Abstract
Individual variation in drug response is considered to have multiple origins arising from interactions among susceptible genes and environmental factors. A total of 726 hypertensive patients who took benazepril 10 mg once a day for 15 days and their families from Huoqiu county of Anhui Province, China, were used to study the association between D919G polymorphism of methionine synthase (MTR) gene and the antihypertensive effect of this angiotensin-converting enzyme inhibitor. Compared to the 919D allele, both population-based (P=0.010) and family-based association tests (additive model P=0.018, dominant model P=0.025) demonstrated that the 919G allele was associated with a significantly less diastolic blood pressure reduction. No significant association was found between the extent of systolic blood pressure reduction and benazepril therapy. Our finding suggests that the D919G polymorphism of the MTR gene may be a useful genetic marker to predict the antihypertensive effect of short-term benazepril therapy in hypertensive patients of Anhui Province, China.
Similar content being viewed by others
Introduction
Essential hypertension is a major independent risk factor of cardiovascular and cerebrovascular diseases such as myocardial infarction and stroke. In order to achieve a maximal reduction in the risk of morbidity and mortality, hypertensive patients usually need constant drug therapy to get their blood pressure well controlled. Benazepril, a long-acting angiotensin-converting enzyme (ACE) inhibitor, has been widely used in the treatment of hypertension. However, there is a significant individual variation in response to this drug. It has been estimated that an oral administration of 10 mg benazepril once a day is only effective in about 50% patients with mild-to-moderate hypertension (Balfour and Goa 1991). The causes underlying such variations remain to be elucidated, and interactions among multiple genetic and environmental factors could be involved.
Methionine synthase (MTR), a key enzyme in homocysteine (Hcy) metabolism, catalyzes the remethylation of Hcy to form methionine. An enhanced function of MTR will lead to a decreased plasma Hcy level. An A-to-G substitution of the MTR gene, which results in the conversion of an aspartic acid residue to a glycine residue at codon 919 (D919G), was found to play a role in regulating plasma Hcy levels. A higher level of Hcy was shown in subjects with the 919D allele than in those with the 919G allele (Chen et al. 2001; Harmon et al. 1999). As a risk factor of myocardial infarction (Stampfer et al. 1992) and stroke (Perry et al. 1995), Hcy was also found to be associated with blood pressure levels in several studies (Fiorina et al. 1998; Lim and Cassano 2002; Nygard et al. 1995; Sutton-Tyrrell et al. 1997). Since shared factors can underlie both disease pathogenesis and its associated drug’s efficacy, this naturally leads us to the hypothesis whether the D919G polymorphism of the MTR gene could explain part of the individual variation in response to benazepril.
Recently, we conducted a large-scale, genetic epidemiologic study in Chinese hypertensive patients and their families to investigate the association between the D919G polymorphism of the MTR gene and the antihypertensive effect of benazepril using both population-based and family-based association tests.
Materials and methods
Study population
The study was conducted in Huoqiu and Yuexi Counties of Anhui Province, China, from July 2000 to May 2001, and the study design has been described previously (Li et al. 2004). In brief, hypertensive patients were enrolled into the study using the following criteria: The study subject (1) was 21–65 years old, (2) resided in Huoqiu or Yuexi County for at least 2 years, (3) took no antihypertensive or other medications 2 weeks prior to this study, (4) had a baseline systolic blood pressure (SBP) of 140–200 mmHg or had a baseline diastolic blood pressure (DBP) of 90–120 mmHg, (5) had a body mass index (BMI) ≤33 kg/m2, (6) was not diagnosed with secondary hypertension, and (7) was not diagnosed with severe liver dysfunction, severe kidney dysfunction, myocardial infarction, or other diseases that may either interfere with the drug efficacy or the patient’s safety. Patients’ family members (parents and/or siblings) were also invited to our study. All participants signed informed consents prior to their inclusion. The study procedure was approved by the Institutional Review Boards of both Anhui Medical University and Harvard School of Public Health.
