DCs from the spleen or peripheral LN, expressed CCR9 and high levels of α4β7.37, 38 We have shown that FTY720 administration prevented Treg from accumulating in the trachea as the result of BV treatment and resulted in an increase in the number of Treg in the mesenteric LN. Altogether, these results suggest that BV promotes the development of Treg in the mesenteric LN and that these cells have the ability to relocalize in the airways. It remains to be determined whether CCR9 is required for their homing to the trachea. However, although the lungs of asthmatic patients express high levels of CCL25 that promotes the recruitment of the CCR9+ Vα24 invariant natural killer T cells,39 the lungs of naive mice express very low levels of CCL25,36 making unlikely the involvement of CCR9 in the homing of Treg into the trachea.
Mice injected with GFP− CD4+ T cells from DEREG mice and further fed with BV exhibited GFP+ CD4+ T cells in trachea, demonstrating that BV treatment induces the conversion of FoxP3− CD4+ T cells into FoxP3+ CD4+ T cells. Although it is still unclear which cells induce the conversion of FoxP3− into FoxP3+ CD4+ T cells, we found that the mesenteric LN of BV-treated animals contained twofold more CD103+ DCs than those from vehicle-treated mice. As previously reported for CD103+ DCs from naive WT mice, these DCs were capable of inducing FoxP3+ CD4+ T cells. Therefore, it is very likely that the increased number of CD103+ DCs induced by BV is responsible for the increased numbers of Treg that accumulated in the trachea as the result of BV treatment.
The role of Treg in BV-induced protection was demonstrated by the transfer of CD4+ T cells purified from the airway mucosa of BV-treated mice into sensitized mice. Although the transfer of total CD4+ T cells into sensitized mice from BV-treated mice led to protection, injection of CCR9-depleted CD4+ T cells did not, demonstrating that a particular subset of CCR9-expressing Treg has the ability to suppress allergic airway inflammation. Interestingly, it has been shown in healthy individuals that circulating CCR9+ CD4+ T cells with a memory phenotype exhibit characteristics of mucosa T cells and that some of them are T regulatory type 1-like cells.41 In agreement with this finding, we found that >20% of trachea Treg in BV-treated mice produced IL-10. Most importantly, we showed that BV-induced protection from airway inflammation was dependent on IL-10. This cytokine has been shown to be essential for the suppression of airway inflammation in several mouse models as well as in immunotherapy in humans.42, 43, 44, 45, 46 Although it remains to be confirmed whether BV-induced protection is dependent on IL-10 produced by Treg, it is noteworthy that the deletion of IL-10 in Treg promoted allergic airway inflammation.47
Altogether, our results show that oral treatment with BV suppresses the cardinal features of asthma via the induction of IL-10-producing CCR9+ Treg.
This study provides evidence for the use of BV as a safe, ready, and efficient treatment in children to prevent the development of allergic diseases.
Methods
Animals. BALB/c ByJ mice (6 weeks old) were purchased from the Centre d’Elevage Janvier, France. LACK TCR transgenic mice (WT15 RAG-1 KO) and MyD88 KO mice48 on the BALB/c background, and DEREG mice19 on the C57BL/6 background, were bred in our animal facility at the Institut de Pharmacologie Moléculaire et Cellulaire (Valbonne, France). All experimental protocols were approved by the local animal ethics committee.
Reagents. BV was made of extracts from H. influenzae, D. pneumoniae, K. pneumoniae, K. ozaenae, S. aureus, S. pyogenes, S. viridans, and N. catarrhalis. LACK recombinant protein was produced in Escherichia coli and purified as described49 and detoxified using Endotrap (Profos, Regensburg, Germany). Lipopolysaccharide contents in LACK protein samples were <5 ng mg–1 as determined using a Limulus Amoebocyte Lysate (LAL) assay (Pierce; Perbio Science, Brebieres, France). T1/ST2 mAbs were purchased from MD Biosciences (Zurich, Switzerland). Monoclonal antibodies to CD4, CD25, TCR Vβ, CD103, CTLA-4, GITR, and TCRγδ were purchased from BD Biosciences (Le Pont de Claix, France). Antibodies to α4β7, CCR9, and CCR10 were purchased from eBiosciences (Paris, France). CD1d tetramers and PBS-57, an analog of α-galactosylceramide, were obtained from the NIH tetramer core facility (Bethesda, MD). Anti-IL-10R mAb (1B1.2 clone) is a kind gift of DNAX Research Institute (Palo Alto, CA). LACK156−173 peptide was purchased from Mimotopes (Victoria, Australia).
