Abstract
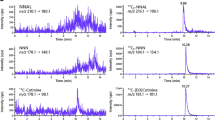
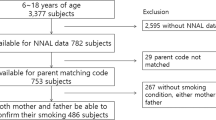
Carcinogenic tobacco-specific nitrosamines (TSNAs) such as 4-(methylnitrosamino)-1-(3-pyridyl)-1-butanone (NNK) are found only in tobacco and derived products. Food and Drug Administration of the United States (US FDA) lists NNK as one of the 93 harmful and potentially harmful constituents (HPHCs) found in tobacco products and tobacco smoke. The aim of this study was to use the urinary concentration of 4-(methylnitrosamino)-1-(3-pyridyl)-1-butanol (NNAL), a major metabolite of NNK, to quantitatively estimate exposure to NNK in the US general population. In 2011–2012, the Centers for Disease Control and Prevention’s National Health and Nutrition Examination Survey (NHANES) collected urine and serum samples from a representative sample of US residents. We used a serum cotinine cutoff of 10 ng/ml with combination of questionnaire data to select non-users from cigarette users and used self-reported data to determine different tobacco product user groups. We estimated the absorbed total daily dose of NNK using a probabilistic method based on a two-compartment model. The geometric mean (GM) for the daily dose of NNK among smokers aged 12–16 years was significantly higher than that for non-users at the same age stage exposed to second-hand smoke (SHS) (P<0.001). Among those exposed to SHS, the GM for daily dose of NNK in young children (6–11 years) was nearly three times of those for adults in the age range 21–59 years. Among cigarette users, non-Hispanic Whites had the highest NNK daily dose and Mexican Americans had the lowest levels. Exclusive snuff or chewing product users had significantly higher daily dose of NNK than did cigarette smokers. Our study found that the maximum daily dose of NNK for children aged from 6 to 11 years and that for a significant percentage of cigarette users, chewing product and snuff users were higher than an estimated provisional “reference” risk level.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 6 print issues and online access
$259.00 per year
only $43.17 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Hecht SS, Carmella SG, Stepanov I, Jensen J, Anderson A, Hatsukami DK . Metabolism of the tobacco-specific carcinogen 4-(methylnitrosamino)-1-(3-pyridyl)-1-butanone to its biomarker total nnal in smokeless tobacco users. Cancer Epidemiol Biomarkers Rev 2008; 17: 732–735.
Hecht SS . Biochemistry, biology, and carcinogenicity of tobacco-specific N-nitrosamines. Chem Res Toxicol 1998; 11: 559–603.
Hecht SS, Trushin N, Rigotty J, Carmella SG, Borukhova A, Akerkar S, Rivenson A . Complete inhibition of 4-(methylnitrosamino)-1-(3-pyridyl)-1-butanone-induced rat lung tumorigenesis and favorable modification of biomarkers by phenethyl isothiocyanate. Cancer Epidemiol Biomarkers Rev 1996; 5: 645–652.
Hoffmann D, Rivenson A, Amin S, Hecht SS . A study of tobacco carcinogenesis .27. Dose-response study of the carcinogenicity of tobacco-specific N-nitrosamines in F344 rats. J Cancer Res Clin 1984; 108: 81–86.
Derby KS, Cuthrell K, Caberto C, Carmella S, Murphy SE, Hecht SS,. Exposure to the carcinogen 4-(methylnitrosamino)-1-(3-pyridyl)-1-butanone (NNK) in smokers from 3 populations with different risks of lung cancer. Int J Cancer 2009; 125: 2418–2424.
Benowitz NL, Renner CC, Lanier AP, Tyndale RF, Hatsukami DK, Lindgren B et al. Exposure to nicotine and carcinogens among Southwestern Alaskan native cigarette smokers and smokeless tobacco users. Cancer Epidemiol Biomarkers Rev 2012; 21: 934–942.
E.P.A., U.S. US Environmental Protection Agency. Respiratory Health Effects of Passive Smoking: Lung Cancer and Other Disorders 1992. Available from Website: http://cfpub2.epa.gov/ncea/cfm/recordisplay.cfm?deid=2835 (accessed 15 December 2014).
US-FDA. Harmful and Potentially Harmful Constituents in Tobacco Products and Tobacco Smoke: Established List 2012. Available from: http://www.fda.gov/TobaccoProducts/GuidanceComplianceRegulatoryInformation/ucm297786.htm (accessed 11 June 2014).