Study procedure
Patients came to the study center around 8:00A.M. with an overnight fasting on the first day and had their baseline blood pressure measured. After a detailed examination and questionnaire were administered, they began to take benazepril (Ciba-Geigy, Beijing, China) at a dose of 10 mg around 9:30 A.M. once a day for a consecutive period of 15 days. Patients took no other medication that may influence blood pressure during this period. They visited our study center every 3 days both to get examined and to receive benazepril for the next 3 days. Patients were also asked to keep their diaries to collect the information on drug responses, medical history, and lifestyle changes in the follow-up period. On the 16th day, patients returned to the study center around 8:00 A.M. and had their end-point blood pressure measured 24 h after the last benazepril tablet was taken. Blood pressure measurement was performed following a standard protocol. In brief, supine blood pressure was measured on patient’s arm by mercury sphygmomanometer after he or she rested for 60 min in supine position. SBP was defined as Korotkoff phase I (appearance of sound), and DBP was defined as Korotkoff phase V (disappearance of sound), respectively. The average of triplicate measurements was used in our analysis.Participants’ blood samples were also collected on the first day for genotyping and biochemical measurements.
Genotyping
The D919G polymorphism of the MTR gene was genotyped using a polymerase chain reaction-restriction fragment length polymorphism (PCR-RFLP) method. PCR was carried out on a PTC-225 thermal cycler (MJ Research Inc., Waltham, MA, USA) using the following primers: 5′-GAACTAGAAGACAGAAATTCTCTA-3′ and 5′-CATGGAAGAATATCAAGATATTAGA-3′. The 10-μl PCR reaction mix contained 30 ng genomic DNA, 1× reaction PCR buffer, 2.5 mM MgCl2, 200 μM of each deoxynucleotide triphosphate, 200 nM of each primer, and 0.25 U of Taq DNA polymerase (Applied Biosystems, Foster City, CA, USA). The initial denaturation was performed at 94°C for 3 min, followed by 14 touchdown cycles of 94°C for 30 s, 61–54°C for 30 s (a decrease of 0.5°C/cycle), 72°C for 30 s, and then followed by 30 cycles of 94°C for 30 s, 54°C for 30 s, and 72°C for 30 s, and a final extension at 72°C for 7 min. Each PCR product was digested by 3 U of HaeIII (New England Biolabs, Beverly, MA, USA) at 37°C for 15 h. Digested PCR products were then loaded onto 3% agarose gel, stained with ethidium bromide, and visualized under UV illumination. The 919D allele resulted in a single band of 189 bp while the 919G allele resulted in two bands of 30 and 159 bp.
Statistical analysis
Data were analyzed using the POLYMORPHISM 2.2 software (Niu et al. 2001), the SAS 8.02 software package (SAS Institute, NC, USA) and the FBAT software (http://www.biostat.harvard.edu/~fbat/default.html). Hardy–Weinberg equilibrium (HWE), heterozygosity (HET) and polymorphism information content (PIC) of the MTR D919G polymorphism were assessed using genotype data of patient’s parents. Blood pressure reduction was calculated by subtracting the end-point blood pressure on the 16th day from the blood pressure at baseline. Multivariate linear regression analysis was performed to estimate the association between different genotypes and blood pressure reduction by adjusting for major demographic and environmental factors including age, gender, height, weight, BMI, smoking status, alcohhol consumption, education, occupation, and baseline blood pressure. Because several enrolled patients came from one family, the generalized estimating equation (GEE) model was used to adjust for intrafamilial correlation. Since a major limitation in population-based association test is the potential confounding by population admixture, we also performed FBAT under dominant, recessive, and genetic models. Residuals of blood pressure reduction calculated from multivariate regression models were used as phenotypic traits of interest in FBAT. P<0.05 was considered statistically significant.