Experimental asthma and treatments. Mice were sensitized by two i.p. injections of 10 μg of LACK (or OVA (Profos)) in 2 mg of aluminum hydroxide (Pierce) at days 0 and 7. At days 17–21, sensitized mice were exposed to LACK (1.5 mg ml–1) or OVA (2 mg ml–1) aerosols for 20 min using an ultrasonic nebulizer (AeroNeb; Emka Technologies, Paris, France). BV-treated and vehicle-treated mice were every day administered 25 mg of BV in 500 μl of water or 500 μl of water only (vehicle), respectively. Treatment started at day −1 and stopped at day 14. In some experiments, mice were treated i.p. with the IL-1R antagonist Kineret (Biovitrum AB, Stockholm, Sweden) at 150 mg kg–1 every day from day −1 to day 16. In the indicated experiments, mice were treated i.p. with FTY720 (Cayman, Interchim, Montigny, France) at 1 mg kg–1 every 3 days for 15 days. Neutralization of IL-10R was performed by i.p. injection of 0.5 mg of anti-IL-10R mAbs every other day starting at day 16 until day 20.
Antibody titers. Mice were bled one day after the last aerosol. LACK- and OVA-specific IgG1, IgG2a, and IgE were measured by enzyme-linked immunosorbent assay using specific mAbs purchased from BD Biosciences as previously described.50
Number and phenotype of BALF cells. Mice were bled and a canula was inserted into the trachea. Lungs were washed three times with 1 ml of PBS. For differential BAL cell counts, cells were stained with mAb anti-CCR3 (R&D), anti-Gr1, anti-CD3, and anti-CD19 mAbs (BD Biosciences) and analyzed by fluorescence-activated cell sorting (FACS) using a FACScalibur flow cytometer and Cellquest software. Eosinophils were defined as CCR3+ CD3−CD19−, neutrophils as Gr-1high CCR3− CD3−CD19−, lymphocytes as CD3+CD19+, and alveolar macrophages as large autofluorescent cells.
Airway hyperreactivity. One day after the last LACK aerosol challenge, AHR was measured by either whole-body or invasive plethysmography (Emka Technologies) in response to inhaled methacholine (Sigma, Lyon, France) as described previously.51 For dynamic lung resistance and compliance, measurements were performed using a Flexivent apparatus (SCIREQ; Emka Technologies). Mice were anesthetized (5 ml per kg body weight (ml kg–1) of 10% medetomidine (Dormitor; Pfizer, Paris, France) and 10% ketamine (Imalgene; Merial, Lyon, France)), tracheotomized, paralyzed (5 ml kg–1 pancuronium bromide 1% (Pavulon; Organon, Puteaux, France)), and immediately intubated with an 18-gauge catheter, followed by mechanical ventilation. Respiratory frequency was set at 150 breaths per min with a tidal volume of 0.2 ml, and a positive-end expiratory pressure of 2 ml H2O was applied. Increasing concentrations of methacholine (0–50 mg ml–1) were administered at the rate of 20 puffs per 10 s, with each puff of aerosol delivery lasting 10 ms, via a nebulizer aerosol system with a 2.5–4 μm aerosol particle size generated by a nebulizer head (Aeroneb; Aerogen, Emka Technologies). Baseline resistance was restored before administering subsequent doses of methacholine.
Histology. Lung samples were harvested and treated as described previously.52 Sections (4 μm) were performed and stained with May Grünwald Giemsa for morphology or with periodic acid Schiff for detection of mucopolysaccharide accumulation as described previously.52 Mucus index was calculated, and analyzed using Image J software (NIH, Bethesda, MD) as follows: (average intensity of epithelial periodic acid Schiff staining) × (area of epithelial periodic acid Schiff staining) × (total area of conducting airways).