Hoffmann D, Rivenson A, Wynder EL, Hecht SS . Formation of tobacco-specific nitrosamines—carcinogenicity and role of dietary-fat in their carcinogenicity. Nitrosamines Related N-Nitroso Compounds 1994; 553: 267–278.
Hecht SS, Chen CB, Hirota N, Ornaf RM, Tso TC, Hoffmann D . Tobacco-specific nitrosamines: formation from nicotine in vitro and during tobacco curing and carcinogenicity in strain A mice. J Natl Cancer Inst 1978; 60: 819–824.
Wu W, Zhang L, Jain RB, Ashley DL, Watson CH . Determination of carcinogenic tobacco-specific nitrosamines in mainstream smoke from US-brand and non-US-brand cigarettes from 14 countries. Nicotine Tob Res 2005; 7: 443–451.
IARCInternational Agency for Research on Cancer. Smokeless tobacco and some tobacco-specific N-nitrosamines. IARC monographs on the evaluation of carcinogenic risks to humans. Available from: http://monographs.iarc.fr/ENG/Monographs/vol89/index.php (accessed 15 December 2014), 2007.
Hecht SS, Carmella SG, Ye M, Le KA, Jensen JA, Zimmerman CL et al. Quantitation of metabolites of 4-(methylnitrosamino)-1-(3-pyridyl)-1-butanone after cessation of smokeless tobacco use. Cancer Res 2002; 62: 129–134.
Helen G, Bernert JT, Hall DB, Sosnoff CS, Xia Y, Balmes JR et al. Exposure to secondhand smoke outside of a bar and a restaurant and tobacco exposure biomarkers in nonsmokers. Environ Health Perspect 2012; 120: 1010–1016.
Vardavas CI, Fthenou E, Patelarou E, Bagkeris E, Murphy S, Hecht SS et al. Exposure to different sources of second-hand smoke during pregnancy and its effect on urinary cotinine and tobacco-specific nitrosamine (NNAL) concentrations. Tob Control. 2012; 22: 194–200.
Hecht SS, Carmella SG, Chen M, Dor Koch JF, Miller AT, Murphy SE et al. Quantitation of urinary metabolites of a tobacco-specific lung carcinogen after smoking cessation. Cancer Res 1999; 59: 590–596.
Jacob P, Havel C, Lee DH, Yu L, Eisner MD, Benowitz NL . Subpicogram per milliliter determination of the tobacco-specific carcinogen metabolite 4-(methylnitrosamino)-1-(3-pyridyl)-1-butanol in human urine using liquid chromatography-tandem mass spectrometry. Anal Chem 2008; 80: 8115–8121.
Goniewicz ML, Havel CM, Peng MW, Jacob P, 3rd, Dempsey D, Yu L et al. Elimination kinetics of the tobacco-specific biomarker and lung carcinogen 4-(methylnitrosamino)-1-(3-pyridyl)-1-butanol. Cancer Epidemiol Biomarkers Rev 2009; 18: 3421–3425.
Hukkanen J, Jacob P, Benowitz NL . Metabolism and disposition kinetics of nicotine. Pharmacol Rev 2005; 57: 79–115.
McGuffey JE, Wei B, Bernert JT, Morrow JC, Xia B, Wang L et al. Validation of a lc-ms/ms method for quantifying urinary nicotine, six nicotine metabolites and the minor alkaloids—anatabine and anabasine—in smokers’ urine. PLoS One 2014; 9 (7): e101816.
Wei B, Feng J, Rehmani IJ, Miller S, McGuffey JE, Blount BC et al. A high-throughput robotic sample preparation system and HPLC-MS/MS for measuring urinary anatabine, anabasine, nicotine and major nicotine metabolites. Clin Chim Acta 2014; 436 (25): 290–297.
Xia Y, McGuffey JE, Bhattacharyya S, Sellergren B, Yilmaz E, Wang L et al. Analysis of the tobacco-specific nitrosamine 4-(methylnitrosamino)-1-(3-pyridyl)-1-butanol in urine by extraction on a molecularly imprinted polymer column and liquid chromatography/atmospheric pressure ionization tandem mass spectrometry. Anal Chem 2005; 77: 7639–7645.