Results
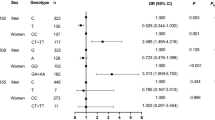
A total of 726 Huoqiu hypertensive patients with complete genotype and phenotype data and their family relatives were finally included in the analysis. The frequencies of DD, DG and GG genotypes were 75.9, 22.8, and 1.3%, respectively. The genotype distribution of MTR D919G polymorphism was in HWE (χ 2=0.15, P>0.05), and the HET and PIC were 0.222 and 0.197, respectively. Since only as few as 5 patients carried the GG genotype, we combined them with those patients carrying the DG genotype in the further analysis. Characteristics of the enrolled hypertensive patients stratified by genotypes are summarized in Table 1. Except for 16th-day blood pressure, there were no significant differences in age, gender, height, weight, BMI, occupation, education, smoking, drinking, and baseline blood pressure between the DD and DG + GG groups. Compared to the reference DD subjects, the DG + GG subjects had a significantly less DBP reduction (β=−2.3, P=0.010) resulting from benazepril therapy (Table 2). Because lipid profiles and diabetes status could also potentially influence blood pressure response to benazepril, we performed further analysis after adjustment for hypercholesterolemia (TC>5.72 mmol/l) and hyperglycemia (fasting glucose ≥7.0 mmol/l). The regression coefficients of these two variables were not statistically significant (P values for hypercholesterolemia and hyperglycemia were 0.388 and 0.669, respectively). Also, there were no significant differences in terms of both β and P values before or after the adjustment for hypercholesterolemia and hyperglycemia. (Table is not shown.) A total of 1,951 subjects from 654 families were included in FBAT analysis, and their family structures are shown in Table 3. Finally, 157, 153, and 7 informative families were analyzed using FBAT under additive, dominant, and recessive models, respectively (Table 4). Consistent with the result of population-based association test, both additive (Z=−2.4, P=0.018) and dominant (Z=−2.2, P=0.025) models in FBAT analysis showed that patients who carried the 919G allele had much less DBP reduction than those who carried the 919D allele. No significant association was found between SBP reduction response to benazepril and the D919G polymorphism of the MTR gene using both methods.
Discussion
Many demographic and environmental factors such as age, gender, race, diet, and drug interactions are known to influence the individual response to medications (Massie 1987). Although ACE inhibitors are generally effective in the treatment of hypertension, there is still a remarkable heterogeneity among different ethnic groups, including both therapeutic and adverse effects. For instance, there appear to be more potent antihypertensive effects and more frequent side effects such as ACE-inhibitor-induced cough in Chinese hypertensives than in Caucasian hypertensives (Ding et al. 2000). It has also been suggested that individual variation in response to drugs can be influenced by genetic factors (Evans and McLeod 2003; Weinshilboum 2003). Polymorphisms in those genes encoding phase I and phase II drug-metabolizing enzymes may alter the structure, configuration, activity, or amount of protein products, and these may lead to different responses as well. Such examples include CYP2C9 variants and warfarin sensitivity (Aithal et al. 1999) and ADRB2 polymorphisms and isoproterenol efficacy (Dishy et al. 2001). In addition, polymorphisms in other genes involved in the pharmacodynamic pathway of drugs can also influence their efficacy, such as the association between FokI (+/−) polymorphism in the GNAS1 gene and blood pressure response to beta blockers (Jia et al. 1999).
The MTR gene encodes an enzyme that catalyzes the remethylation of Hcy to methionine using a methyl group donated by 5-methyltetrahydrofolate. Based on the study in bacteria, the 919th amino acid residue is located within the adenosylmethionine-binding domain of the enzyme (Drummond et al. 1993). Although the 919G allele of the MTR gene was also found to be associated with a lower plasma Hcy level (Chen et al. 2001; Harmon et al. 1999) and a decreased risk of juvenile hypertension (Kahleova et al. 2002) in previous human studies, its functional significance remains to be determined. Trough/peak ratio, calculated as the percentage of blood pressure reduction at the end of the dose interval divided by the maximum blood pressure reduction during this interval after subtracting placebo effect, is recommended to evaluate the appropriate dose interval for safe and effective control of blood pressure. But the estimates may result in a spuriously high trough/peak ratio when nonresponders are included (Menard et al. 1994). Since benazepril has been well approved for once-daily use (Balfour and Goa 1991), the individual variation in drug response was then evaluated by the trough effect.
In this study, we demonstrated that the 919G allele of the MTR gene was associated with a significantly less DBP reduction after 15 days of benazepril treatment in Huoqiu hypertensive patients. Although the trend of SBP reduction was similar to that of DBP reduction, no significant association was found. It is well known that population admixture may give rise to spurious association in population-based regression analysis. FBAT, a method with the advantage of avoiding false-positive results due to population admixture, was then performed (Rabinowitz and Laird 2000). The fact that both population-based and family-based association tests produced similar results made our findings much more convincing.