Cytokine measurement. Lung samples were homogenized in calcium- and magnesium-free Hanks balanced salt solution. Multiplex IL-4, IL-5, IL-6, IL-10, IL-13, and IFN-γ analyses were performed with cytometric bead array using FACS array (BD Bisociences). Eotaxin levels were measured by enzyme-linked immunosorbent assay using its pairs from BD Biosciences.
Cell preparations. LN, spleen, trachea, lung, or vagina were cut to small pieces in Hanks balanced salt solution containing 400 U type I collagenase and 1 mg ml–1 DNAse I and digested for 30 min at 37 °C. Cells were strained through a 70-μm cell strainer, and erythrocytes were lysed with ACK lysis buffer. Intestinal LP cells were obtained after digestion of the small intestine in RPMI containing fetal calf serum 5%, 5 mM EDTA, and 2 mM dithiothreitol as described previously.53 Briefly, the small intestines were minced after the removal of the Peyer's patches. The pieces were incubated under agitation in the digestion buffer for 30 min and filtered. The retained fraction was further digested in RPMI containing fetal calf serum 5%, 400 U of type I collagenase, and 0.1 mg ml–1 of DNase I, and filtered after a 30-min incubation period. Both flow through were centrifuged and stained for cytometry analysis.
Suppression assay. GFP+CD4+ cells from DEREG mice cells were enriched by positive selection using CD4-coated magnetic beads (MACS, Miltenyi, Paris, France) and sorted from the trachea and the mesenteric LN of vehicle- and BV-treated BALB/c and DEREG mice, respectively, using a FACSVantage following staining with anti-CD4 mAb. Cells were incubated at different ratios with carboxyfluorescein succinimidyl ester-labeled GFP−CD4+ effector cells purified from the spleen of syngenic mice, in the presence of anti-CD3 (10 μg ml–1) and anti-CD28 (1 μg ml–1) for 72 h. Effector cell proliferation was assessed by monitoring the decrease in carboxyfluorescein succinimidyl ester fluorescence using a FACScalibur.
Treg induction by MesLN DCs. MesLN cells were prepared as described above. CD11c+ cells were enriched by positive selection using magnetic beads (MACS, Miltenyi) and further purified after staining with antibodies to CD11c and CD103 using a cell sorter (Vantage SE; BD Biosciences). CD103+CD11c+ and CD103−CD11c+ populations were pure at 95%. 1 × 105 CD103+ CD11c+ or CD103−CD11c+ DCs were incubated with 2 × 105 of carboxyfluorescein succinimidyl ester-labeled CD4+ TCR transgenic WT15 cells in the presence of 0.2 ng ml–1 of LACK peptide. Cells were washed after 4 days and analyzed by FACS 3 days later following staining with anti-FoxP3 and anti-CD4 mAbs.
Transfer experiments. Cells were prepared from the trachea or the MesLN of BV- or vehicle-treated mice as described above, and CD4+ T cells were enriched by positive selection using magnetic beads (MACS, Miltenyi), and further purified using a cell sorter after staining with anti-CD3 and anti-CD4 mAbs. CD4 purity was >95%. In some experiments, enriched CD4+ T cells were stained with antibodies to CCR9, CD4, and CD3, and CCR9− CD4+ T cells were sorted by FACS. Sorted cells were then injected intravenously into BALB/c mice (6 × 106 cells per mouse).
Statistic analysis. Analysis of variance for repeated measures was used to determine the levels of difference between groups of mice for plethysmography measurements. Comparisons for all pairs were performed by unpaired two-tailed Student's t-test. Significance levels were set at a P-value of 0.05.
References
Strachan, D.P. Family size, infection and atopy: the first decade of the “hygiene hypothesis”. Thorax 55 (Suppl 1), S2–S10 (2000).
von Mutius, E. Environmental factors influencing the development and progression of pediatric asthma. J. Allergy Clin. Immunol. 109, S525–S532 (2002).
Kitagaki, K. et al. Intestinal helminths protect in a murine model of asthma. J. Immunol. 177, 1628–1635 (2006).
Smits, H.H. et al. Protective effect of Schistosoma mansoni infection on allergic airway inflammation depends on the intensity and chronicity of infection. J. Allergy Clin. Immunol. 120, 932–940 (2007).
