Abstract
Objective:
Maternal obesity significantly increases the susceptibility of offspring to develop obesity and chronic diseases in adulthood. The offspring of obese mothers are shown to prefer high fat diet (HFD) due to their altered neural circuitry, creating a ‘feed-forward cycle’ across generations. We hypothesized that the ‘feed-forward cycle’ caused by multigenerational HFD feeding would have exacerbated effects in adipose tissue of the offspring.
Methods:
Three generations (F0, F1 and F2) of HFD (60% Kcal fat)-fed and corresponding normal chow (NC)-fed C57BL/6 mice were generated. Body weight (BW) and food intake were monitored weekly. Parametrial adipose tissue (pAT) weight and endocrine parameters were measured in 9-month-old female offspring. Gene expression microarray, quantitative RT-PCR and bisulfite sequencing were performed using pAT.
Results:
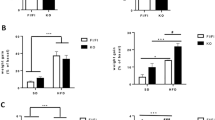
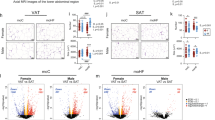
BW and pAT weight increased in female mice across generations under continuous HFD stress, with the most severe phenotype found in the F2 generation. Genes involved in inflammatory response showed increased expression across generations in the pAT, accompanied by increased macrophage infiltration. The promoters of Toll-like receptor 1 (Tlr1), Tlr2 and linker for activation of T cells (Lat) were hypomethylated in the HF groups compared with the NC group, with additional hypomethylation on some specific CpG sites in the F2 generation.
Conclusions:
A feed-forward cycle exists in female mice after continuous HFD stress as demonstrated by increased adiposity and progressive inflammation in adipose tissue across generations. DNA hypomethylation over generations lead to epigenetically altered expression of Tlr1, Tlr2 and Lat, which may contribute to the inflammation in adipose tissue. Our study provides a potential mechanism for enhanced inflammation in adipose tissue under multigenerational HFD-fed stress.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Accession codes
References
Kelly T, Yang W, Chen CS, Reynolds K, He J . Global burden of obesity in 2005 and projections to 2030. Int J Obes (Lond) 2008; 32: 1431–1437.
Wang YC, McPherson K, Marsh T, Gortmaker SL, Brown M . Health and economic burden of the projected obesity trends in the USA and the UK. Lancet 2011; 378: 815–825.
Barker DJ . The origins of the developmental origins theory. J Intern Med 2007; 261: 412–417.
Muhlhausler BS, Ong ZY . The fetal origins of obesity: early origins of altered food intake. Endocr Metab Immune Disord Drug Targets 2011; 11: 189–197.
Vucetic Z, Kimmel J, Totoki K, Hollenbeck E, Reyes TM . Maternal high-fat diet alters methylation and gene expression of dopamine and opioid-related genes. Endocrinology 2010; 151: 4756–4764.
Sullivan EL, Smith MS, Grove KL . Perinatal exposure to high-fat diet programs energy balance, metabolism and behavior in adulthood. Neuroendocrinology 2011; 93: 1–8.
Li J, Huang J, Li JS, Chen H, Huang K, Zheng L . Accumulation of endoplasmic reticulum stress and lipogenesis in the liver through generational effects of high fat diets. J Hepatol 2012; 56: 900–907.
Takasaki M, Honma T, Yanaka M, Sato K, Shinohara N, Ito J et al. Continuous intake of a high-fat diet beyond one generation promotes lipid accumulation in liver and white adipose tissue of female mice. J Nutr Biochem 2011; 23: 640–645.
Massiera F, Barbry P, Guesnet P, Joly A, Luquet S, Moreilhon-Brest C et al. A Western-like fat diet is sufficient to induce a gradual enhancement in fat mass over generations. J Lipid Res 2010; 51: 2352–2361.
Matsuzawa Y, Shimomura I, Nakamura T, Keno Y, Tokunaga K . Pathophysiology and pathogenesis of visceral fat obesity. Diabetes Res Clin Pract 1994; 24 (Suppl): S111–S116.
Hamdy O, Porramatikul S, Al-Ozairi E . Metabolic obesity: the paradox between visceral and subcutaneous fat. Curr Diabetes Rev 2006; 2: 367–373.
Chen HC, Farese RV Jr . Determination of adipocyte size by computer image analysis. J Lipid Res 2002; 43: 986–989.