Carmella SG, Le KA, Upadhyaya P, Hecht SS . Analysis of N- and O-glucuronides of 4-(methylnitrosamino)-1-(3-pyridyl)-1-butanol (NNAL) in human urine. Chem Res Toxicol 2002; 15: 545–550.
Xia Y, Bernert JT, Jain RB, Ashley DL, Pirkle JL . Tobacco-specific nitrosamine 4-(methylnitrosamino)-1-(3-pyridyl)-1-butanol (NNAL) in smokers in the United States: NHANES 2007-2008. Biomarkers 2011; 16: 112–119.
Bernert JT, Pirkle JL, Xia Y, Jain RB, Ashley DL, Sampson EJ . Urine concentrations of a tobacco-specific nitrosamine carcinogen in the US population from secondhand smoke exposure. Cancer Epidemiol Biomarkers Rev 2010; 19: 2969–2977.
US-CDC. National Health and Nutrition Examination Survey. National Center for Health Statistics. Centers for Disease Control and Prevention. Available from: http://www.cdc.gov/nchs/nhanes.htm (accessed 24 September 2014), 2014.
Xia Y, Bernert JT . Stability of the tobacco-specific nitrosamine 4-(methylnitrosamino)-1-(3-pyridyl)-1-butanol in urine samples stored at various temperatures. J Anal Toxicol 2010; 34: 411–415.
Bernert JT, Turner WE, Pirkle JL, Sosnoff CS, Akins JR, Waldrep MK et al. Development and validation of sensitive method for determination of serum cotinine in smokers and nonsmokers by liquid chromatography atmospheric pressure ionization tandem mass spectrometry. Clin Chem 1997; 43: 2281–2291.
Bernert JT, McGuffey JE, Morrison MA, Pirkle JL . Comparison of serum and salivary cotinine measurements by a sensitive high-performance liquid chromatography-tandem mass spectrometry method as an indicator of exposure to tobacco smoke among smokers and nonsmokers. J Anal Toxicol 2000; 24: 333–339.
Caudill SP, Schleicher RL, Pirkle JL . Multi-rule quality control for the age-related eye disease study. Stat Med 2008; 27: 4094–4106.
David RM . Exposure to phthalate esters. Environ Health Perspect 2000; 108: A440–A440.
Kohn MC, Parham F, Masten SA, Portier CJ, Shelby MD, Brock JW et al. Human exposure estimates for phthalates. Environ Health Perspect 2000; 108: A440–A442.
Wei B, Isukapalli SS, Weisel CP . Studying permethrin exposure in flight attendants using a physiologically based pharmacokinetic model. J Expo Sci Environ Epidemiol 2013; 23: 416–427.
Pirkle JL, Flegal KM, Bernert JT, Brody DJ, Etzel RA, Maurer KR . Exposure of the US population to environmental tobacco smoke. JAMA 1996; 275: 1233–1240.
Pirkle JL, Bernert JT, Caudill SA, Sosnoff CS, Pechacek TF . Trends in the exposure of nonsmokers in the US population to secondhand smoke: 1988-2002. Environ Health Perspect 2006; 114: 853–858.
Hornung RW, Reed LD . Estimation of average concentration in the presence of nondetectable values. Appl Occup Environ Hygiene 1990; 5: 46–51.
Wei BN, Mohan KR, Weisel CP . Exposure of flight attendants to pyrethroid insecticides on commercial flights: Urinary metabolite levels and implications. Int J Hygiene Environ Health 2012; 215: 465–473.
Pirkle JL, Flegal KM, Bernert JT, Brody DJ, Etzel RA, Maurer KR . Exposure of the US population to environmental tobacco smoke—The Third National Health and Nutrition Examination Survey, 1988 to 1991. JAMA 1996; 275: 1233–1240.
Hecht SS, Trushin N, Reid-Quinn CA, Burak ES, Jones AB, Southers JL et al. Metabolism of the tobacco-specific nitrosamine 4-(methylnitrosamino)-1-(3-pyridyl)-1-butanone in the patas monkey: pharmacokinetics and characterization of glucuronide metabolites. Carcinogenesis 1993; 14: 229–236.