Dittrich, A.M. et al. Helminth infection with litomosoides sigmodontis induces regulatory T cells and inhibits allergic sensitization, airway inflammation, and hyperreactivity in a murine asthma model. J. Immunol. 180, 1792–1799 (2008).
Matricardi, P.M. et al. Exposure to foodborne and orofecal microbes versus airborne viruses in relation to atopy and allergic asthma: epidemiological study. Br. Med. J. 320, 412–417 (2000).
Pelosi, U. et al. The inverse association of salmonellosis in infancy with allergic rhinoconjunctivitis and asthma at school-age: a longitudinal study. Allergy 60, 626–630 (2005).
Sudo, N. et al. The requirement of intestinal bacterial flora for the development of an IgE production system fully susceptible to oral tolerance induction. J. Immunol. 159, 1739–1745 (1997).
Forsythe, P., Inman, M.D. & Bienenstock, J. Oral treatment with live Lactobacillus reuteri inhibits the allergic airway response in mice. Am. J. Respir. Crit Care. Med. 175, 561–569 (2007).
Karimi, K., Inman, M.D., Bienenstock, J. & Forsythe, P. Lactobacillus reuteriinduced regulatory T cells protect against an allergic airway response in mice. Am. J. Respir. Crit. Care Med. 179, 186–193 (2009).
Bousquet, J. & Fiocchi, A. Prevention of recurrent respiratory tract infections in children using a ribosomal immunotherapeutic agent: a clinical review. Paediatr. Drugs 8, 235–243 (2006).
Steurer-Stey, C., Lagler, L., Straub, D.A., Steurer, J. & Bachmann, L.M. Oral purified bacterial extracts in acute respiratory tract infections in childhood: a systematic quantitative review. Eur. J. Pediatr. 166, 365–376 (2007).
Bowman, L.M. & Holt, P.G. Selective enhancement of systemic Th1 immunity in immunologically immature rats with an orally administered bacterial extract. Infect. Immun. 69, 3719–3727 (2001).
Gutierrez-Tarango, M.D. & Berber, A. Safety and efficacy of two courses of OM-85 BV in the prevention of respiratory tract infections in children during 12 months. Chest 119, 1742–1748 (2001).
Schaad, U.B., Mutterlein, R. & Goffin, H. Immunostimulation with OM-85 in children with recurrent infections of the upper respiratory tract: a double-blind, placebo-controlled multicenter study. Chest 122, 2042–2049 (2002).
Del-Rio-Navarro, B.E. et al. Use of OM-85 BV in children suffering from recurrent respiratory tract infections and subnormal IgG subclass levels. Allergol. Immunopathol. (Madr) 31, 7–13 (2003).
Julia, V. et al. A restricted subset of dendritic cells captures airborne antigens and remains able to activate specific T cells long after antigen exposure. Immunity 16 Revised ID MI-10-001 Page 23 271–283 (2002).
Schmitz, N., Kurrer, M. & Kopf, M. The IL-1 receptor 1 is critical for Th2 cell type airway immune responses in a mild but not in a more severe asthma model. Eur. J. Immunol. 33, 991–1000 (2003).
Lahl, K. et al. Selective depletion of Foxp3+ regulatory T cells induces a scurfy-like disease. J. Exp. Med. 204, 57–63 (2007).
Cook, D.N. et al. Generation and analysis of mice lacking the chemokine fractalkine. Mol. Cell Biol. 21, 3159–3165 (2001).
Matloubian, M. et al. Lymphocyte egress from thymus and peripheral lymphoid organs is dependent on S1P receptor 1. Nature 427, 355–360 (2004).
Graler, M.H. & Goetzl, E.J. The immunosuppressant FTY720 down-regulates sphingosine 1-phosphate G-protein-coupled receptors. FASEB J. 18, 551–553 (2004).
Wei, S.H. et al. Sphingosine 1-phosphate type 1 receptor agonism inhibits transendothelial migration of medullary T cells to lymphatic sinuses. Nat. Immunol. 6, 1228–1235 (2005).
Jaensson, E. et al. Small intestinal CD103+ dendritic cells display unique functional properties that are conserved between mice and humans. J. Exp. Med. 205, 2139–2149 (2008).