Chen H, Zheng C, Zhang X, Li J, Zheng L, Huang K . Apelin alleviates diabetes-associated endoplasmic reticulum stress in the pancreas of Akita mice. Peptides 2011; 32: 1634–1639.
Kosteli A, Sugaru E, Haemmerle G, Martin JF, Lei J, Zechner R et al. Weight loss and lipolysis promote a dynamic immune response in murine adipose tissue. J Clin Invest 2010; 120: 3466–3479.
Peng X, Wood CL, Blalock EM, Chen KC, Landfield PW, Stromberg AJ . Statistical implications of pooling RNA samples for microarray experiments. BMC Bioinformatics 2003; 4: 26.
Bruce KD, Cagampang FR, Argenton M, Zhang J, Ethirajan PL, Burdge GC et al. Maternal high-fat feeding primes steatohepatitis in adult mice offspring, involving mitochondrial dysfunction and altered lipogenesis gene expression. Hepatology 2009; 50: 1796–1808.
Cedar H, Bergman Y, Linking DNA . methylation and histone modification: patterns and paradigms. Nat Rev Genet 2009; 10: 295–304.
Huang da W, Sherman BT, Lempicki RA . Systematic and integrative analysis of large gene lists using DAVID bioinformatics resources. Nat Protoc 2009; 4: 44–57.
Wang LL, Chen H, Huang K, Zheng L . Elevated histone acetylations in Muller cell contribute to inflammation: a novel inhibitory effect of minocycline. Glia 2012; 60: 1896–1905.
Rozen S, Skaletsky H . Primer3 on the WWW for general users and for biologist programmers. Methods Mol Biol 2000; 132: 365–386.
Barber RD, Harmer DW, Coleman RA, Clark BJ . GAPDH as a housekeeping gene: analysis of GAPDH mRNA expression in a panel of 72 human tissues. Physiol Genomics 2005; 21: 389–395.
Schmittgen TD, Livak KJ . Analyzing real-time PCR data by the comparative C(T) method. Nat Protoc 2008; 3: 1101–1108.
Li LC, Dahiya R . MethPrimer: designing primers for methylation PCRs. Bioinformatics 2002; 18: 1427–1431.
Murano I, Barbatelli G, Parisani V, Latini C, Muzzonigro G, Castellucci M et al. Dead adipocytes, detected as crown-like structures, are prevalent in visceral fat depots of genetically obese mice. J Lipid Res 2008; 49: 1562–1568.
Gordon S, Hamann J, Lin HH, Stacey M . F4/80 and the related adhesion-GPCRs. Eur J Immunol 2011; 41: 2472–2476.
Balagopalan L, Ashwell BA, Bernot KM, Akpan IO, Quasba N, Barr VA et al. Enhanced T-cell signaling in cells bearing linker for activation of T-cell (LAT) molecules resistant to ubiquitylation. Proc Natl Acad Sci USA 2011; 108: 2885–2890.
Wardle J, Sanderson S, Guthrie CA, Rapoport L, Plomin R . Parental feeding style and the inter-generational transmission of obesity risk. Obes Res 2002; 10: 453–462.
Hong J, Stubbins RE, Smith RR, Harvey AE, Nunez NP . Differential susceptibility to obesity between male, female and ovariectomized female mice. Nutr J 2009; 8: 11.
Stubbins RE, Najjar K, Holcomb VB, Hong J, Nunez NP . Oestrogen alters adipocyte biology and protects female mice from adipocyte inflammation and insulin resistance. Diabetes Obes Metab 2012; 14: 58–66.
Medrikova D, Jilkova ZM, Bardova K, Janovska P, Rossmeisl M, Kopecky J . Sex differences during the course of diet-induced obesity in mice: adipose tissue expandability and glycemic control. Int J Obes (Lond) 2012; 36: 262–272.
Bayol SA, Simbi BH, Bertrand JA, Stickland NC . Offspring from mothers fed a 'junk food' diet in pregnancy and lactation exhibit exacerbated adiposity that is more pronounced in females. J Physiol 2008; 586: 3219–3230.
Khan IY, Taylor PD, Dekou V, Seed PT, Lakasing L, Graham D et al. Gender-linked hypertension in offspring of lard-fed pregnant rats. Hypertension 2003; 41: 168–175.
Olefsky JM, Glass CK . Macrophages, inflammation, and insulin resistance. Annu Rev Physiol 2010; 72: 219–246.
