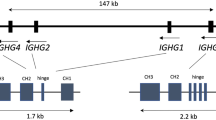
Abstract
We used next-generation sequencing to identify immunoglobulin heavy chain (IGH) genetic variation in two closely related hypertensive rat lines that differ in susceptibility to end-organ disease (SHR-A3 and SHR-B2). The two SHR lines differ extensively at the IGH locus from the rat reference genome sequence and from each other, creating 306 sequence unique IGH genes. Compared with IGH genes mapped in the rat reference genome sequence, 98 are null gene alleles (31 are null in both SHR lines, 45 are null in SHR-A3 only and 23 are null in SHR-B2 only). Of the 306 divergent gene sequences, 126 result in amino acid substitution and, among these, SHR-A3 and SHR-B2 differ from one another at the amino acid level in 96 segments. Twelve pseudogenes in the rat reference genome sequence had changes displacing the stop codon and creating probable functional genes in either or both SHR-A3 and SHR-B2. A further five alleles that encoded functional rat reference genome sequence genes or open reading frames were converted to pseudogenes in either or both SHR-A3 and SHR-B2. These studies reveal that the preimmune immunoglobulin repertoire is highly divergent among SHR lines differing in end-organ injury susceptibility and this may modify immune mechanisms in hypertensive renal injury.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 6 digital issues and online access to articles
$119.00 per year
only $19.83 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Eichler EE, Flint J, Gibson G, Kong A, Leal SM, Moore JH et al. Missing heritability and strategies for finding the underlying causes of complex disease. Nat Rev Genet 2010; 11: 446–450.
Manolio TA, Collins FS, Cox NJ, Goldstein DB, Hindorff LA, Hunter DJ et al. Finding the missing heritability of complex diseases. Nature 2009; 461: 747–753.
Watson CT, Steinberg KM, Huddleston J, Warren RL, Malig M, Schein J et al. Complete haplotype sequence of the human immunoglobulin heavy-chain variable, diversity, and joining genes and characterization of allelic and copy-number variation. Am J Hum Genet 2013; 92: 530–546.
Reveille JD . Genetics of spondyloarthritis—beyond the MHC. Nat Rev Rheumatol 2012; 8: 296–304.
Sollid LM, Markussen G, Ek J, Gjerde H, Vartdal F, Thorsby E . Evidence for a primary association of celiac disease to a particular HLA-DQ alpha/beta heterodimer. J Exp Med 1989; 169: 345–350.
Vandiedonck C, Knight JC . The human major histocompatibility complex as a paradigm in genomics research. Brief Funct Genomic Proteomic 2009; 8: 379–394.
Bunn HF . The triumph of good over evil: protection by the sickle gene against malaria. Blood 2013; 121: 20–25.
Genovese G, Friedman DJ, Ross MD, Lecordier L, Uzureau P, Freedman BI et al. Association of trypanolytic ApoL1 variants with kidney disease in African Americans. Science 2010; 329: 841–845.
Carlsson B, Kindberg E, Buesa J, Rydell GE, Lidon MF, Montava R et al. The G428A nonsense mutation in FUT2 provides strong but not absolute protection against symptomatic GII.4 Norovirus infection. PLoS ONE 2009; 4: e5593.
Mordes JP, Cort L, Norowski E, Leif J, Fuller JM, Lernmark A et al. Analysis of the rat Iddm14 diabetes susceptibility locus in multiple rat strains: identification of a susceptibility haplotype in the Tcrb-V locus. Mamm Genome 2009; 20: 162–169.
Tirabassi RS, Guberski DL, Blankenhorn EP, Leif JH, Woda BA, Liu Z et al. Infection with viruses from several families triggers autoimmune diabetes in LEW*1WR1 rats: prevention of diabetes by maternal immunization. Diabetes 2010; 59: 110–118.
Okamoto K, Yamori Y, Nagaoka A . Establishment of the stroke-prone spontaneously hypertensive rat (SHR). Circ Res 1974; 14 (Suppl I): I143–I153.
Herring SM, Gokul N, Monita M, Bell R, Boerwinkle E, Wenderfer SE et al. Immunoglobulin locus associates with serum IgG levels and albuminuria. J Am Soc Nephrol 2011; 22: 881–889.
Braun MC, Herring SM, Gokul N, Monita M, Bell R, Hicks MJ et al. Hypertensive renal disease: susceptibility and resistance in inbred hypertensive rat lines. J Hypertension 2013; 31: 2050–2059.
Atanur SS, Birol I, Guryev V, Hirst M, Hummel O, Morrissey C et al. The genome sequence of the spontaneously hypertensive rat: analysis and functional significance. Genome Res 2010; 20: 791–803.
Atanur SS, Diaz AG, Maratou K, Sarkis A, Rotival M, Game L et al. Genome sequencing reveals loci under artificial selection that underlie disease phenotypes in the laboratory rat. Cell 2013; 154: 691–703.
Guo X, Brenner M, Zhang X, Laragione T, Tai S, Li Y et al. Whole-genome sequences of DA and F344 rats with different susceptibilities to arthritis, autoimmunity, inflammation and cancer. Genetics 2013; 194: 1017–1028.
Baud A, Hermsen R, Guryev V, Stridh P, Graham D, McBride MW et al. Combined sequence-based and genetic mapping analysis of complex traits in outbred rats. Nat Genet 2013; 45: 767–775.