Johansson-Lindbom, B. et al. Functional specialization of gut CD103+ dendritic cells in the regulation of tissue-selective T cell homing. J. Exp. Med. 202, 1063–1073 (2005).
Coombes, J.L. et al. A functionally specialized population of mucosal CD103+ DCs induces Foxp3+ regulatory T cells via a TGF-beta and retinoic acid-dependent mechanism. J. Exp. Med. 204, 1757–1764 (2007).
Kang, S.G., Wang, C., Matsumoto, S. & Kim, C.H. High and low vitamin a therapies induce distinct FoxP3(+) T-Cell subsets and effectively control intestinal inflammation. Gastroenterology 137, 1391–1402 (2009).
Huber, M., Mossmann, H. & Bessler, W.G. Th1-orientated immunological properties of the bacterial extract OM-85-BV. Eur. J. Med. Res. 10, 209–217 (2005).
Berry, L.J. et al. Transcutaneous immunization with combined cholera toxin and CpG adjuvant protects against Chlamydia muridarum genital tract infection. Infect. Immun. 72, 1019–1028 (2004).
Skelding, K.A. et al. Comparison of intranasal and transcutaneous immunization for induction of protective immunity against Chlamydia muridarum respiratory tract infection. Vaccine 24, 355–366 (2006).
Gockel, C.M., Bao, S. & Beagley, K.W. Transcutaneous immunization induces mucosal and systemic immunity: a potent method for targeting immunity to the female reproductive tract. Mol. Immunol. 37, 537–544 (2000).
Yanagita, M. et al. Nasopharyngeal-associated lymphoreticular tissue (NALT) immunity: fimbriae-specific Th1 and Th2 cell-regulated IgA responses for the inhibition of bacterial attachment to epithelial cells and subsequent inflammatory cytokine production. J. Immunol. 162, 3559–3565 (1999).
Lazarus, N.H. et al. A common mucosal chemokine (mucosae-associated epithelial chemokine/CCL28) selectively attracts IgA plasmablasts. J. Immunol. 170, 3799–3805 (2003).
Kunkel, E.J. et al. CCR10 expression is a common feature of circulating and mucosal epithelial tissue IgA Ab-secreting cells. J. Clin. Invest. 111, 1001–1010 (2003).
Strickland, D.H. et al. Reversal of airway hyperresponsiveness by induction of airway mucosal CD4+CD25+ regulatory T cells. J. Exp. Med. 203, 2649–2660 (2006).
Svensson, M. et al. CCL25 mediates the localization of recently activated CD8alphabeta(+) lymphocytes to the small-intestinal mucosa. J. Clin. Invest. 110 1113-Revised ID MI-10-001 Page 24 21 (2002).
Johansson-Lindbom, B. et al. Selective generation of gut tropic T cells in gut associated lymphoid tissue (GALT): requirement for GALT dendritic cells and adjuvant. J. Exp. Med. 198, 963–969 (2003).
Mora, J.R. et al. Reciprocal and dynamic control of CD8 T cell homing by dendritic cells from skin- and gut-associated lymphoid tissues. J. Exp. Med. 201, 303–316 (2005).
Sen, Y. et al. V alpha 24-invariant NKT cells from patients with allergic asthma express CCR9 at high frequency and induce Th2 bias of CD3+ T cells upon CD226 engagement. J. Immunol. 175, 4914–4926 (2005).
Denning, T.L., Wang, Y.C., Patel, S.R., Williams, I.R. & Pulendran, B. Lamina propria macrophages and dendritic cells differentially induce regulatory and interleukin 17-producing T cell responses. Nat. Immunol. 8, 1086–1094 (2007).
Papadakis, K.A. et al. CC chemokine receptor 9 expression defines a subset of peripheral blood lymphocytes with mucosal T cell phenotype and Th1 or T-regulatory 1 cytokine profile. J. Immunol. 171, 159–165 (2003).
Campbell, J.D. et al. Peptide immunotherapy in allergic asthma generates IL-10- dependent immunological tolerance associated with linked epitope suppression. J. Exp. Med. 206, 1535–1547 (2009).