The underlying pathophysiological mechanism by which the 919G allele of the MTR gene decreases DBP response to benazepril in hypertensive subjects is still unknown. When clinically healthy subjects were under a 2-year Hcy-lowering therapy with folic acid plus pyridoxine, their blood pressure was significantly dropped (van Dijk et al. 2001). Thus, we postulate that the 919G allele may decrease blood pressure response to benazepril through its influence on plasma Hcy level, which has the potential to affect vascular reactivity in several ways. First, Hcy can exacerbate angiotensin II-induced vasoconstriction. When aortic vascular smooth-muscle cells were pretreated with Hcy, intracellular Ca2+ can be raised by angiotensin II well below the physiologic range (Mujumdar et al. 2000). Therefore, angiotensin II may have an enhanced vasoconstrictive effect on patients with high Hcy levels, which could lead to a better response to benazepril. Second, a high-plasma Hcy level has deleterious effects on vascular endothelium function. Flow-mediated (endothelium dependent), not glyceryl trinitrate-mediated (endothelium independent) brachial artery vasodilation was impaired in both methionine-induced acute (Chambers et al. 1999) and chronic hyperhomocysteinaemia (Woo et al. 1997). Acute hyperhomocysteinaemia also has no effect on microvascular vasodilation response to acetylcholine (endothelium dependent) or sodium nitroprusside (endothelium independent) in vivo (Davis et al. 2001). On the other hand, Hcy-induced inhibition of endothelium-dependent relaxation in isolated rat aorta can be prevented by captopril (Fu et al. 2003). Since only nitric oxide-mediated vasodilation is damaged, the impact of Hcy on endothelium function may have small influence on blood pressure reduction response to benazepril. Finally, a high-plasma Hcy level might be associated with increased arterial stiffness as well as thickness (Bortolotto et al. 1999; Malinow et al. 1993; Nestel et al. 2003; Smilde et al. 1998), which may lead to a poor response to benazepril. But whether blood pressure can be directly affected by an increased arterial stiffness resulting from hyperhomocysteinemia remains to be elucidated (Davis et al. 2001; Nestel et al. 2003).
Methylenetetrahydrofolate reductase (MTHFR) is another important enzyme involved in Hcy metabolism. The MTHFR gene C677T polymorphism, which is located in the predicted catalytic domain of the MTHFR enzyme, was found to be associated with a decreased enzyme activity and an elevated plasma Hcy level (Frosst et al. 1995). Individuals carrying the mutant T allele with a higher plasma Hcy level might also have a higher blood pressure (Inamoto et al. 2003). Thus, the T allele of the MTHFR C677T polymorphism and the 919G allele of the MTR D919G polymorphism seem to have conflicting effects in blood pressure response to benazepril. A previous study showed that subjects with the variants on both MTHFR and MTR could have extremely high-plasma Hcy levels (Feix et al. 2001). So based on our hypothesis, it would be interesting in the future to test the association between blood pressure response to benazepril and the MTHFR polymorphism alone or in conjunction with MTR polymorphism.
In summary, our study suggests that the D919G polymorphism of the MTR gene may be a useful genetic predictor of the antihypertensive effect of short-term benazepril therapy in hypertensive patients of Anhui Province, China. It is of note that a recent investigation found additional regulatory, splicing-site, or missense polymorphisms in the MTR gene (Watkins et al. 2002). Since haplotype studies may offer more statistical power and reveal new biological insights than single-marker studies (Niu et al. 2002), it is also important to further study the relationship between benazepril efficacy and haplotypic effects (i.e., “combined effects” of multiple polymorphisms located within the same gene) of the MTR gene.