Bell R, Herring SM, Gokul N, Monita M, Grove ML, Boerwinkle E et al. High-resolution identity by descent mapping uncovers the genetic basis for blood pressure differences between spontaneously hypertensive rat lines. Circ Cardiovasc Genet 2011; 4: 223–231.
IMGT®, the International ImMunoGeneTics Information Systemhttp://www.imgt.org Accessed 2 February 2014.
Giudicelli V, Chaume D, Lefranc MP . IMGT/GENE-DB: a comprehensive database for human and mouse immunoglobulin and T cell receptor genes. Nucleic Acids Res 2005; 33 (Database issue): D256–D261.
Lefranc MP, Giudicelli V, Ginestoux C, Jabado-Michaloud J, Folch G, Bellahcene F et al. IMGT, the international ImMunoGeneTics information system. Nucleic Acids Res 2009; 37 (Database issue): D1006–D1012.
Guzik TJ, Hoch NE, Brown KA, McCann LA, Rahman A, Dikalov S et al. Role of the T cell in the genesis of angiotensin II induced hypertension and vascular dysfunction. J Exp Med 2007; 204: 2449–2460.
Crowley SD, Song YS, Lin EE, Griffiths R, Kim HS, Ruiz P . Lymphocyte responses exacerbate angiotensin II-dependent hypertension. Am J Physiol 2010; 298: R1089–R1097.
Barhoumi T, Kasal DA, Li MW, Shbat L, Laurant P, Neves MF et al. T regulatory lymphocytes prevent angiotensin II-induced hypertension and vascular injury. Hypertension 2011; 57: 469–476.
Hurn PD, Subramanian S, Parker SM, Afentoulis ME, Kaler LJ, Vandenbark AA et al. T- and B-cell-deficient mice with experimental stroke have reduced lesion size and inflammation. J Cereb Blood Flow Metab 2007; 27: 1798–1805.
Shichita T, Sugiyama Y, Ooboshi H, Sugimori H, Nakagawa R, Takada I et al. Pivotal role of cerebral interleukin-17-producing gammadeltaT cells in the delayed phase of ischemic brain injury. Nat Med 2009; 15: 946–950.
Jackman SD, Birol I . Assembling genomes using short-read sequencing technology. Genome Biol 2010; 11: 202.
Alkan C, Sajjadian S, Eichler EE . Limitations of next-generation genome sequence assembly. Nat Meth 2011; 8: 61–65.
Lefranc M-P, Lefranc G . The Immunoglobulin FactsBook. Academic Press: London, UK, 2001.
Raposo B, Dobritzsch D, Ge C, Ekman D, Xu B, Lindh I et al. Epitope-specific antibody response is controlled by immunoglobulin VH polymorphisms. J Exp Med 2014; 211: 405–411.
Jiang N, He J, Weinstein JA, Penland L, Sasaki S, He XS et al. Lineage structure of the human antibody repertoire in response to influenza vaccination. Sci Transl Med 2013; 5: 171ra19.
Bell R, Herring SM, Gokul N, Monita M, Grove ML, Boerwinkle E et al. High resolution identity by descent mapping uncovers the genetic basis for blood pressure differences between SHR lines. Circ Cardiovasc Genet 2011; 4: 223–231.
Novoalign software. http://www.novocraft.com Accessed 19 September 2013.
SAMtools software. http://samtools.sourceforge.net/ Accessed 19 September 2013.
Picard software http://picard.sourceforge.net/ Accessed 19 September 2013.
McKenna A, Hanna M, Banks E, Sivachenko A, Cibulskis K, Kernytsky A et al. The Genome Analysis Toolkit: a MapReduce framework for analyzing next-generation DNA sequencing data. Genome Res 2010; 20: 1297–1303.
Cingolani P, Platts A, Wang le L, Coon M, Nguyen T, Wang L et al. A program for annotating and predicting the effects of single nucleotide polymorphisms, SnpEff: SNPs in the genome of Drosophila melanogaster strain w1118; iso-2; iso-3. Fly 2012; 6: 80–92.
Thorvaldsdottir H, Robinson JT, Mesirov JP . Integrative Genomics Viewer (IGV): high-performance genomics data visualization and exploration. Brief Bioinform 2013; 14: 178–192.
Kent WJ . BLAT–the BLAST-like alignment tool. Genome Res 2002; 12: 656–664.
Artimo P, Jonnalagedda M, Arnold K, Baratin D, Csardi G, de Castro E et al. ExPASy: SIB bioinformatics resource portal. Nucleic Acids Res 2012; 40 (Web Server issue): W597–W603.
Acknowledgements
The work reported has been supported by grants from NIH (R01DK069632-05 and R01DK081866) to PAD. PAD is grateful for additional endowed support from the Cullen Chair in Molecular Medicine at the University of Texas HSC Houston that provided support for rat genomic DNA sequencing.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies this paper on Genes and Immunity website
Rights and permissions
About this article
Cite this article
Gonzalez-Garay, M., Cranford, S., Braun, M. et al. Diversity in the preimmune immunoglobulin repertoire of SHR lines susceptible and resistant to end-organ injury. Genes Immun 15, 528–533 (2014). https://doi.org/10.1038/gene.2014.40
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/gene.2014.40
This article is cited by
-
Adaptive Immunity in Hypertension
Current Hypertension Reports (2019)
-
Sequencing of the human IG light chain loci from a hydatidiform mole BAC library reveals locus-specific signatures of genetic diversity
Genes & Immunity (2015)