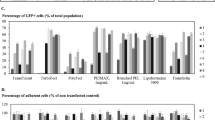
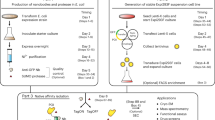
Abstract
Lentivirus can be engineered to be a highly potent vector for gene therapy applications. However, generation of clinical grade vectors in enough quantities for therapeutic use is still troublesome and limits the preclinical and clinical experiments. As a first step to solve this unmet need we recently introduced a baculovirus-based production system for lentiviral vector (LV) production using adherent cells. Herein, we have adapted and optimized the production of these vectors to a suspension cell culture system using recombinant baculoviruses delivering all elements required for a safe latest generation LV preparation. High-titer LV stocks were achieved in 293T cells grown in suspension. Produced viruses were accurately characterized and the functionality was also tested in vivo. Produced viruses were compared with viruses produced by calcium phosphate transfection method in adherent cells and polyethylenimine transfection method in suspension cells. Furthermore, a scalable and cost-effective capture purification step was developed based on a diethylaminoethyl monolithic column capable of removing most of the baculoviruses from the LV pool with 65% recovery.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Naldini L, Blomer U, Gallay P, Ory D, Mulligan R, Gage FH et al. In vivo gene delivery and stable transduction of nondividing cells by a lentiviral vector. Science 1996; 272: 263–267.
Burns JC, Friedmann T, Driever W, Burrascano M, Yee JK . Vesicular stomatitis virus G glycoprotein pseudotyped retroviral vectors: concentration to very high titer and efficient gene transfer into mammalian and nonmammalian cells. Proc Natl Acad Sci USA 1993; 90: 8033–8037.
Ory DS, Neugeboren BA, Mulligan RC . A stable human-derived packaging cell line for production of high titer retrovirus/vesicular stomatitis virus G pseudotypes. Proc Natl Acad Sci USA 1996; 93: 11400–11406.
Miyoshi H, Blomer U, Takahashi M, Gage FH, Verma IM . Development of a self-inactivating lentivirus vector. J Virol 1998; 72: 8150–8157.
Dull T, Zufferey R, Kelly M, Mandel RJ, Nguyen M, Trono D et al. A third-generation lentivirus vector with a conditional packaging system. J Virol 1998; 72: 8463–8471.
Follenzi A, Naldini L . Generation of HIV-1 derived lentiviral vectors. Methods Enzymol 2002; 346: 454–465.
Kutner RH, Zhang XY, Reiser J . Production, concentration and titration of pseudotyped HIV-1-based lentiviral vectors. Nat Protoc 2009; 4: 495–505.
Tiscornia G, Singer O, Verma IM . Production and purification of lentiviral vectors. Nat Protoc 2006; 1: 241–245.
Koldej R, Cmielewski P, Stocker A, Parsons DW, Anson DS . Optimisation of a multipartite human immunodeficiency virus based vector system; control of virus infectivity and large-scale production. J Gene Med 2005; 7: 1390–1399.
Kuroda H, Kutner RH, Bazan NG, Reiser J . Simplified lentivirus vector production in protein-free media using polyethylenimine-mediated transfection. J Virol Methods 2009; 157: 113–121.
Geraerts M, Michiels M, Baekelandt V, Debyser Z, Gijsbers R . Upscaling of lentiviral vector production by tangential flow filtration. J Gene Med 2005; 7: 1299–1310.
Slepushkin V, Chang N, Cohen R, Gan Y, Jiang B, Deausen D et al. Large-scale purification of a lentiviral vector by size exclusion chromatography or mustang Q ion exchange capsule. BioProcessing J 2003; 2: 89–95.
Negrete A, Kotin RM . Production of recombinant adeno-associated vectors using two bioreactor configurations at different scales. J Virol Methods 2007; 145: 155–161.
Segura MM, Garnier A, Durocher Y, Coelho H, Kamen A . Production of lentiviral vectors by large-scale transient transfection of suspension cultures and affinity chromatography purification. Biotechnol Bioeng 2007; 98: 789–799.
Godbey WT, Wu KK, Mikos AG . Poly(ethylenimine)-mediated gene delivery affects endothelial cell function and viability. Biomaterials 2001; 22: 471–480.
Airenne KJ, Mahonen AJ, Laitinen OH, Ylä-Herttuala S . Baculovirus-mediated gene transfer: an emerging universal concept. In: Smyth Templeton N (ed). Gene and Cell Therapy. CRC Press, Taylor & Francis Group: Boca Raton, 2009, pp 263–292.