References
Aithal GP, Day CP, Kesteven PJ, Daly AK (1999) Association of polymorphisms in the cytochrome P450 CYP2C9 with warfarin dose requirement and risk of bleeding complications. Lancet 353:717–719
Balfour JA, Goa KL (1991) Benazepril. A review of its pharmacodynamic and pharmacokinetic properties, and therapeutic efficacy in hypertension and congestive heart failure. Drugs 42:511–539
Bortolotto LA, Safar ME, Billaud E, Lacroix C, Asmar R, London GM, Blacher J (1999) Plasma homocysteine, aortic stiffness, and renal function in hypertensive patients. Hypertension 34:837–842
Chambers JC, McGregor A, Jean-Marie J, Obeid OA, Kooner JS (1999) Demonstration of rapid onset vascular endothelial dysfunction after hyperhomocysteinemia: an effect reversible with vitamin C therapy. Circulation 99:1156–1160
Chen J, Stampfer MJ, Ma J, Selhub J, Malinow MR, Hennekens CH, Hunter DJ (2001) Influence of a methionine synthase (D919G) polymorphism on plasma homocysteine and folate levels and relation to risk of myocardial infarction. Atherosclerosis 154:667–672
Davis KR, Pearson H, Moat S, Bonham JR, Donnelly R (2001) Acute hyperhomocysteinaemia affects pulse pressure but not microvascular vasodilator function. Br J Clin Pharmacol 52:327–332
van Dijk RA, Rauwerda JA, Steyn M, Twisk JW, Stehouwer CD (2001) Long-term homocysteine-lowering treatment with folic acid plus pyridoxine is associated with decreased blood pressure but not with improved brachial artery endothelium-dependent vasodilation or carotid artery stiffness: a 2-year, randomized, placebo-controlled trial. Arterioscler Thromb Vasc Biol 21:2072–2079
Ding PY, Hu OY, Pool PE, Liao W (2000) Does Chinese ethnicity affect the pharmacokinetics and pharmacodynamics of angiotensin-converting enzyme inhibitors? J Hum Hypertens 14:163–170
Dishy V, Sofowora GG, Xie HG, Kim RB, Byrne DW, Stein CM, Wood AJ (2001) The effect of common polymorphisms of the beta2-adrenergic receptor on agonist-mediated vascular desensitization. N Engl J Med 345:1030–1035
Drummond JT, Huang S, Blumenthal RM, Matthews RG (1993) Assignment of enzymatic function to specific protein regions of cobalamin-dependent methionine synthase from Escherichia coli. Biochemistry 32:9290–9295
Evans WE, McLeod HL (2003) Pharmacogenomics—drug disposition, drug targets, and side effects. N Engl J Med 348:538–549
Feix A, Fritsche-Polanz R, Kletzmayr J, Vychytil A, Horl WH, Sunder-Plassmann G, Fodinger M (2001) Increased prevalence of combined MTR and MTHFR genotypes among individuals with severely elevated total homocysteine plasma levels. Am J Kidney Dis 38:956–964
Fiorina P, Lanfredini M, Montanari A, Peca MG, Veronelli A, Mello A, Astorri E, Craveri A (1998) Plasma homocysteine and folate are related to arterial blood pressure in type 2 diabetes mellitus. Am J Hypertens 11:1100–1107
Frosst P, Blom HJ, Milos R, Goyette P, Sheppard CA, Matthews RG, Boers GJ, den Heijer M, Kluijtmans LA, van den Heuvel LP (1995) A candidate genetic risk factor for vascular disease: a common mutation in methylenetetrahydrofolate reductase. Nat Genet 10:111–113
Fu YF, Xiong Y, Fu SH (2003) Captopril restores endothelium-dependent relaxation of rat aortic rings after exposure to homocysteine. J Cardiovasc Pharmacol 42:566–572
Harmon DL, Shields DC, Woodside JV, McMaster D, Yarnell JW, Young IS, Peng K, Shane B, Evans AE, Whitehead AS (1999) Methionine synthase D919G polymorphism is a significant but modest determinant of circulating homocysteine concentrations. Genet Epidemiol 17:298–309
Inamoto N, Katsuya T, Kokubo Y, Mannami T, Asai T, Baba S, Ogata J, Tomoike H, Ogihara T (2003) Association of methylenetetrahydrofolate reductase gene polymorphism with carotid atherosclerosis depending on smoking status in a Japanese general population. Stroke 34:1628–1633
Jia H, Hingorani AD, Sharma P, Hopper R, Dickerson C, Trutwein D, Lloyd DD, Brown MJ (1999) Association of the G(s)alpha gene with essential hypertension and response to beta-blockade. Hypertension 34:8–14
Kahleova R, Palyzova D, Zvara K, Zvarova J, Hrach K, Novakova I, Hyanek J, Bendlova B, Kozich V (2002) Essential hypertension in adolescents: association with insulin resistance and with metabolism of homocysteine and vitamins. Am J Hypertens 15:857–864