Kearley, J., Robinson, D.S. & Lloyd, C.M. CD4+CD25+ regulatory T cells reverse established allergic airway inflammation and prevent airway remodeling. J. Allergy Clin. Immunol. 122, 617–624 e6 (2008).
Leech, M.D., Benson, R.A., De Vries, A., Fitch, P.M. & Howie, S.E. Resolution of Der p1-induced allergic airway inflammation is dependent on CD4+CD25+Foxp3+ regulatory cells. J. Immunol. 179, 7050–7058 (2007).
Joetham, A. et al. Naturally occurring lung CD4(+)CD25(+) T cell regulation of airway allergic responses depends on IL-10 induction of TGF-beta. J. Immunol. 178, 1433–1442 (2007).
Till, S.J., Francis, J.N., Nouri-Aria, K. & Durham, S.R. Mechanisms of immunotherapy. J. Allergy Clin. Immunol. 113, 1025–1034. quiz 1035 (2004).
Rubtsov, Y.P. et al. Regulatory T cell-derived interleukin-10 limits inflammation at environmental interfaces. Immunity 28, 546–558 (2008).
Kawai, T., Adachi, O., Ogawa, T., Takeda, K. & Akira, S. Unresponsiveness of MyD88-deficient mice to endotoxin. Immunity 11, 115–122 (1999).
Mougneau, E. et al. Expression cloning of a Leishmania major protective T cell antigen. Science 268, 563–566 (1995).
Verhasselt, V. et al. Breast milk-mediated transfer of an antigen induces tolerance and protection from allergic asthma. Nat. Med. 14, 170–175 (2008).
Hamelmann, E. et al. Noninvasive measurement of airway responsiveness in allergic mice using barometric plethysmography. Am. J. Respir Crit. Care Med. 156, 766–775 (1997).
Honda, K., Marquillies, P., Capron, M. & Dombrowicz, D. Peroxisome proliferatoractivated receptor gamma is expressed in airways and inhibits features of airway remodeling in a mouse asthma model. J. Allergy Clin. Immunol. 113, 882–888 (2004).
Weigmann, B. et al. Isolation and subsequent analysis of murine lamina propria mononuclear cells from colonic tissue. Nat. Protoc. 2, 2307–2311 (2007).
Acknowledgements
We thank Frank Aguila for his help with figures, and Nicolas Guy, Véronique Thieffin, and Alain Barbot for their excellent animal care. This work was supported in part by grants from the Fondation pour la Recherche Médicale (to V.J.) and by OM Pharma, Geneva, Switzerland. S.N. was supported by a fellowship from the Ministère de l’Enseignement Supérieur, de la Recherche et de la Technologie.
Author contributions: V.J. conceived the experiments and discussed data with N.G., D.D., and C.C. V.J. and N.G. prepared the manuscript. S.N. performed all the experiments with the technical assistance of G.C. and A.L. A.K. and S.F. performed the invasive plethysmography experiments and histochemical analysis. J.C. performed cell sorting experiments. T.S. provided the DEREG mice.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declared no conflict of interest.
Additional information
SUPPLEMENTARY MATERIAL is linked to the online version of the paper
Supplementary information
Rights and permissions
About this article
Cite this article
Navarro, S., Cossalter, G., Chiavaroli, C. et al. The oral administration of bacterial extracts prevents asthma via the recruitment of regulatory T cells to the airways. Mucosal Immunol 4, 53–65 (2011). https://doi.org/10.1038/mi.2010.51
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/mi.2010.51
This article is cited by
-
Impact of Polyvalent Mechanical Bacterial Lysate on lymphocyte number and activity in asthmatic children: a randomized controlled trial
Allergy, Asthma & Clinical Immunology (2021)
-
Viral infections and wheezing–asthma inception in childhood: is there a role for immunomodulation by oral bacterial lysates?
Clinical and Translational Allergy (2020)
-
Evidence that a primary anti-viral stimulation of the immune response by OM-85 reduces susceptibility to a secondary respiratory bacterial infection in mice
Italian Journal of Pediatrics (2018)
-
OM-85 is an immunomodulator of interferon-β production and inflammasome activity
Scientific Reports (2017)
-
The Immunotherapeutic Role of Bacterial Lysates in a Mouse Model of Asthma
Lung (2017)