Meger M, Richter E, Zwickenpflug W, Oehlmann C, Hargaden MB, A-Rahim YI et al. Metabolism and disposition of 4-(methylnitrosamino)-1-(3-pyridyl)-1- butanone (NNK) in rhesus monkeys. Drug Metab Dispos 1999; 27: 471–478.
Wilson KM, Klein JD, Blumkin AK, Gottlieb M, Winickoff JP . Tobacco-smoke exposure in children who live in multiunit housing. Pediatrics 2011; 127: 85–92.
Matt GE, Quintana PJ, Zakarian JM, Fortmann AL, Chatfield DA, Hoh E et al. When smokers move out and non-smokers move in: residential thirdhand smoke pollution and exposure. Tob Control 2011; 20: e1.
Sleiman M, Gundel LA, Pankow JF, Jacob P, 3rd, Singer BC, Destaillats H . Formation of carcinogens indoors by surface-mediated reactions of nicotine with nitrous acid, leading to potential thirdhand smoke hazards. Proc Natl Acad Sci USA 2010; 107: 6576–6581.
US-EPA. US Environmental Protection Agency Exposure Factors Handbook 2011 Edition. (Final): Chapter 4—Non-Dietary Ingestion Factors 2011.
Caraballo RS, Giovino GA, Pechacek TF, Mowery PD, Richter PA, Strauss WJ et al. Racial and ethnic differences in serum cotinine levels of cigarette smokers. JAMA 1998; 280: 135–139.
Haiman CA, Stram DO, Wilkens LR, Pike MC, Kolonel LN, Henderson BE et al. Ethnic and racial differences in the smoking-related risk of lung cancer. N Engl J Med 2006; 354: 333–342.
Benowitz NL, Dains KM, Dempsey D, Wilson M, Jacob P . Racial differences in the relationship between number of cigarettes smoked and nicotine and carcinogen exposure. Nicotine Tob Res 2011; 13: 772–783.
Kresty LA, Carmella SG, Borukhova A, Akerkar SA, Gopalakrishnan R, Harris RE et al. Metabolites of a tobacco-specific nitrosamine, 4-(methylnitrosamino)-1-(3-pyridyl)-1-butanone (NNK), in the urine of smokeless tobacco users: relationship between urinary biomarkers and oral leukoplakia. Cancer Epidemiol Biomarkers Rev 1996; 5: 521–525.
Stepanov I, Hecht SS . Tobacco-specific nitrosamines and their pyridine-N-glucuronides in the urine of smokers and smokeless tobacco users. Cancer Epidemiol Biomarkers Rev 2005; 14: 885–891.
Rivenson A, Hoffmann D, Prokopczyk B, Amin S, Hecht SS . Induction of lung and exocrine pancreas tumors in F344 rats by tobacco-specific and Areca-derived N-nitrosamines. Cancer Res 1988; 48: 6912–6917.
Hoffmann D, Rivenson A, Abbi R, Wynder EL . A study of tobacco carcinogenesis: effect of the fat content of the diet on the carcinogenic activity of 4-(methylnitrosamino)-1-(3-pyridyl)-1-butanone in F344 rats. Cancer Res 1993; 53: 2758–2761.
Belinsky SA, Foley JF, White CM, Anderson MW, Maronpot RR . Dose-response relationship between O6-methylguanine formation in Clara cells and induction of pulmonary neoplasia in the rat by 4-(methylnitrosamino)-1-(3-pyridyl)-1-butanone. Cancer Res 1990; 50: 3772–3780.
Winickoff JP, Friebely J, Tanski SE, Sherrod C, Matt GE, Hovell MF et al. Beliefs about the health effects of “thirdhand” smoke and home smoking bans. Pediatrics 2009; 123: e74–e79.
Eisner MD, Balmes J, Katz PP, Trupin L, Yelin EH, Blanc PD . Lifetime environmental tobacco smoke exposure and the risk of chronic obstructive pulmonary disease. Environ Health 2005; 4: 7.
Johansson A, Hermansson G, Ludvigsson J . How should parents protect their children from environmental tobacco-smoke exposure in the home? Pediatrics 2004; 113: e291–e295.
Yolton K, Dietrich K, Auinger P, Lanphear BP, Hornung R . Exposure to environmental tobacco smoke and cognitive abilities among US children and adolescents. Environ Health Perspect 2005; 113: 98.