Kost TA, Condreay JP, Jarvis DL . Baculovirus as versatile vectors for protein expression in insect and mammalian cells. Nat Biotechnol 2005; 23: 567–575.
Airenne KJ, Makkonen KE, Mahonen AJ, Ylä-Herttuala S . In vivo application and tracking of baculovirus. Curr Gene Ther 2010; 10: 187–194.
Sollerbrant K, Elmen J, Wahlestedt C, Acker J, Leblois-Prehaud H, Latta-Mahieu M et al. A novel method using baculovirus-mediated gene transfer for production of recombinant adeno-associated virus vectors. J Gen Virol 2001; 82: 2051–2060.
Huang KS, Lo WH, Chung YC, Lai YK, Chen CY, Chou ST et al. Combination of baculovirus-mediated gene delivery and packed-bed reactor for scalable production of adeno-associated virus. Hum Gene Ther 2007; 18: 1161–1170.
Lesch HP, Turpeinen S, Niskanen EA, Mahonen AJ, Airenne KJ, Ylä-Herttuala S . Generation of lentivirus vectors using recombinant baculoviruses. Gene Therapy 2008; 15: 1280–1286.
Yamada K, McCarty DM, Madden VJ, Walsh CE . Lentivirus vector purification using anion exchange HPLC leads to improved gene transfer. Biotechniques 2003; 34: 1074–1078, 1080.
Sena-Esteves M, Tebbets JC, Steffens S, Crombleholme T, Flake AW . Optimized large-scale production of high titer lentivirus vector pseudotypes. J Virol Methods 2004; 122: 131–139.
Reiser J . Production and concentration of pseudotyped HIV-1-based gene transfer vectors. Gene Therapy 2000; 7: 910–913.
Miyake K, Miyake N, Shimada T . Development of targeted gene transfer into human primary T lymphocytes and macrophages using high-titer recombinant HIV vectors. J Biotechnol 2007; 129: 532–538.
Baekelandt V, Eggermont K, Michiels M, Nuttin B, Debyser Z . Optimized lentiviral vector production and purification procedure prevents immune response after transduction of mouse brain. Gene Therapy 2003; 10: 1933–1940.
Rodrigues T, Carvalho A, Carmo M, Carrondo MJ, Alves PM, Cruz PE . Scaleable purification process for gene therapy retroviral vectors. J Gene Med 2007; 9: 233–243.
Condreay JP, Witherspoon SM, Clay WC, Kost TA . Transient and stable gene expression in mammalian cells transduced with a recombinant baculovirus vector. Proc Natl Acad Sci USA 1999; 96: 127–132.
Hsu CS, Ho YC, Wang KC, Hu YC . Investigation of optimal transduction conditions for baculovirus-mediated gene delivery into mammalian cells. Biotechnol Bioeng 2004; 88: 42–51.
Cronin J, Zhang XY, Reiser J . Altering the tropism of lentiviral vectors through pseudotyping. Curr Gene Ther 2005; 5: 387–398.
Ansorge S, Henry O, Kamen A . Recent progress in lentiviral vector mass production. Biochem Eng J 2010; 48: 362–377.
Vicente T, Peixoto C, Carrondo MJ, Alves PM . Purification of recombinant baculoviruses for gene therapy using membrane processes. Gene Therapy 2009; 16: 766–775.
Jungbauer A, Hahn R . Polymethacrylate monoliths for preparative and industrial separation of biomolecular assemblies. J Chromatogr A 2008; 1184: 62–79.
Etzel MR, Riordan WT . Viral clearance using monoliths. J Chromatogr A 2009; 1216: 2621–2624.
Kutner RH, Puthli S, Marino MP, Reiser J . Simplified production and concentration of HIV-1-based lentiviral vectors using HYPERFlask vessels and anion exchange membrane chromatography. BMC Biotechnol 2009; 9: 10.
Coleman JE, Huentelman MJ, Kasparov S, Metcalfe BL, Paton JF, Katovich MJ et al. Efficient large-scale production and concentration of HIV-1-based lentiviral vectors for use in vivo. Physiol Genomics 2003; 12: 221–228.
Karolewski BA, Watson DJ, Parente MK, Wolfe JH . Comparison of transfection conditions for a lentivirus vector produced in large volumes. Hum Gene Ther 2003; 14: 1287–1296.