Franchi L, Eigenbrod T, Munoz-Planillo R, Nunez G . The inflammasome: a caspase-1-activation platform that regulates immune responses and disease pathogenesis. Nat Immunol 2009; 10: 241–247.
Vandanmagsar B, Youm YH, Ravussin A, Galgani JE, Stadler K, Mynatt RL et al. The NLRP3 inflammasome instigates obesity-induced inflammation and insulin resistance. Nat Med 2011; 17: 179–188.
Kuo LH, Tsai PJ, Jiang MJ, Chuang YL, Yu L, Lai KT et al. Toll-like receptor 2 deficiency improves insulin sensitivity and hepatic insulin signalling in the mouse. Diabetologia 2011; 54: 168–179.
Shi H, Kokoeva MV, Inouye K, Tzameli I, Yin H, Flier JS . TLR4 links innate immunity and fatty acid-induced insulin resistance. J Clin Invest 2006; 116: 3015–3025.
Feingold KR, Moser A, Shigenaga JK, Grunfeld C . Inflammation inhibits the expression of phosphoenolpyruvate carboxykinase in liver and adipose tissue. Innate Immun 2011; 18: 231–240.
Cinti S . Transdifferentiation properties of adipocytes in the adipose organ. Am J Physiol Endocrinol Metab 2009; 297: E977–E986.
Cinti S . The adipose organ. Prostaglandins Leukot Essent Fatty Acids 2005; 73: 9–15.
Cinti S . The role of brown adipose tissue in human obesity. Nutr Metab Cardiovasc Dis 2006; 16: 569–574.
Ozanne SE, Constancia M . Mechanisms of disease: the developmental origins of disease and the role of the epigenotype. Nat Clin Pract Endocrinol Metab 2007; 3: 539–546.
Ng SF, Lin RC, Laybutt DR, Barres R, Owens JA, Morris MJ . Chronic high-fat diet in fathers programs beta-cell dysfunction in female rat offspring. Nature 2010; 467: 963–966.
Carone BR, Fauquier L, Habib N, Shea JM, Hart CE, Li R et al. Paternally induced transgenerational environmental reprogramming of metabolic gene expression in mammals. Cell 2010; 143: 1084–1096.
Cropley JE, Suter CM, Beckman KB, Martin DI . Germ-line epigenetic modification of the murine A vy allele by nutritional supplementation. Proc Natl Acad Sci USA 2006; 103: 17308–17312.
Anway MD, Cupp AS, Uzumcu M, Skinner MK . Epigenetic transgenerational actions of endocrine disruptors and male fertility. Science 2005; 308: 1466–1469.
Sharma S, Kelly TK, Jones PA . Epigenetics in cancer. Carcinogenesis 2010; 31: 27–36.
Acknowledgements
We thank Bo Yang (College of Life Sciences, Wuhan University, Wuhan, People’s Republic of China) for technical assistance with the heat map and Professor Xin Chen (UCSF, USA) for critical reading the manuscript. This work was supported by the National Basic Research Program of China (2009BC918304 and 2012CB524901), the Natural Science Foundation of China (Nos. 30970607, 81172971, 81222043 and 31271370) and the Program for New Century Excellent Talents in University (NECT10-0623 and NECT11-0170).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies this paper on International Journal of Obesity website
Supplementary information
Rights and permissions
About this article
Cite this article
Ding, Y., Li, J., Liu, S. et al. DNA hypomethylation of inflammation-associated genes in adipose tissue of female mice after multigenerational high fat diet feeding. Int J Obes 38, 198–204 (2014). https://doi.org/10.1038/ijo.2013.98
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ijo.2013.98
Keywords
This article is cited by
-
Developmental programming of offspring adipose tissue biology and obesity risk
International Journal of Obesity (2021)
-
Epigenetic signatures underlying inflammation: an interplay of nutrition, physical activity, metabolic diseases, and environmental factors for personalized nutrition
Inflammation Research (2021)
-
Prolonged high-fat diet induces gradual and fat depot-specific DNA methylation changes in adult mice
Scientific Reports (2017)
-
Epigenetic regulation of ASC/TMS1 expression: potential role in apoptosis and inflammasome function
Cellular and Molecular Life Sciences (2014)
-
Maternal Nutrition and Epigenetic Perturbation: Modeling Trends to Translation
Current Pediatrics Reports (2013)