Ng M, Freeman MK, Fleming TD, Robinson M, Dwyer-Lindgren L, Thomson B et al. Smoking prevalence and cigarette consumption in 187 countries, 1980-2012. JAMA 2014; 311: 183–192.
Melikian AA, Djordjevic MV, Hosey J, Zhang J, Chen S, Zang E et al. Gender differences relative to smoking behavior and emissions of toxins from mainstream cigarette smoke. Nicotine Tob Res 2007; 9: 377–387.
Bernert JT, Pirkle JL, Xia Y, Jain RB, Ashley DL, Sampson EJ . Urine concentrations of a tobacco-specific nitrosamine carcinogen in the U.S. population from secondhand smoke exposure. Cancer Epidemiol Biomarkers Rev 2010; 19: 2969–2977.
Ter-Minassian M, Asomaning K, Zhao Y, Chen F, Su L, Carmella SG et al. Genetic variability in the metabolism of the tobacco-specific nitrosamine 4-(methylnitrosamino)-1-(3-pyridyl)-1-butanone (NNK) to 4-(methylnitrosamino)-1-(3-pyridyl)-1-butanol (NNAL). Int J Cancer 2012; 130: 1338–1346.
Richie JP, Carmella SG, Muscat JE, Scott DG, Akerkar SA, Hecht SS . Differences in the urinary metabolites of the tobacco-specific lung carcinogen 4-(methylnitrosamino)-1-(3-pyridyl)-1-butanone in black and white smokers. Cancer Epidemiol Biomarkers Rev 1997; 6: 783–790.
Muscat JE, Djordjevic MV, Colosimo S, Stellman SD, Richie JP . Racial differences in exposure and glucuronidation of the tobacco‐specific carcinogen 4‐(methylnitrosamino)‐1‐(3‐pyridyl)‐1‐butanone (NNK). Cancer 2005; 103: 1420–1426.
Carmella SG, Akerkar SA, Richie J, Hecht SS . Intraindividual and interindividual differences in metabolites of the tobacco-specific lung carcinogen 4-(methylnitrosamino)-1-(3-pyridyl)-1-butanone (NNK) in smokers' urine. Cancer Epidemiol Biomarkers Rev 1995; 4: 635–642.
Morse MA, Eklind KI, Toussaint M, Amin SG, Chung F-L . Characterization of a glucuronide metabolite of 4-(methylnitrosamino)-1-(3-pyridyl)-1-butanone (NNK) and its dose-dependent excretion in the urine of mice and rats. Carcinogenesis 1990; 11: 1819–1823.
Acknowledgements
We thank Yang Xia and Connie S. Sosnoff for measuring urinary total NNAL and serum cotinine, respectively. We also thank Ernest L. Martin, Patricia Ruiz and Rey Decastro at CDC for their valuable comments. The findings and conclusions in this presentation are those of the authors and do not necessarily represent the views of the Centers for Disease Control and Prevention (CDC). Use of trade names and commercial sources is for identification only, and such use does not constitute endorsement by the US Department of Health and Human Services or the Centers for Disease Control and Prevention.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies the paper on the Journal of Exposure Science and Environmental Epidemiology website
Supplementary information
Rights and permissions
About this article
Cite this article
Wei, B., Blount, B., Xia, B. et al. Assessing exposure to tobacco-specific carcinogen NNK using its urinary metabolite NNAL measured in US population: 2011–2012. J Expo Sci Environ Epidemiol 26, 249–256 (2016). https://doi.org/10.1038/jes.2014.88
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/jes.2014.88
Keywords
This article is cited by
-
Carcinogenic and tobacco smoke-derived particulate matter biomarker uptake and associated healthcare patterns among children
Pediatric Research (2023)
-
Association between DNA methylation variability and self-reported exposure to heavy metals
Scientific Reports (2022)
-
LKB1 phosphorylation and deactivation in lung cancer by NNAL, a metabolite of tobacco-specific carcinogen, in an isomer-dependent manner
Oncogene (2022)
-
Association between Urinary Metabolite Levels of Organophosphorus Flame Retardants and Serum Sex Hormone Levels Measured in a Reference Sample of the US General Population
Exposure and Health (2020)
-
Dietary phytochemicals as the potential protectors against carcinogenesis and their role in cancer chemoprevention
Clinical and Experimental Medicine (2020)