Harper DM, Franco EL, Wheeler C, Ferris DG, Jenkins D, Schuind A et al. Efficacy of a bivalent L1 virus-like particle vaccine in prevention of infection with human papillomavirus types 16 and 18 in young women: a randomised controlled trial. Lancet 2004; 364: 1757–1765.
Ansorge S, Lanthier S, Transfiguracion J, Durocher Y, Henry O, Kamen A . Development of a scalable process for high-yield lentiviral vector production by transient transfection of HEK293 suspension cultures. J Gene Med 2009; 11: 868–876.
Dukkipati A, Park HH, Waghray D, Fischer S, Garcia KC . BacMam system for high-level expression of recombinant soluble and membrane glycoproteins for structural studies. Protein Expr Purif 2008; 62: 160–170.
Mahonen AJ, Makkonen KE, Laakkonen JP, Ihalainen TO, Kukkonen SP, Kaikkonen MU et al. Culture medium induced vimentin reorganization associates with enhanced baculovirus-mediated gene delivery. J Biotechnol 2010; 145: 111–119.
Baekelandt V, Claeys A, Eggermont K, Lauwers E, De SB, Nuttin B et al. Characterization of lentiviral vector-mediated gene transfer in adult mouse brain. Hum Gene Ther 2002; 13: 841–853.
Rodrigues T, Carrondo MJ, Alves PM, Cruz PE . Purification of retroviral vectors for clinical application: biological implications and technological challenges. J Biotechnol 2007; 127: 520–541.
Vicente T, Peixoto C, Alves PM, Carrondo MJ . Modeling electrostatic interactions of baculovirus vectors for ion-exchange process development. J Chromatogr A 2010; 1217: 3754–3764.
Tran NT, Taverna M, Chevalier M, Ferrier D . One-step capillary isoelectric focusing for the separation of the recombinant human immunodeficiency virus envelope glycoprotein glycoforms. J Chromatogr A 2000; 866: 121–135.
Airenne KJ, Hiltunen MO, Turunen MP, Turunen AM, Laitinen OH, Kulomaa MS et al. Baculovirus-mediated periadventitial gene transfer to rabbit carotid artery. Gene Therapy 2000; 7: 1499–1504.
Kaikkonen MU, Lesch HP, Pikkarainen J, Raty JK, Vuorio T, Huhtala T et al. (Strept) avidin-displaying lentiviruses as versatile tools for targeting and dual imaging of gene delivery. Gene Therapy 2009; 16: 894–904.
Rodrigues AF, Carmo M, Alves PM, Coroadinha AS . Retroviral vector production under serum deprivation: the role of lipids. Biotechnol Bioeng 2009; 104: 1171–1181.
Acknowledgements
We thank Joonas Malinen, Tarja Taskinen, Jaana Siponen, Siiri Väistö, Juha Ruuskanen, Anne Martikainen and Anneli Miettinen for excellent technical assistance. This work was supported by grants from Finnish Academy, the European Union (Baculogenes, LSH-2005-1.4.4.6 and Clinigene Network of Excellence, LSHB-CT-2006-018933), Portuguese Fundação para a Ciência e Tecnologia (PTDC/EQU-EQU/71645/2006 and SFRH/BD/31257/2006) and Ark Therapeutics Group plc.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
HPL, JTP, HS and LL are employees of Ark Therapeutics Ltd. CP, TV, PMA and MJTC are employees of IBET.
Rights and permissions
About this article
Cite this article
Lesch, H., Laitinen, A., Peixoto, C. et al. Production and purification of lentiviral vectors generated in 293T suspension cells with baculoviral vectors. Gene Ther 18, 531–538 (2011). https://doi.org/10.1038/gt.2010.162
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/gt.2010.162
Keywords
This article is cited by
-
Rapid isolation of lentivirus particles from cell culture media via a hydrophobic interaction chromatography method on a polyester, capillary-channeled polymer fiber stationary phase
Analytical and Bioanalytical Chemistry (2021)
-
Assessment of selected media supplements to improve F/HN lentiviral vector production yields
Scientific Reports (2017)
-
Transgene expression in Penaeus monodon cells: evaluation of recombinant baculoviral vectors with shrimp specific hybrid promoters
Cytotechnology (2016)
-
p21 and CK2 interaction-mediated HDAC2 phosphorylation modulates KLF4 acetylation to regulate bladder cancer cell proliferation
Tumor Biology (2016)
-
Efficient gene delivery into cell lines and stem cells using baculovirus
Nature Protocols (2014)