Li D, Xing H, Hao K, Peng S, Wu D, Guang W, Huang A, Wang X, Zhang Y, Yun Y, Li J, Chen C, Wang B, Zhu G, Huo Y, Chen D, Ronnenberg A, Niu T, Xu X (2004) Hypertensive patients from two rural Chinese counties respond differently to benazepril: the Anhui hypertension health care study. Ann Epidemiol 14:123–128
Lim U, Cassano PA (2002) Homocysteine and blood pressure in the Third National Health and Nutrition Examination Survey, 1988–1994. Am J Epidemiol 156:1105–1113
Malinow MR, Nieto FJ, Szklo M, Chambless LE, Bond G (1993) Carotid artery intimal-medial wall thickening and plasma homocyst(e)ine in asymptomatic adults. The atherosclerosis risk in communities study. Circulation 87:1107–1113
Massie BM (1987) Demographic considerations in the selection of antihypertensive therapy. Am J Cardiol 60:121I–126I
Menard J, Chatellier G, Day M, Vaur L (1994) Self-measurement of blood pressure at home to evaluate drug effects by the trough: peak ratio. J Hypertens Suppl 12:S21–S25
Mujumdar VS, Hayden MR, Tyagi SC (2000) Homocyst(e)ine induces calcium second messenger in vascular smooth muscle cells. J Cell Physiol 183:28–36
Nestel PJ, Chronopoulos A, Cehun M (2003) Arterial stiffness is rapidly induced by raising the plasma homocysteine concentration with methionine. Atherosclerosis 171:83–86
Niu T, Struk B, Lindpaintner K (2001) Statistical considerations for genome-wide scans: design and application of a novel software package POLYMORPHISM. Hum Hered 52:102–109
Niu T, Qin ZS, Xu X, Liu JS (2002) Bayesian haplotype inference for multiple linked single-nucleotide polymorphisms. Am J Hum Genet 70:157–169
Nygard O, Vollset SE, Refsum H, Stensvold I, Tverdal A, Nordrehaug JE, Ueland M, Kvale G (1995) Total plasma homocysteine and cardiovascular risk profile. The Hordaland Homocysteine Study. JAMA 274:1526–1533
Perry IJ, Refsum H, Morris RW, Ebrahim SB, Ueland PM, Shaper AG (1995) Prospective study of serum total homocysteine concentration and risk of stroke in middle-aged British men. Lancet 346:1395–1398
Rabinowitz D, Laird N (2000) A unified approach to adjusting association tests for population admixture with arbitrary pedigree structure and arbitrary missing marker information. Hum Hered 50:211–223
Smilde TJ, van den Berkmortel FW, Boers GH, Wollersheim H, de Boo T, van Langen H, Stalenhoef AF (1998) Carotid and femoral artery wall thickness and stiffness in patients at risk for cardiovascular disease, with special emphasis on hyperhomocysteinemia. Arterioscler Thromb Vasc Biol 18:1958–1963
Stampfer MJ, Malinow MR, Willett WC, Newcomer LM, Upson B, Ullmann D, Tishler PV, Hennekens CH (1992) A prospective study of plasma homocyst(e)ine and risk of myocardial infarction in US physicians. JAMA 268:877–881
Sutton-Tyrrell K, Bostom A, Selhub J, Zeigler-Johnson C (1997) High homocysteine levels are independently related to isolated systolic hypertension in older adults. Circulation 96:1745–1749
Watkins D, Ru M, Hwang HY, Kim CD, Murray A, Philip NS, Kim W, Legakis H, Wai T, Hilton JF, Ge B, Dore C, Hosack A, Wilson A, Gravel RA, Shane B, Hudson TJ, Rosenblatt DS (2002) Hyperhomocysteinemia due to methionine synthase deficiency, cblG: structure of the MTR gene, genotype diversity, and recognition of a common mutation, P1173L. Am J Hum Genet 71:143–153
Weinshilboum R (2003) Inheritance and drug response. N Engl J Med 348:529–537
Woo KS, Chook P, Lolin YI, Cheung AS, Chan LT, Sun YY, Sanderson JE, Metreweli C, Celermajer DS (1997) Hyperhomocyst(e)inemia is a risk factor for arterial endothelial dysfunction in humans. Circulation 96:2542–2544
Acknowledgements
We gratefully acknowledge the assistance and cooperation of the faculties and staffs of Anhui Medical University, University of Science and Technology in China, Peking University First Hospital, and Harvard School of Public Health. We are thankful to all the patients in our study and their families.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zhang, Y., Zhang, M., Niu, T. et al. D919G polymorphism of methionine synthase gene is associated with blood pressure response to benazepril in Chinese hypertensive patients. J Hum Genet 49, 296–301 (2004). https://doi.org/10.1007/s10038-004-0149-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10038-004-0149-0
Keywords
This article is cited by
-
Effects of methionine synthase and methionine synthase reductase polymorphisms on hypertension susceptibility
Genes & Genomics (2020)
-
The methionine synthase polymorphism c.2756A>G (D919G) influences diastolic blood pressure
Journal of Human Hypertension (2007)